-
PDF
- Split View
-
Views
-
Cite
Cite
Dimitrios P. Kontoyiannis, Russell E. Lewis, Oliver Lotholary, Brad Spellberg, Georgios Petrikkos, Emmanuel Roillides, Ashraf Ibrahim, Thomas J. Walsh, Future Directions in Mucormycosis Research, Clinical Infectious Diseases, Volume 54, Issue suppl_1, February 2012, Pages S79–S85, https://doi.org/10.1093/cid/cir886
Close - Share Icon Share
Abstract
Mucormycosis has emerged as an important opportunistic infection, especially in severely immunosuppressed hosts. The evolving epidemiology, immunopathogenesis, molecular virulence studies, early diagnosis, and pitfalls in designing clinical studies of mucormycosis are discussed in this article.
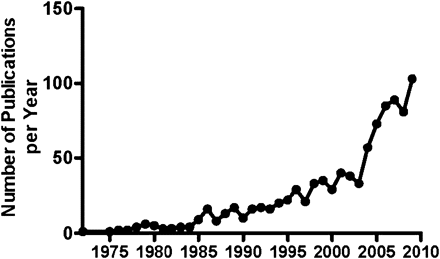
Mucormycosis (zygomycosis) has emerged as a formidable infection in an increasing population of patients with various forms of chronic and/or severe immunosuppression [1, 2]. An explosion in the number of published studies on this devastating opportunistic fungal infection over the past decade (Figure 1) reflects the ever-increasing awareness by the infectious disease community. In this article we examine the clinical and research issues regarding this rapidly changing field (Table 1).
Clinical and Research Issues Relating to Mucormycosis Research
| Epidemiology |
| Prospective international registries to determine the burden of mucormycosis and impact of antifungal use in diagnosis and identify host groups at risk |
| Development of molecular fingerprinting methods to better define nosocomial versus community-acquired mucormycosis |
| Pathogenesis |
| Comparative studies of the immunopathogenesis of mucormycosis versus aspergillosis |
| Ability to identify innate immune defects in the host that increase risk |
| Innovative animal models in phylogenetically disparate hosts |
| Molecular tool sets for studying the molecular Koch’s postulates regarding Zygomycetes virulence |
| Diagnosis |
| DNA- or antigen-based detection for early diagnosis (eg, before the first radiologic appearance of lesions) |
| Host and radiologic predictors for diagnosis of mucormycosis versus other mold infections |
| Clinical research |
| Development of guidelines to decrease practice divergence |
| Innovative study design |
| Definition of the comparators in a trial with monotherapy arms or monotherapy versus combination therapy based on risk stratification |
| Epidemiology |
| Prospective international registries to determine the burden of mucormycosis and impact of antifungal use in diagnosis and identify host groups at risk |
| Development of molecular fingerprinting methods to better define nosocomial versus community-acquired mucormycosis |
| Pathogenesis |
| Comparative studies of the immunopathogenesis of mucormycosis versus aspergillosis |
| Ability to identify innate immune defects in the host that increase risk |
| Innovative animal models in phylogenetically disparate hosts |
| Molecular tool sets for studying the molecular Koch’s postulates regarding Zygomycetes virulence |
| Diagnosis |
| DNA- or antigen-based detection for early diagnosis (eg, before the first radiologic appearance of lesions) |
| Host and radiologic predictors for diagnosis of mucormycosis versus other mold infections |
| Clinical research |
| Development of guidelines to decrease practice divergence |
| Innovative study design |
| Definition of the comparators in a trial with monotherapy arms or monotherapy versus combination therapy based on risk stratification |
Clinical and Research Issues Relating to Mucormycosis Research
| Epidemiology |
| Prospective international registries to determine the burden of mucormycosis and impact of antifungal use in diagnosis and identify host groups at risk |
| Development of molecular fingerprinting methods to better define nosocomial versus community-acquired mucormycosis |
| Pathogenesis |
| Comparative studies of the immunopathogenesis of mucormycosis versus aspergillosis |
| Ability to identify innate immune defects in the host that increase risk |
| Innovative animal models in phylogenetically disparate hosts |
| Molecular tool sets for studying the molecular Koch’s postulates regarding Zygomycetes virulence |
| Diagnosis |
| DNA- or antigen-based detection for early diagnosis (eg, before the first radiologic appearance of lesions) |
| Host and radiologic predictors for diagnosis of mucormycosis versus other mold infections |
| Clinical research |
| Development of guidelines to decrease practice divergence |
| Innovative study design |
| Definition of the comparators in a trial with monotherapy arms or monotherapy versus combination therapy based on risk stratification |
| Epidemiology |
| Prospective international registries to determine the burden of mucormycosis and impact of antifungal use in diagnosis and identify host groups at risk |
| Development of molecular fingerprinting methods to better define nosocomial versus community-acquired mucormycosis |
| Pathogenesis |
| Comparative studies of the immunopathogenesis of mucormycosis versus aspergillosis |
| Ability to identify innate immune defects in the host that increase risk |
| Innovative animal models in phylogenetically disparate hosts |
| Molecular tool sets for studying the molecular Koch’s postulates regarding Zygomycetes virulence |
| Diagnosis |
| DNA- or antigen-based detection for early diagnosis (eg, before the first radiologic appearance of lesions) |
| Host and radiologic predictors for diagnosis of mucormycosis versus other mold infections |
| Clinical research |
| Development of guidelines to decrease practice divergence |
| Innovative study design |
| Definition of the comparators in a trial with monotherapy arms or monotherapy versus combination therapy based on risk stratification |
Number of articles published annually on mucormycosis since 1975 (source: SCOPUS, accessed 16 April 2010).
EPIDEMIOLOGY
The epidemiology of mucormycosis over the past 2 decades has been complex and, at times, controversial [3]. The exact prevalence and incidence of this infection are unclear because mucormycosis is not reportable. Also, difficulties in diagnosis of mucormycosis in the current era of very low autopsy rates [4] and relative inaccuracy of hospital discharge codes [5] make assessment of the true burden of this infection challenging.
The results of a recent population-based study of mucormycosis in France that relied heavily on hospital discharge codes [6] echoed those in several recent reports from the United States. These studies noted an increasing incidence of mucormycosis, especially in hematology and hematopoietic stem cell transplant (HSCT) units [1, 2, 7]. Although this increased incidence coincided with the introduction of Aspergillus-active agents such as the echinocandins and voriconazole [2], attributing this trend solely to use of these antifungals may be overly simplistic [8]. Changes in patient profiles and qualitative elements of their immunosuppression may also play equally important roles [8]. In fact, the incidence of this infection was already increasing in the mid-1990s [7, 9]. In addition, the population-based study in France also evidenced an increased incidence in patients with diabetes who were not typically exposed to either voriconazole or the echinocandins [6]. Furthermore, it remains to be seen whether widespread use of posaconazole in prophylaxis (as adopted in several guidelines [10]) decreases the incidence of documented mucormycosis and whether the incidence of posaconazole-resistant mucormycosis would increase. The latter may be a reasonable concern in view of the reported frequent underexposure of patients to adequate levels of posaconazole, which is absorbed poorly [11–13], and because only 1 point mutation on the cytochrome P450 14α-demethylase can cause resistance of Aspergillus species to posaconazole [14]. In fact, researchers have already reported cases of breakthrough mucormycosis to posaconazole [15].
Other changes in the management of mucormycosis in patients with hematologic cancers at high risk for this infection may increase or decrease the incidence of mucormycosis in this population. Some examples of these changes are worth mentioning. For example, researchers recently found that preexisting iron overload is a predictor of poor outcome and increased adverse effects, such as bacterial and fungal infections (including mucormycosis) and organ toxicity in patients with hematologic malignancies or recipients of HSCTs [16]. Therefore, in view of the availability of the relatively nontoxic oral iron chelator deferasirox [17], hematologists may be quick to administer this agent early in the natural history of a hematologic cancer [18]. Because deferasirox has activity against Zygomycetes [19, 20], its use to treat iron overload in patients with hematologic malignancies could theoretically decrease the occurrence of mucormycosis. Others have formulated hypotheses about the indirect consequences of using nonantifungal drugs on the epidemiology of mucormycosis. Specifically, despite the explosion of the diabetes epidemic over the last 2 decades, the incidence of mucormycosis has been shown to be in constant decline in the diabetic population in some [7], but not all [6], studies during this time. Also, because statins are routinely prescribed for diabetic patients receiving treatment of hyperlipidemia and with anti-Zygomycetes effects [21, 22], the association may be more than a coincidence and may illustrate the complexity and “fluidity” of the epidemiology of mucormycosis.
We have only begun to scratch the surface regarding the complicated observation of mucormycosis cases in some but not all tertiary cancer centers [6] (B. Parks and D. P. Kontoyiannis, unpublished data). Could this inconsistency be related to the intensity of diagnostic workup for mucormycosis? The recently published TRANSNET registry showed that this indeed may be the case for the more common mold infection invasive aspergillosis (IA) (ie, centers that performed high numbers of HSCTs had increased incidences of IA) [23]. Could this be related to unique geographical exposure to Zygomycetes in the environment or due to weather patterns? A recent study showed that the latter partially explains differences in the incidence of IA between the transplant center at Fred Hutchinson Cancer Research Center in Seattle, Washington, and the HSCT service at The University of Texas MD Anderson Cancer Center in Houston [24]. Only carefully obtained prospective surveillance data from North America, Europe, and other continents could capture the true burden of mucormycosis and the dynamics of the evolution of its epidemiology; researchers have begun to report on such data [25–28]. These studies are particularly important in evaluating the feasibility of and target populations for future clinical trials of mucormycosis treatment. Finally, molecular tools with improved definition of the clonality of mucormycosis cases and its hospital acquisition would help tremendously in improving description of the epidemiology of mucormycosis [2]. This area of study is in its infancy, so more work is needed.
PATHOGENESIS
Investigators have recently made tremendous strides in deciphering the pathogenesis of mucormycosis [3, 29, 30]. They built these efforts on the results of classic studies from the 1980s documenting the important, unique role of iron acquisition in this pathogenesis [31–34]. Findings point to iron chelation as an important therapeutic strategy deserving of further exploration [19]. The recent sequencing of the Rhizopus genome [35] and development of molecular tool sets for studying gene function [36] are expected to rapidly increase our understanding of the unique virulence properties of the fungus. Evidence of widespread genome duplication in Rhizopus [35] has shed light on the known attributes of Zygomycetes virulence, specifically, versatility, robust growth, and multidrug resistance [3, 29, 37]. Table 2 lists some specific steps to take when studying the molecular pathogenesis of mucormycosis in the postgenomic era. We believe that comparative study of the immunopathogenesis of IA and mucormycosis in relevant experimental systems and under different immunosuppression scenarios (eg, corticosteroid use versus neutropenia) is equally important.
Identification of Molecular Mechanisms of Rhizopus Species Pathogenesis and Immunopathogenesis in the Postgenomic Era
| Define the complete transcriptome for early colonization and adhesion to epithelia |
| Identify patterns of gene expression during hyphal growth, tissue invasion |
| Identify the blood factors (eg, neutrophils, complement factor) that are able to control Rhizopus species growth and prevent its dissemination |
| Identify signaling pathways involved in escape of Zygomycetes from the blood compartments across the endothelial lining to grow in deep-seated organs (eg, iron acquisition–regulating genes, hyphally associated genes, thermal stress genes, starvation-responsive pathways) |
| Define the complete transcriptome for early colonization and adhesion to epithelia |
| Identify patterns of gene expression during hyphal growth, tissue invasion |
| Identify the blood factors (eg, neutrophils, complement factor) that are able to control Rhizopus species growth and prevent its dissemination |
| Identify signaling pathways involved in escape of Zygomycetes from the blood compartments across the endothelial lining to grow in deep-seated organs (eg, iron acquisition–regulating genes, hyphally associated genes, thermal stress genes, starvation-responsive pathways) |
Identification of Molecular Mechanisms of Rhizopus Species Pathogenesis and Immunopathogenesis in the Postgenomic Era
| Define the complete transcriptome for early colonization and adhesion to epithelia |
| Identify patterns of gene expression during hyphal growth, tissue invasion |
| Identify the blood factors (eg, neutrophils, complement factor) that are able to control Rhizopus species growth and prevent its dissemination |
| Identify signaling pathways involved in escape of Zygomycetes from the blood compartments across the endothelial lining to grow in deep-seated organs (eg, iron acquisition–regulating genes, hyphally associated genes, thermal stress genes, starvation-responsive pathways) |
| Define the complete transcriptome for early colonization and adhesion to epithelia |
| Identify patterns of gene expression during hyphal growth, tissue invasion |
| Identify the blood factors (eg, neutrophils, complement factor) that are able to control Rhizopus species growth and prevent its dissemination |
| Identify signaling pathways involved in escape of Zygomycetes from the blood compartments across the endothelial lining to grow in deep-seated organs (eg, iron acquisition–regulating genes, hyphally associated genes, thermal stress genes, starvation-responsive pathways) |
Zygomycetes species have great affinity for blood vessels, invade rapidly, and disseminate widely [3]. Furthermore, hemorrhagic necrosis is the hallmark of mucormycotic tissue lesions [38]. However, important questions remain. For example, which endothelial receptors are utilized by Zygomycetes for invasion [39, 40]? Previous data demonstrated the ability of Mucorales to bind, invade, and damage human umbilical vein endothelial cells in vitro [39]. A recent study demonstrated that glucose regulated protein 78 (GRP78) serves as a receptor that promotes the ability of Mucorales to invade endothelial cells lining blood vessels [40]. Elevated concentrations of glucose and iron, consistent with those seen during diabetic ketoacidosis, enhance GRP78 expression and resulting invasion and damage of endothelial cells in a receptor-dependent manner [40]. These findings likely explain the unique susceptibility of diabetic ketoacidosis to mucormycosis.
Also, what is the exact mechanism of immune evasion by Zygomycetes? And, finally, can Zygomycetes disrupt endothelial integrity via mycotoxin production as shown recently in cases of experimental IA [41]? Development of novel models of this infection would be welcomed because researchers have relied heavily on murine models of hyperacute disseminated (via intravenous introduction) and pulmonary (via inhalation of high fungal loads) mucormycosis [42]. Mini–host models of mucormycosis (Drosophila) have shed light on the Toll-independent mechanisms of evasion of Zygomycetes species in contrast with those seen for IA [43]. Also, the recent development of subacute, subcutaneous murine models of mucormycosis (D. P. Kontoyiannis, unpublished data) may help in the study of tissue- and inoculum-specific differences in the pathogenesis of mucormycosis. Finally, preclinical studies of the immunomodulatory effects and comparative pharmacokinetics of antifungals are needed [44, 45]. The upcoming decade is expected to be an era of intense activity in studying mucormycosis that hopefully will lead to new avenues for diagnosis, immune monitoring, and antifungal drug discovery.
DIAGNOSIS
Early diagnosis of mucormycosis remains challenging and is a major unmet need, causing a bottleneck in devising innovative, effective clinical trials. This is important because delayed treatment clearly impacts the outcome of mucormycosis [46]. Major problems in the diagnosis of mucormycosis include its elusive clinical presentation and frequent occult dissemination [9], a lack of sensitive nonculture-based diagnostic methods (eg, antigen and molecular detection platforms), and the fact that culture of samples obtained from nonsterile sites (eg, sputum) is neither sensitive [9, 47] nor specific [9, 47, 48]. Tissue-based diagnosis remains the gold standard, although patients frequently are not candidates for biopsy analysis because of thrombocytopenia or hemodynamic instability. Even with tissue-based diagnostic methods, sampling errors and occasional difficulties in differentiating Zygomycetes from distorted hyalohyphomycetes in tissue samples result in false-negative or false-positive results [49]. Furthermore, paradoxically, two-thirds of histopathologically proven mucormycosis cases are culture negative [47]. It has not yet been determined whether optimal processing of infected tissue samples and adaptation of culture conditions to simulate semi-anaerobic growth [50] require further validation.
Morphologic identification of Zygomycetes at the species level is reasonably accurate, especially when performed in high-volume [2] or reference clinical microbiology [51] laboratories. Of note, carbon-assimilation profiles have been reported to be a promising tool for the precise identification of Zygomycetes [52]. Polymerase chain reaction–based detection of Zygomycetes fungi remains investigational [53], although recent data reveal that ITS sequencing was promising for identification of Zygomycetes genus/species from culture, frozen sections, or paraffin-embedded sections of infected tissues [54, 55]. Whether detection of volatile compounds produced by Zygomycetes fermentation in patients with mucormycosis is of help in diagnosis is an unexplored area. An equally important unexplored area is identification of diagnostic factors that favor mucormycosis over the more common IA on clinical grounds. Retrospective single-institution studies focusing on patients with leukemia and/or recipients of HSCTs identified prior voriconazole exposure, community-acquired pansinusitis, hyperglycemia (eg, steroid-induced), multiple (>10) nodules detected on chest computed tomography scans, pleural effusion, and the reverse halo sign as markers potentially favoring a diagnosis of mucormycosis over a diagnosis of aspergillosis [56, 57]. Prospective validation and similar studies of other populations at risk for mucormycosis are needed. Development of a “scorecard” for diagnosis of mucormycosis would be ideal.
TREATMENT
The literature on mucormycosis is modestly helpful to clinicians managing this infection. However, “primary” prospective mucormycosis treatment data have yet to be reported [3]. Both salvage and single-institution retrospective studies should be viewed with caution in light of the multiple inherent biases [58]. Multiple controversies remain. For example, host heterogeneity, pleiotropic clinical presentation (at least 6 clinical syndromes), the multitude of Zygomycetes species, factors related to surgery (eg, timing, radicalness), and the influence or correction of underlying metabolic (eg, ketoacidosis) and immunosuppressive (eg, neutrophil recovery in neutropenic patients, steroid tapering in patients receiving corticosteroids) conditions lead to complex and at times highly individualized scenarios for management of mucormycosis [3]. For example, recent studies at The University of Texas MD Anderson Cancer Center showed that a lack of corticosteroid tapering, monocytopenia and persistent neutropenia, and a lack of early treatment of mucormycosis were powerful independent predictors of poor outcome of this infection [56].
In our view, a successful clinical trial of treatment of mucormycosis hinges on the following factors:
Enthusiastic participation of all investigators so that as many patients as possible are enrolled and protocol violations are kept to a minimum.
Risk stratification using a scoring system. Table 3 depicts a possible scoring system that is based on preenrollment patient characteristics. Unfortunately, such scoring systems have yet to be developed and validated not only for mucormycosis but also for other clinically important fungal infections.
Early sensitive diagnosis and staging of mucormycosis at baseline (eg, using computed tomography) to intervene early in the natural history of the infection and assist in risk stratification, respectively.
Use of liberal inclusion criteria to allow a degree of preexisting renal or hepatic dysfunction.
In view of the rarity of mucormycosis, use of an innovative statistical design for power calculation like that used for rare, complex oncologic entities (eg, adaptive randomization) [59].
Preenrollment of possible cases (conditional enrollment) and obtainment of definitive diagnoses within 7–10 days after enrollment, perhaps with analysis of possible mucormycosis cases as supportive evidence.
Superiority trial with a margin of 15%. For example, the pivotal trial of voriconazole versus amphotericin B deoxycholate in treatment of IA had a noninferiority margin of 20% [60].
The timing for assessment of response of mucormycosis to treatment should reflect the natural history of the infection and the treatment effect (not too early or too late). Therefore, different endpoints and scenarios for analysis as a function of time after enrollment (eg, progression-free response rate at week 4, global response rate at week 8, overall mortality rate at week 12) may be used. The lack of a validated outcome measure or surrogate endpoint that is reproducible, free of bias, and correlates with treatment effect rather than underlying disease effect remains a major issue.
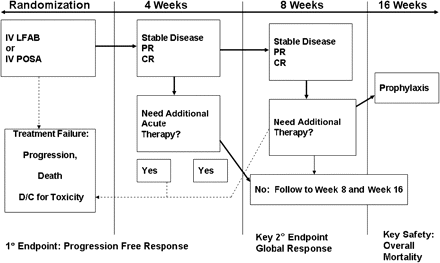
In terms of treatment comparators, different options are available depending in part on the feasibility of industry support for such a complex and relatively infrequent infection and small market. Potential combination strategies have been the subject of a previous article in this supplement. The expected introduction of intravenous and oral posaconazole formulations with reliable pharmacokinetics opens the possibility of performing a randomized study of monotherapy with a lipid formulation of amphotericin B versus that with intravenous posaconazole for severe mucormycosis. Figure 2 shows a design for such a study.
Finally, the inability to control for variability of practice (diagnosis and/or management) across a large number of centers and heterogeneous hosts is perhaps the most important threat to a successful trial, as shown in the recently published Deferasirox-AmBisome Therapy for Mucormycosis study [61]. Perhaps creating regional “centers of excellence” for managing this infection both medically and surgically would result in less variability.
Potential Components of Mucormycosis Risk Stratification at Baseline
| Preenrollment characteristic |
| Age (>40 vs <40 years) |
| Type of hematopoietic stem cell transplant |
| Allogeneic |
| Autologous |
| Relapsed leukemia |
| Interval from diagnosis to surgery |
| Early (1–7 days) |
| Intermediate (8–14 days) |
| Late (>14 days) |
| Diabetes control |
| Good |
| Poor |
| Staging of mucormycosis |
| Disseminated |
| Rhinocerebral |
| Multifocal pulmonary |
| Preenrollment characteristic |
| Age (>40 vs <40 years) |
| Type of hematopoietic stem cell transplant |
| Allogeneic |
| Autologous |
| Relapsed leukemia |
| Interval from diagnosis to surgery |
| Early (1–7 days) |
| Intermediate (8–14 days) |
| Late (>14 days) |
| Diabetes control |
| Good |
| Poor |
| Staging of mucormycosis |
| Disseminated |
| Rhinocerebral |
| Multifocal pulmonary |
Potential Components of Mucormycosis Risk Stratification at Baseline
| Preenrollment characteristic |
| Age (>40 vs <40 years) |
| Type of hematopoietic stem cell transplant |
| Allogeneic |
| Autologous |
| Relapsed leukemia |
| Interval from diagnosis to surgery |
| Early (1–7 days) |
| Intermediate (8–14 days) |
| Late (>14 days) |
| Diabetes control |
| Good |
| Poor |
| Staging of mucormycosis |
| Disseminated |
| Rhinocerebral |
| Multifocal pulmonary |
| Preenrollment characteristic |
| Age (>40 vs <40 years) |
| Type of hematopoietic stem cell transplant |
| Allogeneic |
| Autologous |
| Relapsed leukemia |
| Interval from diagnosis to surgery |
| Early (1–7 days) |
| Intermediate (8–14 days) |
| Late (>14 days) |
| Diabetes control |
| Good |
| Poor |
| Staging of mucormycosis |
| Disseminated |
| Rhinocerebral |
| Multifocal pulmonary |
Potential study design for examining primary monotherapy for mucormycosis. Abbreviations: CR, complete response; D/C, discontinuation; IV, intravenous; LFAB, lipid formulation of amphotericin B; POSA, posaconazole; PR, partial response.
CONCLUSIONS
In the symposium from which this supplement was derived, all of the scientists, physician-investigators, laypersons, Henry Schueler, and relatives and friends of Henry Schueler, the heroic child who died of this devastating infection, had a palpable sense of working in a frontier of clinical mycology. Much work remains ahead of us. By mobilizing resources, organizing, expanding intellectual energy, and thinking innovatively, we can make strides in improving outcomes of mucormycosis.
Notes
Acknowledgments.
We thank our patients for providing the inspiration for doing research related to this disease.
Financial support.
This research is supported in part by the National Institutes of Health through MD Anderson’s Cancer Center Support Grant CA016672.
Supplement sponsorship.
This article was published as part of a supplement entitled “Advances Against Mucormycosis: A Tribute to the Memory and Courage of Hank Schueler,” sponsored by the Henry Schueler 41&9 Foundation.
Potential conflicts of interest.
R. L. has received grant support from Merck & Co, Gilead, Astellas, and Enzon. O. L. has received grant support from Gilead; has received grant support from and has been a consultant for Astellas; and has served on speakers’ bureaus for Astellas, Gilead, Pfizer, Schering-Plough, and Merck. B. S. has received grant support from Gilead, Astellas, and Novartis; has been a consultant for Merck, Pfizer, Arpida, Theravance, Advanced Life Sciences, Basilea, The Medicines Company, Novo Nordisk, Novartis, and Cerexa; and is a shareholder of NovaDigm Therapeutics and Neutropenia Immunotherapy Solutions. T. J. W. has received grant support from Novartis and Astellas, and has been a consultant for Trius, iCo, Sigma Tau, Draius, and Novartis. D. K. has served on the board and received consultancy support from Schering-Plough and Merck. E. R. has been a consultant for Schering-Plough, Gilead, Astellas, Cephalon, Pfizer, Wyeth, Merck, and Aventis, and has lectured at or served on speakers’ bureaus for Pfizer, Gilead, Enzon, Schering-Plough, and Wyeth. A. I. has received grant support from Astellas, Enzon, Gilead, Merck, Pfizer, NovaDigm Therapeutics, and Novartis, and is a shareholder of NovaDigm Therapeutics and Spectral Platforms. G. P. has received research grants from Gilead, Pfizer, Schering-Plough, Aventis, and MSD; has acted as paid consultant to Janssen-Cilag, Gilead, Astellas, and Schering-Plough; and is a member of the speakers’ bureaus for Gilead, Schering-Plough, and MSD.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.





Comments