-
PDF
- Split View
-
Views
-
Cite
Cite
Jenny A. Cadée, Chou-Yue Chang, Chien-Wei Chen, Chien-Ning Huang, Su-Lin Chen, Chin-Kun Wang, Bovine Casein Hydrolysate (C12 Peptide) Reduces Blood Pressure in Prehypertensive Subjects, American Journal of Hypertension, Volume 20, Issue 1, January 2007, Pages 1–5, https://doi.org/10.1016/j.amjhyper.2006.06.005
Close - Share Icon Share
Abstract
About one in four adults suffer from prehypertension. People with prehypertension are at risk of developing hypertension, being a biomarker for cardiovascular disease risk. The use of milk-derived protein hydrolysates containing peptides with angiotensin-converting enzyme (ACE) inhibiting properties may reduce blood pressure (BP) and thus the risk of developing hypertension.
We investigated the BP-lowering effect of a casein-derived protein hydrolysate (C12 Peptide) during a 4-week intervention period in prehypertensive subjects. After a 2-week run-in period, 48 Taiwanese volunteers were randomly assigned to either placebo or C12 Peptide tablets for 4 weeks, followed by a 2-week off-treatment period. After the run-in period, BP was measured weekly.
Baseline values for systolic BP (mean ± SEM) in the placebo and C12 Peptide groups were 137.1 ± 3.1 and 137.9 ± 2.4 mm Hg, respectively; those for diastolic BP were 85.2 ± 2.1 and 86.9 ± 2.0 mm Hg, respectively. Four weeks repeated daily intake of 3.8 g C12 Peptide reduced significantly systolic and diastolic BP by 10.7 ± 1.6 mm Hg and 6.9 ± 1.2 mm Hg, respectively, compared to baseline. Furthermore, plasma angiotensin II and aldosterone levels were reduced significantly (P < .05). The placebo group showed a BP reduction of 3.6 ± 2.4 and 2.7 ± 1.6 mm Hg in systolic and diastolic BP, respectively (P = not significant). No evidence of side effects was observed.
This study shows that C12 Peptide reduces BP in prehypertensive people. Am J Hypertens 2007;20: 1–5 © 2007 American Journal of Hypertension, Ltd.
Hypertension is known as the “silent killer” because it usually does not produce symptoms until severe damage is already done. Worldwide, hypertension causes 7 million deaths, about 13% of the global total.1 It is the number one cause of strokes and can cause heart failure, increased peripheral arteriolar stiffness, and kidney damage. About one in four adults suffers from hypertension and the same ratio is observed for prehypertension, people who are at high risk of developing hypertension.2
In the renin-angiotensin system, angiotensin-converting enzyme (ACE) plays an important role in blood pressure (BP) regulation. It catalyzes the production of a strong vasoconstrictor (angiotensin II) and the inactivation of the vasodilator bradykinin. Inhibiting these enzymes, controlled reactions by specific ACE inhibitors, will reduce BP.
Several food-derived ACE inhibitors have been identified3–10 as potential known antihypertensive agents. Milk proteins, both caseins and whey proteins, are a rich source of ACE inhibiting peptides.11–14 Several spontaneously hypertensive rat studies and a limited number of human studies showed that these so-called casokinins and lactokinins have antihypertensive effects.15–19 Among the casokinins, a bovine casein hydrolysate C12 Peptide has been found having strong in vitro ACE inhibitory activity as well as antihypertensive effects in both animals and humans.15,18,19 This hydrolysate consists of various peptides and C12 refers to the peptide consisting of 12 amino acids having ACE inhibitory activity as well as antihypertensive effects in rodents.15 In addition to C12, other ACE inhibiting peptides are present such as C7 and C6,15 which may contribute to the antihypertensive effect of C12 Peptide in humans. Due to C12 Peptide's antihypertensive effect in humans and its applicability in dietary supplements and functional foods such as beverages, chews, and fermented dairy products, C12 Peptide is a very interesting and promising ingredient to lower BP.
Recently, Townsend et al19 showed that a single administration of C12 Peptide tablets reduced BP in mildly hypertensive people within 4 to 6 hours. The present study investigates the efficacy and safety of a 4-week repeated daily administration of C12 Peptide to prehypertensive volunteers using a double-blind, placebo-controlled, randomized parallel group design.
Methods
Study Population
The procedures followed were in accordance with the ethical standards of Chung Shan Medical University hospital. After approval of the protocol by the Institutional Review Board (IRB) committee of Chung Shan Medical University hospital, all subjects gave written informed consent before starting the study.
Subjects were Taiwanese men and women of at least 32 years of age. Subjects were eligible if they had prehypertensive BP (120 < systolic BP ≤ 140 mm Hg or 80 < diastolic BP ≤ 90 mm Hg) at their first visit and were not on antihypertensive medications. Subjects were excluded if in the past 6 months they had experienced liver, kidney, or heart related diseases such as stroke and transient ischemic attack, had uncontrolled diabetes mellitus (blood glucose >200 mg/dL), milk allergy, melancholy, hemoglobin level <11 g/dL (men) or <10 g/dL (women), had alcoholism, were pregnant or planned to become pregnant during the study, and were lactose intolerant.
Tablets
Tablets were prepared offsite by Uni-President Enterprises Corp. (Tainan, Taiwan). Placebo tablet contained 640 mg of microcrystalline cellulose, 10 mg of magnesium stearate, 0.09 mg of FD&C Blue No. 1 Aluminum lake, and 40 mg of hydroxypropyl methylcellulose. A C12 Peptide tablet contained 633 mg of C12 Peptide, 9.6 mg of lubricants, 0.09 mg of FD&C Blue No. 1 Aluminum lake, and 40 mg of hydroxypropyl methylcellulose. The C12 Peptide is commercially available from DMV International, The Netherlands.
High Performance Liquid Chromatography
Tablets containing C12 Peptide were analyzed by reversed phase high performance liquid chromatography (HPLC; analytical column Lichrospher C18e, 5 μm, 4.6 mm by 250 mm [Merck, Darmstadt, Germany] with a Waters system [Waters 2690 pump and Waters 2996 pda detector, Boston]). At random, a tablet was made into powder with mortar and pestle and repeated ten times. The 10 powder samples were dissolved separately in water to a final concentration of 1 mg/mL and subsequently placed for 1 h in an ultrasonic bath. After filtration using a 0.45-μm filter, 20 μL of the solution was injected onto the column. A linear gradient was run from 100% A (water; 0.1% trifluoracid) to 45% A and 55% B (acetonitril; 0.1% trifluoracid) for 25 min. The flow rate was 0.8 mL/min and the column oven was set at 37°C. The UV detection at a wavelength of 260 nm was applied. Peak areas were determined with Waters Millennium 32 software and resulted in an average content of C12 per tablet of 39.9 mg ± 3.2 mg (average ± SD). This is equal to an average C12 Peptide content of 633 mg per tablet by taken into account that the produced C12 Peptide batch contained 6.3% C12. Therefore the daily intake in the C12 Peptide group was on average 3.8 g of C12 Peptide.
Study Design
During the first (screening) visit, all subjects came into an air-conditioned room. The protocol was reviewed and written informed consent was obtained. After 30 min of rest, BP was measured (mercurial sphygmomanometer, ENNE, E-300; Jen Kuo Inc., Taiwan) in the sitting position (right arm), three times at 2-min intervals. During the whole study, BP of each subject was measured by the same nurse. Body height and weight were measured as well. Electrocardiogram (ECG), physical examination, and basic clinical chemistry, hematology, and urine testing were performed to determine study eligibility as well as whether serious psychological or medical illness was present. When subjects were eligible for inclusion in the study, they were invited back for a baseline visit.
After enrollment, the study commenced with a single blind run-in period of 2 weeks in which all subjects took three placebo tablets, in the morning before breakfast and another three placebo tablets after dinner. The subjects were randomly assigned to a double-blind intervention period of 4 weeks with either three placebo tablets, twice a day, or three tablets containing in total of 1.9 g of C12 Peptide, twice a day (before breakfast and after dinner). The intervention period was followed by a 2-week off-treatment period.
During the intervention and off-treatment period, the subjects visited the clinic weekly for BP and weight measurements. At each visit, subjects received a new bottle of pills and old bottles were returned. At the end of both the run-in period and the 4-week intervention period, baseline values of BP and body weight were measured and blood samples were drawn and urine samples were collected after an overnight fasting. At the end of the intervention period an ECG was taken.
During the whole trial, no restrictions in dietary and physical activity were given.
Biochemical Indices
Fasting (12 h overnight) blood and urine samples were collected at baseline and at the end of the 4-week intervention period. Blood samples were analyzed for serum fat, total cholesterol, HDL cholesterol, triglycerides, blood urea nitrogen, serum creatinine, glutamate oxaloacetate transaminase glutamate pyruvate transaminase, plasma aldosterone,20 plasma angiotensin I,21 and angiotensin II22 levels. The LDL cholesterol was calculated by using the converting equation (LDL cholesterol = Total cholesterol − [HDL cholesterol + 1/5 triglyceride]).
An urine reagent test strip (Bayer Inc., Berkshire, UK) was used to evaluate whether the levels of glucose, protein, pH, blood, ketone, bilirubin, nitrite, urobilinogen, and specific gravity were within the normal range.
Statistical Analysis
Data were analyzed by SPSS statistical software (V10.07C for window, SPSS Inc., Chicago, IL) using two-way analysis of variance with treatment and time as grouping variables. Paired t test was used to evaluate the changes in BP and safety parameters compared to baseline.
Results
Subjects
In total 168 subjects were screened, of which 54 subjects fulfilled the enrollment criteria. During the run-in period, six subjects dropped out due to personal reasons or too difficult to comply with the study demands. In total 48 subjects were randomly assigned to either placebo or C12 Peptide treatment for 4 weeks and finished this study completely. The subjects in the placebo group were 55.6 ± 2.4 years old, had a body weight of 62.7 ± 1.8 kg, a BMI of 25.4 ± 0.6 kg/m2 and a height of 157.0 ± 6.3 cm. The group consisted of four male and 20 female. The subjects of the C12 Peptide group were 56.1 ± 2.3 year old, had a body weight of 62.8 ± 2.5 kg, a body mass index (BMI) of 24.5 ± 0.6 kg/m2, and a height of 159.7 ± 9.3 cm. The group consisted of 6 men and 18 women.
No significant differences were observed between the baseline characteristics of the placebo and C12 Peptide groups.
BP Changes
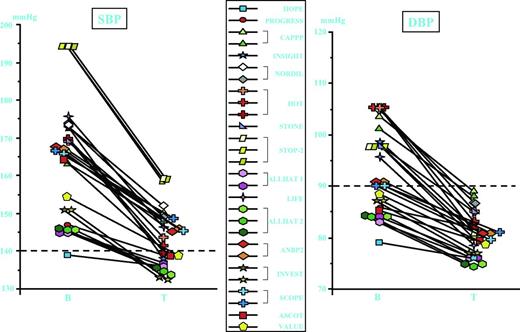
The baseline values for systolic BP in the placebo and C12 Peptide groups were (mean ± SEM) 137.1 ± 3.1 and 137.9 ± 2.4 mm Hg, respectively; those for diastolic BP were 85.2 ± 2.1 and 86.9 ± 2.0 mm Hg, respectively. Figs. 1 and 2 depict weekly measurements of systolic and diastolic DBP during 4 weeks of daily administration of either placebo or C12 Peptide tablets to prehypertensive subjects, respectively.
Effect of 4 weeks of repeated daily administration (solid lines) of placebo tablets (■) or C12 Peptide tablets (•; 3.8 g/d) on systolic blood pressure (SBP) of prehypertensive subjects, followed by a 2-week off-treatment period (dotted lines). Values are mean ± SEM. *P < .05 compared to baseline.
Effect of 4 weeks of repeated daily administration (solid lines) of placebo tablets (■) or C12 Peptide tablets (•; 3.8 g/d) on diastolic blood pressure (DBP) of prehypertensive subjects, followed by a 2-week off-treatment period (dotted lines). Values are mean ± SEM. *P < .05 compared to baseline.
One week of repeated administration of 3.8 g of C12 Peptide/day already reduced systolic and diastolic BP significantly by (mean ± SEM) 9.7 ± 2.5 and 5.9 ± 1.9 mm Hg, respectively, compared to baseline.
During the next 3 weeks of administration both systolic and diastolic BP decreased gradually. After 4 weeks of repeated daily administration of C12 Peptide, systolic and diastolic BP had reduced significantly by 10.7 ± 1.6 and 6.9 ± 1.2 mm Hg, respectively, compared to baseline. Moreover, C12 Peptide reduced diastolic BP of prehypertensive subjects to a normal BP.
The placebo group showed a gradual decrease in both systolic and diastolic BP. After 4 weeks of repeated administration of placebo tablets, systolic and diastolic BP had reduced by 3.6 ± 2.4 mm Hg (P = not significant [NS]) and 2.7 ± 1.6 mm Hg (P = NS), respectively, compared to baseline.
Two-way analysis of variance with treatment and time as the grouping variables revealed for both systolic and diastolic BP, a treatment effect (P < .01), but there was no evidence of interaction between treatment and time.
The 4-week intervention period was followed by a 2-week off-treatment period. During this time, both systolic and diastolic BP returned gradually to baseline value (Figs. 1 and 2).
Angiotensin II and Aldosterone
To support C12 Peptide's mechanism of action in humans, angiotensin I, angiotensin II, and aldosterone levels were measured in blood plasma. In contrast to the placebo group, the angiotensin I level decreased in the C12 Peptide group (P = NS). Fig. 3 shows that after 4 weeks of daily administration of 3.8 g of C12 Peptide both plasma angiotensin II and aldosterone levels were reduced significantly compared to baseline and placebo. This is in contrast to the placebo group, which showed no significant reduction (Fig. 3).
Effect of 4 weeks of repeated daily administration of placebo tablets (solid column) or C12 Peptide tablets (shaded column) on angiotensin II level (A) and aldosterone level (B) in blood plasma of prehypertensive subjects. Values are mean ± SEM. *P < .05 compared to baseline and compared to placebo group.
Safety
Body weight, BMI, and blood chemistry/lipids parameters were not affected by 4 weeks of repeated daily administration of placebo or C12 Peptide tablets. Evaluation of the urine parameters showed no values outside of the normal range.
During the 4-week intervention period, adverse events such as dry cough, skin and gastrointestinal symptoms were not induced by either placebo or C12 Peptide tablets.
Discussion
The present study evaluated the effect of 4 weeks of repeated daily administration of C12 Peptide (3.8 g/d) tablets on systolic and diastolic BP of prehypertensive volunteers with no restrictions in dietary and physical activity. We found that already within 1 week, C12 Peptide reduced both systolic and diastolic BP significantly compared to baseline and was maintained for the next 3 weeks (Figs. 1 and 2). After 4 weeks of administration, systolic and diastolic BP were reduced significantly by 10.7 ± 1.6 and 6.9 ± 1.2 mm Hg, respectively, compared to baseline. This pattern of reduction in BP, within 2 weeks and maintained afterward, was also observed in the Dietary Approach to Stop Hypertension trial.23 In that trial, an 8-week intake of a diet rich in fruits, vegetables, and low-fat dairy foods reduced systolic and diastolic BP by 5.5 and 3.0 mm Hg more than the control diet in prehypertensive subjects. Similar reductions were observed in our study after 4 weeks of repeated administration of C12 Peptide compared to the placebo group. This shows that C12 Peptide offers an additional nutritional approach to preventing and treating hypertension, in addition to life style modifications like conscientious dietary choices with reduced salt intake and increased potassium intake, weight loss, exercise, and stress reduction.23,,,–27
The off-treatment period showed that in 2 weeks the BP rebounded to its baseline value (Figs. 1 and 2). Townsend et al19 showed that, after a single daily dose of C12 Peptide, BP returned to its initial value within 24 h. This indicates that repeated daily administration of C12 Peptide is necessary to obtain a sustained efficacy, which is consistent with (food derived) ACE inhibitors. Furthermore, this study shows that the time frame of the rebound effect, that is, the time to come back to baseline, becomes longer after repeated administration (2 weeks) and shows a gradual increase.
In the renin-angiotensin system, ACE catalyzes the synthesis of angiotensin II, which stimulates the synthesis and secretion of aldosterone. By using an ACE inhibitor, the synthesis of angiotensin II is reduced, which is associated with less aldosterone production. This study shows that after a 4-week intervention period, both plasma angiotensin II and aldosterone levels were reduced significantly in the C12 Peptide group compared to the placebo group (Fig. 3). These results do support the working mechanism of C12 Peptide containing peptides such as C6, C7, and C12 with ACE inhibiting properties.
To assess the safety of 4 weeks of repeated daily administration of 3.8 g of C12 Peptide, several blood and urine parameters were evaluated. In addition to these parameters, no significant effect was observed with respect to weight and BMI. This shows that the BP-lowering effect is induced by C12 Peptide and not, for example, by weight loss.27,28 Moreover, C12 Peptide did not induce adverse effects such as dry cough, skin and gastrointestinal symptoms. These results show that C12 Peptide is both a safe and effective ingredient for lowering BP in humans.
References
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
Author notes
This study was supported by a grant from Uni-President Enterprises Corp, Taiwan.