-
PDF
- Split View
-
Views
-
Cite
Cite
Juhani Knuuti, William Wijns, Antti Saraste, Davide Capodanno, Emanuele Barbato, Christian Funck-Brentano, Eva Prescott, Robert F Storey, Christi Deaton, Thomas Cuisset, Stefan Agewall, Kenneth Dickstein, Thor Edvardsen, Javier Escaned, Bernard J Gersh, Pavel Svitil, Martine Gilard, David Hasdai, Robert Hatala, Felix Mahfoud, Josep Masip, Claudio Muneretto, Marco Valgimigli, Stephan Achenbach, Jeroen J Bax, ESC Scientific Document Group , 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC), European Heart Journal, Volume 41, Issue 3, 14 January 2020, Pages 407–477, https://doi.org/10.1093/eurheartj/ehz425
Close - Share Icon Share
 For the Supplementary Data which include background information and detailed discussion of the data that have provided the basis for the Guidelines see https://academic.oup.com/eurheartj/article-lookup/doi/10.1093/eurheartj/ehz425#supplementary-data
For the Supplementary Data which include background information and detailed discussion of the data that have provided the basis for the Guidelines see https://academic.oup.com/eurheartj/article-lookup/doi/10.1093/eurheartj/ehz425#supplementary-data
Table of contents
1. Preamble 411
2. Introduction 413
2.1 What is new in the 2019 Guidelines? 414
3. Patients with angina and/or dyspnoea, and suspected coronary artery disease 416
3.1 Basic assessment, diagnosis, and risk assessment 416
3.1.1 Step 1: symptoms and signs 417
3.1.1.1 Stable vs. unstable angina 418
3.1.1.2 Distinction between symptoms caused by epicardial vs. microvascular/vasospastic disease 419
3.1.2 Step 2: comorbidities and other causes of symptoms 419
3.1.3 Step 3: basic testing 419
3.1.3.1 Biochemical tests 419
3.1.3.2 Resting electrocardiogram and ambulatory monitoring 420
3.1.3.3 Echocardiography and magnetic resonance imaging at rest 420
3.1.3.4 Chest X-ray 421
3.1.4 Step 4: assess pre-test probability and clinical likelihood of coronary artery disease 421
3.1.5 Step 5: select appropriate testing 422
3.1.5.1 Functional non-invasive tests 422
3.1.5.2 Anatomical non-invasive evaluation 423
3.1.5.3 Role of the exercise electrocardiogram 423
3.1.5.4 Selection of diagnostic tests 424
3.1.5.5 The impact of clinical likelihood on the selection of a diagnostic test 424
3.1.5.6 Invasive testing 425
3.1.6 Step 6: assess event risk 427
3.1.6.1 Definition of levels of risk 428
3.2 Lifestyle management 429
3.2.1 General management of patients with coronary artery disease 429
3.2.2 Lifestyle modification and control of risk factors 429
3.2.2.1 Smoking 429
3.2.2.2 Diet and alcohol 430
3.2.2.3 Weight management 430
3.2.2.4 Physical activity 430
3.2.2.5 Cardiac rehabilitation 430
3.2.2.6 Psychosocial factors 430
3.2.2.7 Environmental factors 431
3.2.2.8 Sexual activity 431
3.2.2.9 Adherence and sustainability 431
3.2.2.10 Influenza vaccination 431
3.3 Pharmacological management 432
3.3.1 Anti-ischaemic drugs 432
3.3.1.1 General strategy 432
3.3.1.2 Available drugs 432
3.3.1.3 Patients with low blood pressure 435
3.3.1.4 Patients with low heart rate 435
3.3.2 Event prevention 436
3.3.2.1 Antiplatelet drugs 436
3.3.2.2 Anticoagulant drugs in sinus rhythm 436
3.3.2.3 Anticoagulant drugs in atrial fibrillation 437
3.3.2.4 Proton pump inhibitors 437
3.3.2.5 Cardiac surgery and antithrombotic therapy 437
3.3.2.6 Non-cardiac surgery and antithrombotic therapy 438
3.3.3 Statins and other lipid-lowering drugs 440
3.3.4 Renin−angiotensin−aldosterone system blockers 440
3.3.5 Hormone replacement therapy 441
3.4 Revascularization 441
4. Patients with new onset of heart failure or reduced left ventricular function 442
5. Patients with a long-standing diagnosis of chronic coronary syndromes 444
5.1 Patients with stabilized symptoms <1 year after an acute coronary syndrome or patients with recent revascularization 444
5.2 Patients >1 year after initial diagnosis or revascularization 444
6. Angina without obstructive disease in the epicardial coronary arteries 446
6.1 Microvascular angina 447
6.1.1 Risk stratification 447
6.1.2 Diagnosis 447
6.1.3 Treatment 447
6.2 Vasospastic angina 448
6.2.1 Diagnosis 448
6.2.2 Treatment 448
7. Screening for coronary artery disease in asymptomatic subjects 449
8. Chronic coronary syndromes in specific circumstances 450
8.1 Cardiovascular comorbidities 450
8.1.1 Hypertension 450
8.1.2 Valvular heart disease (including planned transcatheter aortic valve implantation) 450
8.1.3 After heart transplantation 450
8.2 Non-cardiovascular comorbidities 451
8.2.1 Cancer 451
8.2.2 Diabetes mellitus 451
8.2.3 Chronic kidney disease 452
8.2.4 Elderly 452
8.3 Sex 452
8.4 Patients with refractory angina 453
9. Key messages 454
10. Gaps in the evidence 455
10.1 Diagnosis and assessment 455
10.2 Assessment of risk 455
10.3 Lifestyle management 455
10.4 Pharmacological management 455
10.5 Revascularization 455
10.6 Heart failure and left ventricular dysfunction 455
10.7 Patients with long-standing diagnosis of chronic coronary syndromes 455
10.8 Angina without obstructive coronary artery disease 455
10.9 Screening in asymptomatic subjects 455
10.10 Comorbidities 456
10.11 Patients with refractory angina 456
11. ’What to do’ and ’what not to do’ messages from the Guidelines 456
12. Supplementary data 460
13. Appendix 460
14. References 461
Recommendations
2019 New major recommendations 414
Changes in major recommendations 416
Basic biochemistry testing in the initial diagnostic management of patients with suspected coronary artery disease 419
Resting electrocardiogram in the initial diagnostic management of patients with suspected coronary artery disease 420
Ambulatory electrocardiogram monitoring in the initial diagnostic management of patients with suspected coronary artery disease 420
Resting echocardiography and cardiac magnetic resonance in the initial diagnostic management of patients with suspected coronary artery disease 421
Chest X-ray in the initial diagnostic management of patients with suspected coronary artery disease 421
Use of diagnostic imaging tests in the initial diagnostic management of symptomatic patients with suspected coronary artery disease 426
Performing exercise electrocardiogram in the initial diagnostic management of patients with suspected coronary artery disease 426
Recommendations for risk assessment 428
Recommendations on lifestyle management 431
Recommendations on anti-ischaemic drugs in patients with chronic coronary syndromes 435
Recommendations for event prevention I 438
Recommendations for event prevention II 441
General recommendations for the management of patients with cnronic coronary syndromes and symptomatic heart failure due to ischaemic cardiomyopathy and left ventricular systolic dysfunction 443
Recommendations for patients with a long-standing diagnosis of chronic coronary syndromes 446
Investigations in patients with suspected coronary microvascular angina 448
Recommendations for investigations in patients with suspected vasospastic angina 448
Recommendations for screening for coronary artery disease in asymptomatic subjects 449
Recommendations for hypertension treatment in chronic coronary syndromes 450
Recommendations for valvular disease in chronic coronary syndromes 450
Recommendations for active cancer in chronic coronary syndromes 451
Recommendations for diabetes mellitus in chronic coronary syndromes 451
Recommendations for chronic kidney disease in chronic coronary syndromes 452
Recommendations for elderly patients with chronic coronary syndromes 452
Recommendation for sex issues and chronic coronary syndromes 453
Recommendations for treatment options for refractory angina 454
Recommendations: ’what to do’ and ’what not to do’ 456
List of tables
Table 1 Classes of recommendations 412
Table 2 Levels of evidence 412
Table 3 Traditional clinical classification of suspected anginal symptoms 418
Table 4 Grading of effort angina severity according to the Canadian Cardiovascular Society 418
Table 5 Pre-test probabilities of obstructive coronary artery disease in 15 815 symptomatic patients according to age, sex, and the nature of symptoms in a pooled analysis of contemporary data 422
Table 6 Definitions of high event risk for different test modalities in patients with established chronic coronary syndromes 427
Table 7 Lifestyle recommendations for patients with chronic coronary syndromes. 429
Table 8 Healthy diet characteristics 430
Table 9 Treatment options for dual antithrombotic therapy in combination with aspirin 75-100 mg daily in alphabetical order in patients who have a high or moderate risk of ischaemic events, and do not have a high bleeding risk. 440
Table 10 Blood pressure thresholds for definition of hypertension with different types of blood pressure measurement 450
Table 11 Potential treatment options for refractory angina and summary of trial data 453
List of figures
Figure 1 Schematic illustration of the natural history of chronic coronary syndromes 413
Figure 2 Approach for the initial diagnostic management of patients with angina and suspected coronary artery disease 417
Figure 3 Determinants of clinical likelihood of obstructive coronary artery disease 423
Figure 4 Main diagnostic pathways in symptomatic patients with suspected obstructive coronary artery disease 424
Figure 5 Ranges of clinical likelihood of coronary artery disease in which the test can rule-in or rule-out obstructive coronary artery disease 425
Figure 6 Comparison of risk assessments in asymptomatic apparently healthy subjects (primary prevention) and patients with established chronic coronary syndromes (secondary prevention) 427
Figure 7 The five As of smoking cessation 430
Figure 8 Suggested stepwise strategy for long-term anti-ischaemic drug therapy in patients with chronic coronary syndromes and specific baseline characteristics 434
Figure 9 Decision tree for patients undergoing invasive coronary angiography 442
Figure 10 Proposed algorithm according to patient types commonly observed at chronic coronary syndrome outpatient clinics 445
Abbreviations and acronyms
- ABI
Ankle−brachial index
- ACE
Angiotensin-converting enzyme
- ACS
Acute coronary syndrome(s)
- ACTION
A Coronary disease Trial Investigating Outcome with Nifedipine gastrointestinal therapeutic system
- AF
Atrial fibrillation
- ARB
Angiotensin receptor blocker
- AUGUSTUS
An Open-label, 2 × 2 Factorial, Randomized Controlled, Clinical Trial to Evaluate the Safety of Apixaban vs. Vitamin K Antagonist and Aspirin vs. Aspirin Placebo in Patients With Atrial Fibrillation and Acute Coronary Syndrome or Percutaneous Coronary Intervention
- BARI-2D
Bypass Angioplasty Revascularization Investigation 2 Diabetes
- BEAUTIFUL
If Inhibitor Ivabradine in Patients with Coronary Artery Disease and Left Ventricular Dysfunction
- b.i.d.
Bis in die (twice a day)
- BMI
Body mass index
- BP
Blood pressure
- b.p.m.
Beats per minute
- CABG
Coronary artery bypass grafting
- CAD
Coronary artery disease
- CAPRIE
Clopidogrel vs. Aspirin in Patients at Risk of Ischaemic Events
- CASS
Coronary Artery Surgery Study
- CCB
Calcium channel blocker
- CCS
Chronic coronary syndrome(s)
- CFR
Coronary flow reserve
- CHA2DS2- VASc
Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]
- CHD
Coronary heart disease
- CI
Confidence interval
- CKD
Chronic kidney disease
- CMR
Cardiac magnetic resonance
- COMPASS
Cardiovascular Outcomes for People Using Anticoagulation Strategies
- COURAGE
Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation
- CPG
Committee for Practice Guidelines
- CRT
Cardiac resynchronization therapy
- CT
Computed tomography
- CTA
Computed tomography angiography
- CVD
Cardiovascular disease
- DAPT
Dual antiplatelet therapy
- DES
Drug-eluting stent(s)
- DHP
Dihydropyridine
- ECG
Electrocardiogram
- eGFR
Estimated glomerular filtration rate
- ESC
European Society of Cardiology
- FAME 2
Fractional Flow Reserve versus Angiography for Multivessel Evaluation 2
- FFR
Fractional flow reserve
- FFRCT
Computed tomography-based fractional flow reserve
- GEMINI- ACS
A Study to Compare the Safety of Rivaroxaban Versus Acetylsalicylic Acid in Addition to Either Clopidogrel or Ticagrelor Therapy in Participants With Acute Coronary Syndrome
- GFR
Glomerular filtration rate
- GLS
Global longitudinal strain
- GOSPEL
Global secondary prevention strategies to limit event recurrence after myocardial infarction
- HbA1c
Glycated haemoglobin
- HF
Heart failure
- ICA
Invasive coronary angiography
- IMR
Index of microcirculatory resistance
- IMT
Intima−media thickness
- IONA
Impact Of Nicorandil in Angina
- iwFR
Instantaneous wave-free ratio (instant flow reserve)
- LAD
Left anterior descending
- LBBB
Left bundle branch block
- LDL-C
Low-density lipoprotein cholesterol
- LM
Left main (coronary artery)
- LV
Left ventricular
- LVEF
Left ventricular ejection fraction
- MI
Myocardial infarction
- MRA
Mineralocorticoid receptor antagonist
- NOAC
Non-vitamin K antagonist oral anticoagulant
- NT-proBNP
N-terminal pro-B-type natriuretic peptide
- OAC
Oral anticoagulant
- o.d.
Omni die (once a day)
- ORBITA
Objective Randomised Blinded Investigation with optimal medical Therapy of Angioplasty in stable angina
- PAD
Peripheral artery disease
- PCI
Percutaneous coronary intervention
- PCSK9
Proprotein convertase subtilisin-kexin type 9
- PEGASUS- TIMI 54
Prevention of Cardiovascular Events in Patients with Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin–Thrombolysis in Myocardial Infarction 54
- PET
Positron emission tomography
- PROMISE
Prospective Multicenter Imaging Study for Evaluation of Chest Pain
- PTP
Pre-test probability
- RAS
Renin−angiotensin system
- RCT
Randomized clinical trial
- REACH
Reduction of Atherothrombosis for Continued Health
- RIVER-PCI
Ranolazine for Incomplete Vessel Revascularization Post‐Percutaneous Coronary Intervention
- SCORE
Systematic COronary Risk Evaluation
- SCOT- HEART
Scottish Computed Tomography of the HEART
- SIGNIFY
Study Assessing the Morbidity–Mortality Benefits of the If Inhibitor Ivabradine in Patients with Coronary Artery Disease
- SPECT
Single-photon emission computed tomography
- VKA
Vitamin K antagonist
1 Preamble
Guidelines summarize and evaluate available evidence with the aim of assisting health professionals in proposing the best management strategies for an individual patient with a given condition. Guidelines and their recommendations should facilitate decision making of health professionals in their daily practice. However, the final decisions concerning an individual patient must be made by the responsible health professional(s) in consultation with the patient and caregiver as appropriate.
A great number of guidelines have been issued in recent years by the European Society of Cardiology (ESC), as well as by other societies and organizations. Because of their impact on clinical practice, quality criteria for the development of guidelines have been established in order to make all decisions transparent to the user. The recommendations for formulating and issuing ESC Guidelines can be found on the ESC website (http://www.escardio.org/Guidelines-&-Education/Clinical-Practice-Guidelines/Guidelines-development/Writing-ESC-Guidelines). The ESC Guidelines represent the official position of the ESC on a given topic and are regularly updated.
The ESC carries out a number of registries which are essential to assess, diagnostic/therapeutic processes, use of resources and adherence to Guidelines. These registries aim at providing a better understanding of medical practice in Europe and around the world, based on data collected during routine clinical practice.
The guidelines are developed together with derivative educational material addressing the cultural and professional needs for cardiologists and allied professionals. Collecting high-quality observational data, at appropriate time interval following the release of ESC Guidelines, will help evaluate the level of implementation of the Guidelines, checking in priority the key end points defined with the ESC Guidelines and Education Committees and Task Force members in charge.
The Members of this Task Force were selected by the ESC, including representation from its relevant ESC sub-specialty groups, in order to represent professionals involved with the medical care of patients with this pathology. Selected experts in the field undertook a comprehensive review of the published evidence for management of a given condition according to ESC Committee for Practice Guidelines (CPG) policy. A critical evaluation of diagnostic and therapeutic procedures was performed, including assessment of the riskbenefit ratio. The level of evidence and the strength of the recommendation of particular management options were weighed and graded according to predefined scales, as outlined in Tables 1 and 2.
The experts of the writing and reviewing panels provided declaration of interest forms for all relationships that might be perceived as real or potential sources of conflicts of interest. These forms were compiled into one file and can be found on the ESC website (http://www.escardio.org/guidelines). Any changes in declarations of interest that arise during the writing period were notified to the ESC and updated. The Task Force received its entire financial support from the ESC without any involvement from the healthcare industry.
The ESC CPG supervises and coordinates the preparation of new Guidelines. The Committee is also responsible for the endorsement process of these Guidelines. The ESC Guidelines undergo extensive review by the CPG and external experts. After appropriate revisions the Guidelines are approved by all the experts involved in the Task Force. The finalized document is approved by the CPG for publication in the European Heart Journal. The Guidelines were developed after careful consideration of the scientific and medical knowledge and the evidence available at the time of their dating.
The task of developing ESC Guidelines also includes the creation of educational tools and implementation programmes for the recommendations including condensed pocket guideline versions, summary slides, booklets with essential messages, summary cards for non-specialists and an electronic version for digital applications (smartphones, etc.). These versions are abridged and thus, for more detailed information, the user should always access to the full text version of the Guidelines, which is freely available via the ESC website and hosted on the EHJ website. The National Societies of the ESC are encouraged to endorse, translate and implement all ESC Guidelines. Implementation programmes are needed because it has been shown that the outcome of disease may be favourably influenced by the thorough application of clinical recommendations.
Health professionals are encouraged to take the ESC Guidelines fully into account when exercising their clinical judgment, as well as in the determination and the implementation of preventive, diagnostic or therapeutic medical strategies. However, the ESC Guidelines do not override in any way whatsoever the individual responsibility of health professionals to make appropriate and accurate decisions in consideration of each patient's health condition and in consultation with that patient or the patient's caregiver where appropriate and/or necessary. It is also the health professional's responsibility to verify the rules and regulations applicable in each country to drugs and devices at the time of prescription.
2 Introduction
Coronary artery disease (CAD) is a pathological process characterized by atherosclerotic plaque accumulation in the epicardial arteries, whether obstructive or non-obstructive. This process can be modified by lifestyle adjustments, pharmacological therapies, and invasive interventions designed to achieve disease stabilization or regression. The disease can have long, stable periods but can also become unstable at any time, typically due to an acute atherothrombotic event caused by plaque rupture or erosion. However, the disease is chronic, most often progressive, and hence serious, even in clinically apparently silent periods. The dynamic nature of the CAD process results in various clinical presentations, which can be conveniently categorized as either acute coronary syndromes (ACS) or chronic coronary syndromes (CCS). The Guidelines presented here refer to the management of patients with CCS. The natural history of CCS is illustrated in Figure 1.
Schematic illustration of the natural history of chronic coronary syndromes. ACE = angiotensin-converting enzyme; ACS = acute coronary syndromes; CCS = chronic coronary syndromes; MI = myocardial infarction.
The most frequently encountered clinical scenarios in patients with suspected or established CCS are: (i) patients with suspected CAD and ‘stable’ anginal symptoms, and/or dyspnoea (see section 3); (ii) patients with new onset of heart failure (HF) or left ventricular (LV) dysfunction and suspected CAD (see section 4); (iii) asymptomatic and symptomatic patients with stabilized symptoms <1 year after an ACS, or patients with recent revascularization (see section 5.1); (iv) asymptomatic and symptomatic patients >1 year after initial diagnosis or revascularization (see section 5.2); (v) patients with angina and suspected vasospastic or microvascular disease (see section 6); and (vi) asymptomatic subjects in whom CAD is detected at screening (see section 7).
All of these scenarios are classified as a CCS but involve different risks for future cardiovascular events [e.g. death or myocardial infarction (MI)], and the risk may change over time. Development of an ACS may acutely destabilize each of these clinical scenarios. The risk may increase as a consequence of insufficiently controlled cardiovascular risk factors, suboptimal lifestyle modifications and/or medical therapy, or unsuccessful revascularization. Alternatively, the risk may decrease as a consequence of appropriate secondary prevention and successful revascularization. Hence, CCS are defined by the different evolutionary phases of CAD, excluding situations in which an acute coronary artery thrombosis dominates the clinical presentation (i.e. ACS).
In the present Guidelines, each section deals with the main clinical scenarios of CCS. This structure aims to simplify the use of the Guidelines in clinical practice. Additional information, tables, figures, and references are available in the Supplementary Data on the ESC website (www.escardio.org) as well as in The ESC Textbook of Cardiovascular Medicine.
2.1 What is new in the 2019 Guidelines?
| New/revised concepts in 2019 |
| The Guidelines have been revised to focus on CCS instead of stable CAD. |
| This change emphasizes the fact that the clinical presentations of CAD can be categorized as either ACS or CCS. CAD is a dynamic process of atherosclerotic plaque accumulation and functional alterations of coronary circulation that can be modified by lifestyle, pharmacological therapies, and revascularization, which result in disease stabilization or regression. |
| In the current Guidelines on CCS, six clinical scenarios most frequently encountered in patients are identified: (i) patients with suspected CAD and ‘stable’ anginal symptoms, and/or dyspnoea; (ii) patients with new onset of HF or LV dysfunction and suspected CAD; (iii) asymptomatic and symptomatic patients with stabilized symptoms <1 year after an ACS or patients with recent revascularization; (iv) asymptomatic and symptomatic patients >1 year after initial diagnosis or revascularization; (v) patients with angina and suspected vasospastic or microvascular disease; (vi) asymptomatic subjects in whom CAD is detected at screening. |
| The PTP of CAD based on age, gender and nature of symptoms have undergone major revisions. In addition, we introduced a new phrase 'Clinical likelihood of CAD' that utilizes also various risk factors of CAD as PTP modifiers. The application of various diagnostic tests in different patient groups to rule-in or rule-out CAD have been updated. |
| The Guidelines emphasize the crucial role of healthy lifestyle behaviours and other preventive actions in decreasing the risk of subsequent cardiovascular events and mortality. |
| New/revised concepts in 2019 |
| The Guidelines have been revised to focus on CCS instead of stable CAD. |
| This change emphasizes the fact that the clinical presentations of CAD can be categorized as either ACS or CCS. CAD is a dynamic process of atherosclerotic plaque accumulation and functional alterations of coronary circulation that can be modified by lifestyle, pharmacological therapies, and revascularization, which result in disease stabilization or regression. |
| In the current Guidelines on CCS, six clinical scenarios most frequently encountered in patients are identified: (i) patients with suspected CAD and ‘stable’ anginal symptoms, and/or dyspnoea; (ii) patients with new onset of HF or LV dysfunction and suspected CAD; (iii) asymptomatic and symptomatic patients with stabilized symptoms <1 year after an ACS or patients with recent revascularization; (iv) asymptomatic and symptomatic patients >1 year after initial diagnosis or revascularization; (v) patients with angina and suspected vasospastic or microvascular disease; (vi) asymptomatic subjects in whom CAD is detected at screening. |
| The PTP of CAD based on age, gender and nature of symptoms have undergone major revisions. In addition, we introduced a new phrase 'Clinical likelihood of CAD' that utilizes also various risk factors of CAD as PTP modifiers. The application of various diagnostic tests in different patient groups to rule-in or rule-out CAD have been updated. |
| The Guidelines emphasize the crucial role of healthy lifestyle behaviours and other preventive actions in decreasing the risk of subsequent cardiovascular events and mortality. |
ACS = acute coronary syndromes; CAD = coronary artery disease; CCS = chronic coronary syndromes; HF = heart failure; LV = left ventricular; PTP = pre-test probability.
| New/revised concepts in 2019 |
| The Guidelines have been revised to focus on CCS instead of stable CAD. |
| This change emphasizes the fact that the clinical presentations of CAD can be categorized as either ACS or CCS. CAD is a dynamic process of atherosclerotic plaque accumulation and functional alterations of coronary circulation that can be modified by lifestyle, pharmacological therapies, and revascularization, which result in disease stabilization or regression. |
| In the current Guidelines on CCS, six clinical scenarios most frequently encountered in patients are identified: (i) patients with suspected CAD and ‘stable’ anginal symptoms, and/or dyspnoea; (ii) patients with new onset of HF or LV dysfunction and suspected CAD; (iii) asymptomatic and symptomatic patients with stabilized symptoms <1 year after an ACS or patients with recent revascularization; (iv) asymptomatic and symptomatic patients >1 year after initial diagnosis or revascularization; (v) patients with angina and suspected vasospastic or microvascular disease; (vi) asymptomatic subjects in whom CAD is detected at screening. |
| The PTP of CAD based on age, gender and nature of symptoms have undergone major revisions. In addition, we introduced a new phrase 'Clinical likelihood of CAD' that utilizes also various risk factors of CAD as PTP modifiers. The application of various diagnostic tests in different patient groups to rule-in or rule-out CAD have been updated. |
| The Guidelines emphasize the crucial role of healthy lifestyle behaviours and other preventive actions in decreasing the risk of subsequent cardiovascular events and mortality. |
| New/revised concepts in 2019 |
| The Guidelines have been revised to focus on CCS instead of stable CAD. |
| This change emphasizes the fact that the clinical presentations of CAD can be categorized as either ACS or CCS. CAD is a dynamic process of atherosclerotic plaque accumulation and functional alterations of coronary circulation that can be modified by lifestyle, pharmacological therapies, and revascularization, which result in disease stabilization or regression. |
| In the current Guidelines on CCS, six clinical scenarios most frequently encountered in patients are identified: (i) patients with suspected CAD and ‘stable’ anginal symptoms, and/or dyspnoea; (ii) patients with new onset of HF or LV dysfunction and suspected CAD; (iii) asymptomatic and symptomatic patients with stabilized symptoms <1 year after an ACS or patients with recent revascularization; (iv) asymptomatic and symptomatic patients >1 year after initial diagnosis or revascularization; (v) patients with angina and suspected vasospastic or microvascular disease; (vi) asymptomatic subjects in whom CAD is detected at screening. |
| The PTP of CAD based on age, gender and nature of symptoms have undergone major revisions. In addition, we introduced a new phrase 'Clinical likelihood of CAD' that utilizes also various risk factors of CAD as PTP modifiers. The application of various diagnostic tests in different patient groups to rule-in or rule-out CAD have been updated. |
| The Guidelines emphasize the crucial role of healthy lifestyle behaviours and other preventive actions in decreasing the risk of subsequent cardiovascular events and mortality. |
ACS = acute coronary syndromes; CAD = coronary artery disease; CCS = chronic coronary syndromes; HF = heart failure; LV = left ventricular; PTP = pre-test probability.
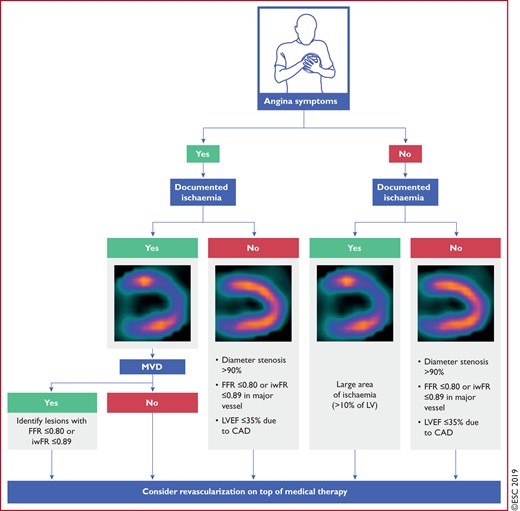
 |
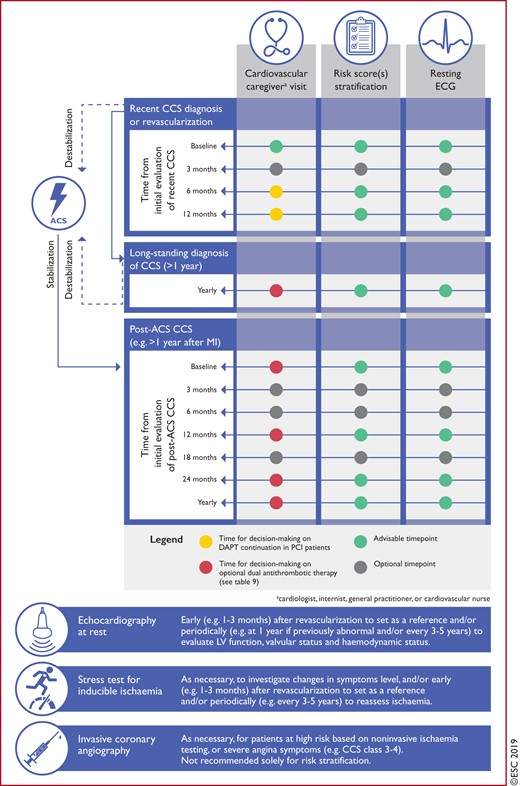
 |
 |
 |
aClass of recommendation.
ACE = angiotensin-converting enzyme; ACS = acute coronary syndromes; AF = atrial fibrillation; b.i.d. = bis in die (twice a day); CAD = coronary artery disease; CCS = chronic coronary syndromes; CHA2DS2-VASc = Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]; CTA = computed tomography angiography; CVD = cardiovascular disease; HF = heart failure; IMT = intima−media thickness; LV = left ventricular; NOAC = non-vitamin K antagonist oral anticoagulant; OAC = oral anticoagulant; o.d. = omni die (once a day); PCI = percutaneous coronary intervention; PCSK9 = proprotein convertase subtilisin-kexin type 9; VKA = vitamin K antagonist.
 |
 |
 |
 |
aClass of recommendation.
ACE = angiotensin-converting enzyme; ACS = acute coronary syndromes; AF = atrial fibrillation; b.i.d. = bis in die (twice a day); CAD = coronary artery disease; CCS = chronic coronary syndromes; CHA2DS2-VASc = Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]; CTA = computed tomography angiography; CVD = cardiovascular disease; HF = heart failure; IMT = intima−media thickness; LV = left ventricular; NOAC = non-vitamin K antagonist oral anticoagulant; OAC = oral anticoagulant; o.d. = omni die (once a day); PCI = percutaneous coronary intervention; PCSK9 = proprotein convertase subtilisin-kexin type 9; VKA = vitamin K antagonist.
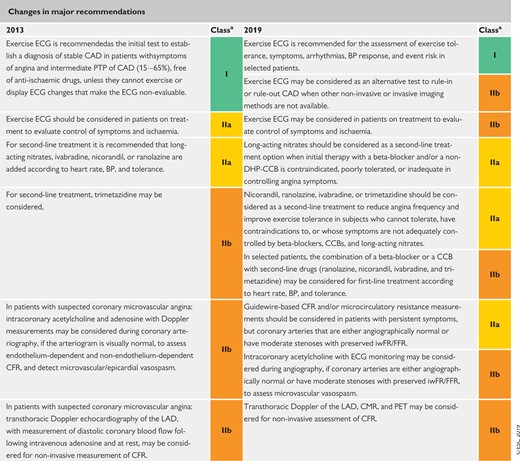 |
 |
aClass of recommendation.
BP = blood pressure; CAD = coronary artery disease; CCB = calcium channel blocker; CFR = coronary flow reserve; CMR = cardiac magnetic resonance; DHP-CCB = dihydropyridine calcium channel blockers; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio (instant flow reserve); LAD = left anterior descending; PET = positron emission tomography; PTP = pre-test probability.
 |
 |
aClass of recommendation.
BP = blood pressure; CAD = coronary artery disease; CCB = calcium channel blocker; CFR = coronary flow reserve; CMR = cardiac magnetic resonance; DHP-CCB = dihydropyridine calcium channel blockers; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio (instant flow reserve); LAD = left anterior descending; PET = positron emission tomography; PTP = pre-test probability.
3 Patients with angina and/or dyspnoea, and suspected coronary artery disease
3.1 Basic assessment, diagnosis, and risk assessment
The general approach for the initial diagnostic management of patients with angina and suspected obstructive CAD is presented in Figure 2. The diagnostic management approach includes six steps. The first step is to assess the symptoms and signs, to identify patients with possible unstable angina or other forms of ACS (step 1). In patients without unstable angina or other ACS, the next step is to evaluate the patient’s general condition and quality of life (step 2). Comorbidities that could potentially influence therapeutic decisions are assessed and other potential causes of the symptoms are considered. Step 3 includes basic testing and assessment of LV function. Thereafter, the clinical likelihood of obstructive CAD is estimated (step 4) and, on this basis, diagnostic testing is offered to selected patients to establish the diagnosis of CAD (step 5). Once a diagnosis of obstructive CAD has been confirmed, the patient’s event risk will be determined (step 6) as it has a major impact on the subsequent therapeutic decisions.
Approach for the initial diagnostic management of patients with angina and suspected coronary artery disease. ACS = acute coronary syndrome; BP = blood pressure; CAD = coronary artery disease; CTA = computed tomography angiography; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio; LVEF = left ventricular ejection fraction. aIf the diagnosis of CAD is uncertain, establishing a diagnosis using non-invasive functional imaging for myocardial ischaemia before treatment may be reasonable. bMay be omitted in very young and healthy patients with a high suspicion of an extracardiac cause of chest pain, and in multimorbid patients in whom the echocardiography result has no consequence for further patient management. cConsider exercise ECG to assess symptoms, arrhythmias, exercise tolerance, BP response, and event risk in selected patients. dAbility to exercise, individual test-related risks, and likelihood of obtaining diagnostic test result. eHigh clinical likelihood and symptoms inadequately responding to medical treatment, high event risk based on clinical evaluation (such as ST-segment depression, combined with symptoms at a low workload or systolic dysfunction indicating CAD), or uncertain diagnosis on non-invasive testing. fFunctional imaging for myocardial ischaemia if coronary CTA has shown CAD of uncertain grade or is non-diagnostic. gConsider also angina without obstructive disease in the epicardial coronary arteries (see section 6).
After these steps, appropriate therapies are to be initiated, which include lifestyle management (see section 3.2), medical therapy (see section 3.3), and revascularization when indicated (see section 3.4).
3.1.1. Step 1: Symptoms and signs
A careful history is the cornerstone of the diagnosis of angina. It is possible to achieve a high degree of certainty on a diagnosis based on history alone, although physical examination and objective tests are most often necessary to confirm the diagnosis, exclude alternative diagnoses, and assess the severity of underlying disease. The history should include any manifestation of cardiovascular disease (CVD) and risk factors (i.e. family history of CVD, dyslipidaemia, diabetes, hypertension, smoking, and other lifestyle factors).
The characteristics of discomfort related to myocardial ischaemia (angina pectoris) may be divided into four categories: location, character, duration, and relationship to exertion, and other exacerbating or relieving factors. The discomfort caused by myocardial ischaemia is usually located in the chest, near the sternum, but may be felt anywhere from the epigastrium to the lower jaw or teeth, between the shoulder blades, or in either arm to the wrist and fingers. The discomfort is often described as pressure, tightness, or heaviness; sometimes strangling, constricting, or burning. It may be useful to ask the patient directly about the presence of ‘discomfort’ as many do not feel ‘pain’ or ‘pressure’ in their chest. Shortness of breath may accompany angina, and chest discomfort may also be accompanied by less-specific symptoms such as fatigue or faintness, nausea, burning, restlessness, or a sense of impending doom. Shortness of breath may be the sole symptom of CAD and it may be difficult to differentiate this from shortness of breath caused by other conditions.
The duration of the discomfort is brief—≤10 min in the majority of cases, and more commonly just a few minutes or less—and chest pain lasting for seconds is unlikely to be due to CAD. An important characteristic is the relationship to exercise. Symptoms classically appear or become more severe with increased levels of exertion—such as walking up an incline or against a breeze, or in cold weather—and rapidly disappear within a few minutes when these causal factors abate. Exacerbations of symptoms after a heavy meal or after waking up in the morning are classic features of angina. Angina may paradoxically be reduced with further exercise (walk-through angina) or on second exertion (warm-up angina).1 Sublingual nitrates rapidly relieve angina. Symptoms are unrelated to respiration or position. The angina threshold, and hence symptoms, may vary considerably from day to day and even during the same day.
Definitions of typical and atypical angina are summarized in Table 3. The classification, although subjective, is practical and of proven value in determining the likelihood of obstructive CAD.2,3 Studies published since 2015 have reported that the majority of patients suspected of having CAD present with atypical or non-anginal chest pain,4–6 with as few as 10 − 15% presenting with typical angina.3,7,8 The Canadian Cardiovascular Society classification is still widely used as a grading system for angina,9 to quantify the threshold at which symptoms occur in relation to physical activities (Table 4).
Traditional clinical classification of suspected anginal symptoms
| Typical angina | Meets the following three characteristics: (i) Constricting discomfort in the front of the chest or in the neck, jaw, shoulder, or arm; (ii) Precipitated by physical exertion; (iii) Relieved by rest or nitrates within 5 min. |
| Atypical angina | Meets two of these characteristics. |
| Non-anginal chest pain | Meets only one or none of these characteristics. |
| Typical angina | Meets the following three characteristics: (i) Constricting discomfort in the front of the chest or in the neck, jaw, shoulder, or arm; (ii) Precipitated by physical exertion; (iii) Relieved by rest or nitrates within 5 min. |
| Atypical angina | Meets two of these characteristics. |
| Non-anginal chest pain | Meets only one or none of these characteristics. |
Traditional clinical classification of suspected anginal symptoms
| Typical angina | Meets the following three characteristics: (i) Constricting discomfort in the front of the chest or in the neck, jaw, shoulder, or arm; (ii) Precipitated by physical exertion; (iii) Relieved by rest or nitrates within 5 min. |
| Atypical angina | Meets two of these characteristics. |
| Non-anginal chest pain | Meets only one or none of these characteristics. |
| Typical angina | Meets the following three characteristics: (i) Constricting discomfort in the front of the chest or in the neck, jaw, shoulder, or arm; (ii) Precipitated by physical exertion; (iii) Relieved by rest or nitrates within 5 min. |
| Atypical angina | Meets two of these characteristics. |
| Non-anginal chest pain | Meets only one or none of these characteristics. |
Grading of effort angina severity according to the Canadian Cardiovascular Society
| Grade . | Description of angina severity . | . |
|---|---|---|
| I | Angina only with strenuous exertion | Presence of angina during strenuous, rapid, or prolonged ordinary activity (walking or climbing the stairs). |
| II | Angina with moderate exertion | Slight limitation of ordinary activities when they are performed rapidly, after meals, in cold, in wind, under emotional stress, or during the first few hours after waking up, but also walking uphill, climbing more than one flight of ordinary stairs at a normal pace, and in normal conditions. |
| III | Angina with mild exertion | Having difficulties walking one or two blocks, or climbing one flight of stairs, at normal pace and conditions. |
| IV | Angina at rest | No exertion needed to trigger angina. |
| Grade . | Description of angina severity . | . |
|---|---|---|
| I | Angina only with strenuous exertion | Presence of angina during strenuous, rapid, or prolonged ordinary activity (walking or climbing the stairs). |
| II | Angina with moderate exertion | Slight limitation of ordinary activities when they are performed rapidly, after meals, in cold, in wind, under emotional stress, or during the first few hours after waking up, but also walking uphill, climbing more than one flight of ordinary stairs at a normal pace, and in normal conditions. |
| III | Angina with mild exertion | Having difficulties walking one or two blocks, or climbing one flight of stairs, at normal pace and conditions. |
| IV | Angina at rest | No exertion needed to trigger angina. |
Grading of effort angina severity according to the Canadian Cardiovascular Society
| Grade . | Description of angina severity . | . |
|---|---|---|
| I | Angina only with strenuous exertion | Presence of angina during strenuous, rapid, or prolonged ordinary activity (walking or climbing the stairs). |
| II | Angina with moderate exertion | Slight limitation of ordinary activities when they are performed rapidly, after meals, in cold, in wind, under emotional stress, or during the first few hours after waking up, but also walking uphill, climbing more than one flight of ordinary stairs at a normal pace, and in normal conditions. |
| III | Angina with mild exertion | Having difficulties walking one or two blocks, or climbing one flight of stairs, at normal pace and conditions. |
| IV | Angina at rest | No exertion needed to trigger angina. |
| Grade . | Description of angina severity . | . |
|---|---|---|
| I | Angina only with strenuous exertion | Presence of angina during strenuous, rapid, or prolonged ordinary activity (walking or climbing the stairs). |
| II | Angina with moderate exertion | Slight limitation of ordinary activities when they are performed rapidly, after meals, in cold, in wind, under emotional stress, or during the first few hours after waking up, but also walking uphill, climbing more than one flight of ordinary stairs at a normal pace, and in normal conditions. |
| III | Angina with mild exertion | Having difficulties walking one or two blocks, or climbing one flight of stairs, at normal pace and conditions. |
| IV | Angina at rest | No exertion needed to trigger angina. |
Physical examination of a patient with suspected CAD is important to assess the presence of anaemia, hypertension, valvular heart disease, hypertrophic cardiomyopathy, or arrhythmias. It is also recommended that practitioners obtain the body mass index (BMI) and search for evidence of non-coronary vascular disease, which may be asymptomatic [includes palpation of peripheral pulses, and auscultation of carotid and femoral arteries, as well as assessment of the ankle−brachial index (ABI)], and other signs of comorbid conditions such as thyroid disease, renal disease, or diabetes. This should be used in the context of other clinical information, such as the presence of cough or stinging pain, making CAD more unlikely. One should also try to reproduce the symptoms by palpation10 and test the effect of sublingual nitroglycerin in order to classify the symptoms (Table 3).
3.1.1.1 Stable vs. unstable angina
Unstable angina may present in one of three ways: (i) as rest angina, i.e. pain of characteristic nature and location occurring at rest and for prolonged periods (>20 min); (ii) new-onset angina, i.e. recent (2 months) onset of moderate-to-severe angina (Canadian Cardiovascular Society grade II or III); or (iii) crescendo angina, i.e. previous angina, which progressively increases in severity and intensity, and at a lower threshold, over a short period of time. Management of angina fulfilling these criteria is dealt with in the ESC Guidelines for the management of ACS.11,12 New-onset angina is generally regarded as unstable angina; however, if angina occurs for the first time with heavy exertion and subsides at rest, the suspected condition falls under the definition of CCS rather than unstable angina. In patients with unstable angina identified as being at low risk, it is recommended that the diagnostic and prognostic algorithms presented in these Guidelines be applied once the period of instability has subsided.11 Low-risk patients with unstable angina are characterized by no recurrence of angina, no signs of HF, no abnormalities in the initial or subsequent electrocardiogram (ECG), and no rise in troponin levels.11 In this setting, a non-invasive diagnostic strategy is recommended before deciding on an invasive strategy. Based on the definition above, stable and unstable angina may overlap, and many CCS patients pass through a period of experiencing unstable angina.
3.1.1.2 Distinction between symptoms caused by epicardial vs. microvascular/vasospastic disease
A distinction between symptoms caused by an epicardial stenosis and symptoms caused by microvascular or vasospastic disease cannot be made with reasonable certainty. Reliance on ischaemia testing or depiction of the coronary anatomy is often unavoidable to exclude obstructive CAD, which can be absent in symptomatic patients.13,14 A diagnostic workup for microvascular or vasospastic disease is discussed in section 6 of these Guidelines.
3.1.2 Step 2: Comorbidities and other causes of symptoms
Before any testing is considered, one must assess the patient’s general health, comorbidities, and quality of life. If revascularization is unlikely to be an acceptable option, further testing may be reduced to a clinically indicated minimum and appropriate therapy should be instituted, which may include a trial of antianginal medication even if a diagnosis of CAD has not been fully demonstrated. Non-invasive functional imaging for ischaemia may be an option if there is need to verify the diagnosis (Figure 2).
If the pain is clearly non-anginal, other diagnostic testing may be indicated to identify gastrointestinal, pulmonary, or musculoskeletal causes of chest pain. Nevertheless, these patients should also receive Guideline-based risk-factor modification based on commonly applied risk charts such as SCORE (Systematic COronary Risk Evaluation) (www.heartscore.org).15
3.1.3 Step 3: Basic testing
Basic (first-line) testing in patients with suspected CAD includes standard laboratory biochemical testing, a resting ECG, possible ambulatory ECG monitoring, resting echocardiography, and, in selected patients, a chest X-ray. Such testing can be done on an outpatient basis.
3.1.3.1 Biochemical tests
Laboratory investigations are used to identify possible causes of ischaemia, to establish cardiovascular risk factors and associated conditions, and to determine prognosis. Haemoglobin as part of a full blood count and—where there is a clinical suspicion of a thyroid disorder—thyroid hormone levels provide information related to possible causes of ischaemia. Fasting plasma glucose and glycated haemoglobin (HbA1c) should be measured in every patient with suspected CAD. If both are inconclusive, an additional oral glucose tolerance test is recommended.16 Knowledge of glucose metabolism is important because of the well-recognized association between diabetes and adverse cardiovascular outcome. Patients with diabetes should be managed according to specific Guidelines.15,16 A lipid profile, including total cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol (LDL-C), and triglycerides, should also be evaluated in any patient with suspected CAD to establish the patient’s risk profile and ascertain the need for treatment.15,17 To characterize severe dyslipidaemia or follow-up on high triglyceridaemia, fasting values are recommended.17
Peripheral artery disease (PAD) and renal dysfunction increase the likelihood of CAD, and have a negative impact on prognosis.18–20 Hence, baseline renal function should be evaluated with estimation of the glomerular filtration rate (GFR). It may also be reasonable to measure the uric acid level, as hyperuricaemia is a frequent comorbid condition and may also affect renal function.
If there is a clinical suspicion of CAD instability, biochemical markers of myocardial injury—such as troponin T or troponin I—should be measured, preferably using high-sensitivity assays, and management should follow the Guidelines for ACS without persistent ST-segment elevation.11 If high-sensitivity assays are employed, low levels of troponin can be detected in many patients with stable angina. Increased troponin levels are associated with adverse outcome21–25 and small studies have indicated a possible incremental value in diagnosing CAD,26,27 but larger trials are needed to verify the utility of systematic assessment in patients suspected of CAD. While multiple biomarkers may be useful for prognostication (see section 5), they do not yet have a role in diagnosing obstructive CAD.
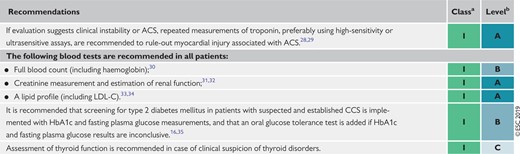
Basic biochemistry testing in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
ACS = acute coronary syndromes; CAD = coronary artery disease; CCS = chronic coronary syndromes; HbA1c = glycated haemoglobin; LDL-C = low-density lipoprotein cholesterol.
Class of recommendation.
Level of evidence.
Basic biochemistry testing in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
ACS = acute coronary syndromes; CAD = coronary artery disease; CCS = chronic coronary syndromes; HbA1c = glycated haemoglobin; LDL-C = low-density lipoprotein cholesterol.
Class of recommendation.
Level of evidence.
3.1.3.2 Resting electrocardiogram and ambulatory monitoring
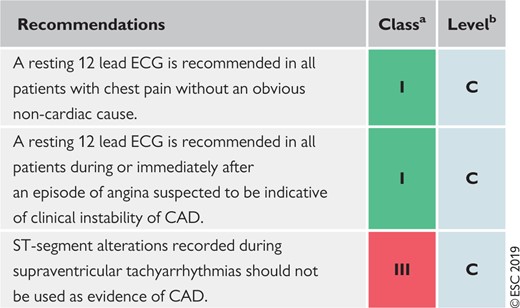
The paradigm of diagnosing myocardial ischaemia has, for almost a century, been based on the detection of repolarization abnormalities, mainly in the form of ST-segment depressions. Thus, the resting 12 lead ECG remains an indispensable component of the initial evaluation of a patient with chest pain without an obviously non-cardiac cause. Two scenarios of clinical evaluation are encountered: (i) a patient without symptoms of chest pain or discomfort, and (ii) a patient with ongoing anginal symptoms.
The former situation is far more prevalent and a normal resting ECG is frequently recorded. However, even in the absence of repolarization abnormalities, an ECG can demonstrate indirect signs of CAD, such as signs of previous MI (pathological Q waves) or conduction abnormalities [mainly left bundle branch block (LBBB) and impairment of atrioventricular conduction]. Atrial fibrillation (AF) is a frequent finding in patients with chest pain (usually atypical). ST-segment depression during supraventricular tachyarrhythmias is not predictive of obstructive CAD.36–39
The ECG can be crucial for diagnosing myocardial ischaemia if dynamic ST-segment changes are recorded during ongoing angina. The diagnosis of Prinzmetal and vasospastic angina is based on the detection of typical transient ST-segment elevation or depression during an angina attack (usually at rest).
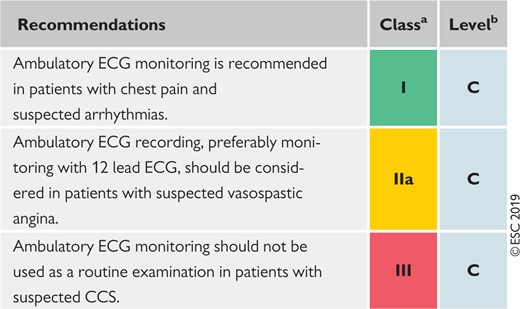
Long-term ambulatory ECG monitoring and recording should not be used to replace exercise testing; however, 12 lead ECG monitoring can be considered in selected patients to detect anginal episodes unrelated to physical exercise. Ambulatory ECG monitoring may reveal evidence of silent myocardial ischaemia in patients with CCS, but rarely adds relevant diagnostic or prognostic information that cannot be derived from stress testing.40 ECG changes suggesting ischaemia on ambulatory ECG monitoring are very frequent in women, but do not correlate with findings during stress testing.41 Most importantly, therapeutic strategies targeting silent ischaemia detected by ambulatory monitoring have not demonstrated clear survival benefits.42,43
Resting electrocardiogram in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CCS = chronic coronary syndromes; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
Resting electrocardiogram in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CCS = chronic coronary syndromes; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
Ambulatory electrocardiogram monitoring in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CCS = chronic coronary syndromes; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
Ambulatory electrocardiogram monitoring in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CCS = chronic coronary syndromes; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
3.1.3.3 Echocardiography and magnetic resonance imaging at rest
An echocardiographic study will provide important information about cardiac function and anatomy. LV ejection fraction (LVEF) is often normal in patients with CCS.44 A decreased LV function and/or regional wall motion abnormalities may increase the suspicion of ischaemic myocardial damage,45 and a pattern of LV dysfunction following the theoretical distribution territory of the coronary arteries is typical in patients who have already had an MI.46,47 The detection of regional wall motion abnormalities can challenging by visual assessment, and detection of early systolic lengthening, decreased systolic shortening, or post-systolic shortening by strain imaging techniques might be helpful in patients with apparently normal LV function but with clinical suspicion of CCS.48–50 Decreased diastolic LV function has been reported to be an early sign of ischaemic myocardial dysfunction and could also be indicative of microvascular dysfunction.51,52
Echocardiography is an important clinical tool for the exclusion of alternative causes of chest pain and also aids in diagnosing concurrent cardiac diseases, such as valvular heart diseases, HF, and most cardiomyopathies,53 but it is important to remember that these diseases often coexist with obstructive CAD. The use of an echocardiographic contrast agent can be useful in patients with poor acoustic windows.54
Cardiac magnetic resonance (CMR) may be considered in patients with suspected CAD when the echocardiogram (having used contrast) is inconclusive.55 CMR will provide useful information on cardiac anatomy and systolic cardiac function, similar to that from an echocardiogram, in patients with no contraindications for CMR. CMR can assess global and regional function,56 and the use of late gadolinium enhancement CMR can reveal a typical pattern of scarred myocardium in patients who have already experienced an MI.57
Assessment of LV function is important in all patients for risk stratification (see Supplementary Datasection 3.2) and should therefore be performed in all symptomatic patients with suspected CAD. Management of patients with either angina or HF symptoms, with reduced LVEF <40% or a mid-range reduced LVEF of 40−49%, is described in section 4 of the Guidelines.
Resting echocardiography and cardiac magnetic resonance in the initial diagnostic management of patients with suspected coronary artery disease
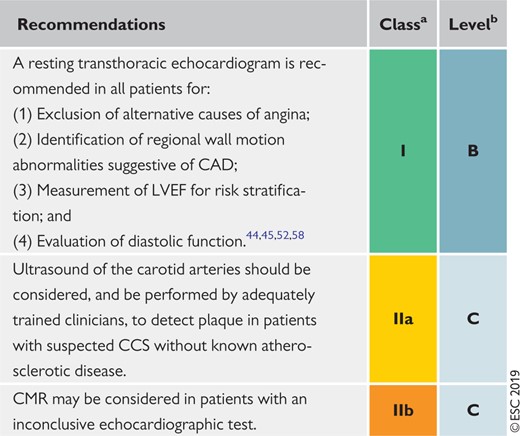 |
 |
CAD = coronary artery disease; CCS = chronic coronary syndromes; CMR = cardiac magnetic resonance imaging; LVEF = left ventricular ejection fraction.
Class of recommendation.
Level of evidence.
Resting echocardiography and cardiac magnetic resonance in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CCS = chronic coronary syndromes; CMR = cardiac magnetic resonance imaging; LVEF = left ventricular ejection fraction.
Class of recommendation.
Level of evidence.
3.1.3.4 Chest X-ray
Chest X-ray is frequently used in the assessment of patients with chest pain. However, in CCS, it does not provide specific information for diagnosis or event risk stratification. The test may occasionally be helpful in assessing patients with suspected HF. Chest X-ray may also be useful in patients with pulmonary problems, which often accompany CAD, or to rule-out another cause of chest pain in atypical presentations.
Chest X-ray in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
HF = heart failure.
Class of recommendation.
Level of evidence.
Chest X-ray in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
HF = heart failure.
Class of recommendation.
Level of evidence.
3.1.4 Step 4: Assessment of pre-test probability and clinical likelihood of coronary artery disease
The performance of the available methods in diagnosing obstructive CAD (i.e. the likelihood that the patient has disease if the test is abnormal, and the likelihood that the patient does not have disease if the test is normal) depends on the prevalence of disease in the population studied and, thus, the likelihood that a given patient will actually have CAD. Diagnostic testing is most useful when the likelihood is intermediate. When likelihood is high, a large number of patients need to be studied to identify the few patients that do not have disease, and a negative test result can seldom rule out the presence of obstructive CAD (i.e. the negative predictive value is low). When the likelihood is low, a negative test can rule out the disease, but the lower the likelihood, the higher the likelihood of a false-positive test (i.e. a positive test in the absence of obstructive CAD). In patients at the extreme ends of the probability range, it is therefore reasonable to refrain from diagnostic testing, and assume that the patient does or does not have obstructive CAD based on clinical evaluation alone.
The likelihood of obstructive CAD is influenced by the prevalence of the disease in the population studied, as well as by clinical features of an individual patient. A simple predictive model can be used to estimate the pre-test probability (PTP) of obstructive CAD based on age, sex, and the nature of symptoms.59 In the previous version of these Guidelines,60 estimation of the PTP was based on data gathered by Genders et al.,61 which updated previous data from Diamond and Forrester.59 Notably, the prevalence of disease for a given constellation of age, sex, and nature of symptoms was lower than in the Diamond and Forrester data. Since the previous version of the Guidelines was published, several studies have indicated that the prevalence of obstructive disease among patients with suspected CAD is lower than in the previous update.7,8,62,63
A pooled analysis64 of three contemporary study cohorts, including patients evaluated for suspected CAD,7,8,62 has indicated that the PTP based on age, sex, and symptoms is approximately one-third of that predicted by the model used in the previous version of the Guidelines.57,62 Overestimation of PTP is an important contributory factor to a low diagnostic yield of non-invasive and invasive testing. The new set of PTPs presented in Table 5 may substantially reduce the need for non-invasive and invasive tests in patients with suspected stable CAD. The table now also includes patients presenting with dyspnoea as their main symptom. However, it should be noted that the PTPs presented in Table 5 (as well as the PTP table in the previous version of the Guidelines) are based mainly on patients from countries with low CVD risk, and may vary between regions and countries.
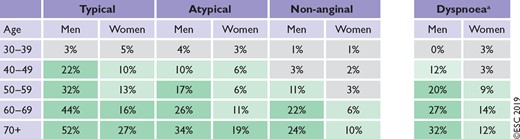 |
 |
CAD = coronary artery disease; PTP = pre-test probability.
In addition to the classic Diamond and Forrester classes,59 patients with dyspnoea only or dyspnoea as the primary symptom are included. The regions shaded dark green denote the groups in which non-invasive testing is most beneficial (PTP >15%). The regions shaded light green denote the groups with PTPs of CAD between 5–15%, in which testing for diagnosis may be considered after assessing the overall clinical likelihood based on the modifiers of PTPs presented in Figure 3.
 |
 |
CAD = coronary artery disease; PTP = pre-test probability.
In addition to the classic Diamond and Forrester classes,59 patients with dyspnoea only or dyspnoea as the primary symptom are included. The regions shaded dark green denote the groups in which non-invasive testing is most beneficial (PTP >15%). The regions shaded light green denote the groups with PTPs of CAD between 5–15%, in which testing for diagnosis may be considered after assessing the overall clinical likelihood based on the modifiers of PTPs presented in Figure 3.
Application of the new PTPs (Table 5) has important consequences for the referral of patients for diagnostic testing. If diagnostic testing was deferred in patients with a new PTP <15%, this would result in a large increase in the proportion of patients for whom diagnostic testing was not recommended, because more patients are classified as having a PTP <15%. In data derived from the PROMISE (Prospective Multicenter Imaging Study for Evaluation of Chest Pain) trial, 50% of patients previously classified as having an intermediate likelihood of obstructive CAD were reclassified to a PTP <15% according to the new PTP.62 In data derived from the pooled analysis64 (Table 5), 57% of all patients were classified to a PTP <15%.
Studies have shown that outcomes in patients classified with the new PTP <15% is good (annual risk of cardiovascular death or MI is <1%).7,62 Hence, it is safe to defer routine testing in patients with PTP <15%, thus reducing unnecessary procedures and costs.
Recent studies have also demonstrated that, when tested, the true observed prevalence of obstructive CAD has been <5% in patients who had a PTP <15% according to the 2013 version of these Guidelines.7,63 Therefore, this Task Force recognizes that the performance of diagnostic testing in patients with a new PTP of 5 − 15% more closely reflects current clinical practice and may be considered, particularly if symptoms are limiting and require clarification.7,63 Patient preference, local resources and the availability of tests, clinical judgement, and appropriate patient information remain important when making a decision to proceed with non-invasive diagnostic testing for an individual patient when the PTP is 5 − 15%, and the higher likelihood of a false-positive test must be considered. Patients with a PTP ≤5% can be assumed to have such a low probability of disease that diagnostic testing should be performed only for compelling reasons. Implementation of the new PTPs also indicates that patients should not be routinely referred directly to invasive assessment unless clinical or other data indicate a high likelihood of obstructive CAD.
Clinical models that incorporate information on risk factors for CVD, resting ECG changes, or coronary calcification have improved the identification of patients with obstructive CAD compared with age, sex, and symptoms alone.3,7,60,65–68 Therefore, the presence of risk factors for CVD (such as family history of CVD, dyslipidaemia, diabetes, hypertension, smoking, and other lifestyle factors) that increase the probability of obstructive CAD can be used as modifiers of the PTP estimate. If available, Q-wave, ST-segment, or T-wave changes on the ECG, LV dysfunction suggestive of ischaemia, and findings on exercise ECG, as well as information on coronary calcium obtained by computed tomography (CT), can be used to improve estimations of the PTP of obstructive CAD.3,69 In particular, the absence of coronary calcium (Agatston score = 0) is associated with a low prevalence of obstructive CAD (<5%), and low risk of death or non-fatal MI (<1% annual risk).69,70 However, it should be noted that coronary calcium imaging does not exclude coronary stenosis caused by a non-calcified atherosclerotic lesion,70 and the presence of coronary calcium is a weak predictor of obstructive CAD.69 Although the optimal use of these factors in improving PTP assessment has not yet been established, they should be considered in addition to the PTP based on sex, age, and the nature of symptoms to determine the overall clinical likelihood of obstructive CAD, as summarized in Figure 3. This is particularly important in refining the likelihood of CAD patients with a PTP of 5–15% based on age, sex, and the nature of symptoms.
Determinants of the clinical likelihood of obstructive coronary artery disease. CAD = coronary artery disease; CT = computed tomography, CVD = cardiovascular disease, ECG = electrocardiogram, LV = left ventricular; PTP = pre-test probability. aWhen available.
3.1.5 Step 5: Selecting appropriate testing
In patients in whom revascularization is futile due to comorbidities and overall quality of life, the diagnosis of CAD can be made clinically and only medical therapy is required. If the diagnosis of CAD is uncertain, establishing a diagnosis using non-invasive functional imaging for myocardial ischaemia before treatment is reasonable (Figure 2).
In a patient with a high clinical likelihood of CAD, symptoms unresponsive to medical therapy or typical angina at a low level of exercise, and an initial clinical evaluation (including echocardiogram and, in selected patients, exercise ECG) that indicates a high event risk, proceeding directly to invasive coronary angiography (ICA) without further diagnostic testing is a reasonable option. Under such circumstances, the indication for revascularization should be based on appropriate invasive confirmation of the haemodynamic significance of a stenosis.71,72
In other patients in whom CAD cannot be excluded by clinical assessment alone, non-invasive diagnostic tests are recommended to establish the diagnosis and assess the event risk. The current Guidelines recommend the use of either non-invasive functional imaging of ischaemia or anatomical imaging using coronary CT angiography (CTA) as the initial test for diagnosing CAD.
3.1.5.1 Functional non-invasive tests
Functional non-invasive tests for the diagnosis of obstructive CAD are designed to detect myocardial ischaemia through ECG changes, wall motion abnormalities by stress CMR or stress echocardiography, or perfusion changes by single-photon emission CT (SPECT), positron emission tomography (PET), myocardial contrast echocardiography, or contrast CMR. Ischaemia can be provoked by exercise or pharmacological stressors, either by increased myocardial work and oxygen demand, or by heterogeneity in myocardial perfusion by vasodilatation. Non-invasive functional tests are associated with high accuracy for the detection of flow-limiting coronary stenosis compared with invasive functional testing [fractional flow reserve (FFR)].73 However, lower-grade coronary atherosclerosis not linked with ischaemia remains undetected by functional testing and, in the presence of a negative functional test, patients should receive risk-factor modification based on commonly applied risk charts and recommendations.
3.1.5.2 Anatomical non-invasive evaluation
Anatomical non-invasive evaluation, by visualizing the coronary artery lumen and wall using an intravenous contrast agent, can be performed with coronary CTA, which provides high accuracy for the detection of obstructive coronary stenoses defined by ICA,73 because both tests are based on anatomy. However, stenoses estimated to be 50–90% by visual inspection are not necessarily functionally significant, i.e. they do not always induce myocardial ischaemia.73,74 Therefore, either non-invasive or invasive functional testing is recommended for further evaluation of angiographic stenosis detected by coronary CTA or invasive angiography, unless a very high-grade (>90% diameter stenosis) stenosis is detected via invasive angiography. The presence or absence of non-obstructive coronary atherosclerosis on coronary CTA provides prognostic information, and can be used to guide preventive therapy.75 The SCOT-HEART (Scottish Computed Tomography of the HEART) trial demonstrated a significantly lower rate of the combined endpoint of cardiovascular death or non-fatal MI (2.3 vs. 3.9% during 5 year follow-up) in patients in whom coronary CTA was performed in addition to routine testing, which consisted predominantly of exercise ECG.6 Other randomized, prospective clinical trials have demonstrated that diagnostic testing with coronary CTA is associated with clinical outcomes similar to those for functional imaging in patients with suspected CAD.4,6,76 In patients with extensive CAD, coronary CTA complemented by CT-based FFR was non-inferior to ICA and FFR for decision-making, and the identification of targets for revascularization.77
3.1.5.3 Role of the exercise electrocardiogram
Exercise ECG has inferior diagnostic performance compared with diagnostic imaging tests, and has limited power to rule-in or rule-out obstructive CAD.73 Since the publication of the previous version of these Guidelines, randomized clinical trials (RCTs) have compared the effects of diagnostic strategies based on exercise ECG or an imaging diagnostic test6,78,79 on clinical outcomes. These studies have shown that the addition of coronary CTA5,6,78,80 or functional imaging79 clarifies the diagnosis, enables the targeting of preventive therapies and interventions, and potentially reduces the risk of MI compared with an exercise ECG. Some, although not all, registry studies have also shown similar benefits regarding the use of an imaging diagnostic test in patients treated in everyday clinical practice.81,82 Therefore, these Guidelines recommend the use of an imaging diagnostic test instead of exercise ECG as the initial test for to diagnose obstructive CAD.
An exercise ECG alone may be considered as an alternative to diagnose obstructive CAD if imaging tests are not available, keeping in mind the risk of false-negative and false-positive test results.73,83 An exercise ECG is of no diagnostic value in patients with ECG abnormalities that prevent interpretation of the ST-segment changes during stress (i.e. LBBB, paced rhythm, Wolff−Parkinson−White syndrome, ≥0.1 mV ST-segment depression on resting ECG, or who are being treated with digitalis). An exercise ECG provides complementary clinically useful information beyond ECG changes and valuable prognostic information. Therefore, application of an exercise ECG may be considered in selected patients to complement clinical evaluation for the assessment of symptoms, ST-segment changes, exercise tolerance, arrhythmias, blood pressure (BP) response, and event risk.
3.1.5.4 Selection of diagnostic tests
Either a functional or anatomical test can be used to establish a diagnosis of obstructive CAD. A summary of the main diagnostic pathways is displayed in Figure 4. For revascularization decisions, information on both anatomy and ischaemia is needed.
Main diagnostic pathways in symptomatic patients with suspected obstructive coronary artery disease. Depending on clinical conditions and the healthcare environment, patient workup can start with either of three options: non-invasive testing, coronary computed tomography angiography, or invasive coronary angiography. Through each pathway, both functional and anatomical information is gathered to inform an appropriate diagnostic and therapeutic strategy. Risk-factor modification should be considered in all patients. CAD = coronary artery disease; CTA = computed tomography angiography; ECG = electrocardiogram; LV = left ventricular. aConsider microvascular angina. bAntianginal medications and/or risk-factor modification.
3.1.5.5 The impact of clinical likelihood on the selection of a diagnostic test
Each non-invasive diagnostic test has a particular range of clinical likelihood of obstructive CAD where the usefulness of its application is maximal. The likelihood ratios of the tests constitute useful parameters of their abilities to correctly classify patients, and can be used to facilitate the selection of the most useful test in any given patient.73,84 Given a clinical likelihood of obstructive CAD and the likelihood ratio of a particular test, one can assess the post-test probability of obstructive CAD after performing such a test. Using this approach, one can estimate the optimal ranges of clinical likelihood for each test, where they can reclassify patients from intermediate to either low or high post-test probability of CAD (Figure 5).73
Ranges of clinical likelihood of coronary artery disease in which a given test can rule-in (red) or rule-out (green) obstructive coronary artery disease. (A) Reference standard is anatomical assessment using invasive coronary angiography. (B) Reference standard is functional assessment using fractional flow reserve. Note in (B) that the data with stress echocardiography and single-photon emission computed tomography are more limited than with the other techniques.73 The crosshairs mark the mean values and their 95% confidence intervals. Figure adapted from Knuuti et al.73 CAD = coronary artery disease; CMR = cardiac magnetic resonance; CTA = computed tomography angiography; ECG = electrocardiogram; FFR = fractional flow reserve; ICA = invasive coronary angiography; PET = positron emission tomography; SPECT = single-photon emission computed tomography.
Coronary CTA is the preferred test in patients with a lower range of clinical likelihood of CAD, no previous diagnosis of CAD, and characteristics associated with a high likelihood of good image quality. It detects subclinical coronary atherosclerosis, but can also accurately rule out both anatomically and functionally significant CAD (Figure 5). It has higher accuracy values when low clinical likelihood populations are subjected to examination.85 Trials evaluating outcomes after coronary CTA to date have mostly included patients with a low clinical likelihood.4,5
The non-invasive functional tests for ischaemia typically have better rule-in power. In outcome trials, functional imaging tests have been associated with fewer referrals for downstream ICA compared with a strategy relying on anatomical imaging.55,76,86 Before revascularization decisions can be made, functional evaluation of ischaemia (either non-invasive or invasive) is required in most patients. Therefore, functional non-invasive testing may be preferred in patients at the higher end of the range of clinical likelihood if revascularization is likely or the patient has previously diagnosed CAD.
Patients in whom CAD is suspected, but who have a very low clinical likelihood (≤5%) of CAD, should have other cardiac causes of chest pain excluded and their cardiovascular risk factors adjusted, based on a risk-score assessment. In patients with repeated, unprovoked attacks of anginal symptoms mainly at rest, vasospastic angina should be considered, diagnosed, and treated appropriately (see section 6).
In addition to diagnostic accuracy and clinical likelihood, the selection of a non-invasive test depends on other patient characteristics, local expertise, and the availability of tests. Some diagnostic tests may perform better in some patients than others. For example, irregular heart rate and the presence of extensive coronary calcification are associated with increased likelihood of non-diagnostic image quality of coronary CTA, and it is not recommended in such patients.85 Stress echocardiography or SPECT perfusion imaging can be combined with dynamic exercise testing, and may be preferred if additional information available from the exercise test, such as exercise tolerance or heart rate response to exercise, is considered important. Exercise ECG cannot be used for diagnostic purposes in the presence of ECG abnormalities that prevent the evaluation of ischaemia. Risks related to different diagnostic tests need to be weighed against the benefits to the individual.87 For example, exposure to ionizing radiation associated with coronary CTA and nuclear perfusion imaging needs to be taken into account, especially in young individuals.87 Similarly, contraindications to pharmacological stressors and contrast agents (iodine-based contrast agents and gadolinium-based chelates) need to be taken into account. When testing is used appropriately, the clinical benefit from accurate diagnosis and therapy exceeds the projected risks of testing itself.87
3.1.5.6 Invasive testing
For diagnostic purposes, ICA is only necessary in patients with suspected CAD in cases of inconclusive non-invasive testing or, exceptionally, in patients from particular professions, due to regulatory issues.88 However, ICA may be indicated if non-invasive assessment suggests high event risk for determination of options for revascularization.88
In a patient with a high clinical likelihood of CAD, and symptoms unresponsive to medical therapy or with typical angina at a low level of exercise, and initial clinical evaluation indicates a high event risk, early ICA without previous non-invasive risk stratification may be reasonable to identify lesions potentially amenable to revascularization (Figure 4). Invasive functional assessment should complement ICA, especially in patients with coronary stenoses of 50 − 90% or multivessel disease, given the frequent mismatch between the angiographic and haemodynamic severities of coronary stenoses.89–91 Systematic integration of ICA with FFR has been shown to result in changes to the management strategies of 30 − 50% of patients undergoing elective ICA.92,93 Methods used to perform ICA have improved substantially, resulting in a reduction of complication rates with rapid ambulation. This is especially true for ICA performed via the radial artery.94 The composite rate of major complications associated with routine femoral diagnostic catheterization—mainly bleeding requiring blood transfusions—is still 0.5–2%.95 The composite rate of death, MI, or stroke is of the order of 0.1–0.2%.96 ICA should not be performed in patients with angina who refuse invasive procedures, prefer to avoid revascularization, who are not candidates for percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG), or in whom revascularization is not expected to improve functional status or quality of life. Intracoronary techniques for the diagnostic assessment of coronary anatomy are briefly mentioned in the Supplementary Data of this document.
Use of diagnostic imaging tests in the initial diagnostic management of symptomatic patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CT = computed tomography; CTA = computed tomography angiography.
Class of recommendation.
Level of evidence
Stress echocardiography, stress cardiac magnetic resonance, single-photon emission CT, or positron emission tomography.
Characteristics determining ability to exercise, likelihood of good image quality, expected radiation exposure, and risks or contraindications.
Use of diagnostic imaging tests in the initial diagnostic management of symptomatic patients with suspected coronary artery disease
 |
 |
CAD = coronary artery disease; CT = computed tomography; CTA = computed tomography angiography.
Class of recommendation.
Level of evidence
Stress echocardiography, stress cardiac magnetic resonance, single-photon emission CT, or positron emission tomography.
Characteristics determining ability to exercise, likelihood of good image quality, expected radiation exposure, and risks or contraindications.
Use of exercise electrocardiogram in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
BP = blood pressure; CAD = coronary artery disease; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
When this information will have an impact on diagnostic strategy or management.
Use of exercise electrocardiogram in the initial diagnostic management of patients with suspected coronary artery disease
 |
 |
BP = blood pressure; CAD = coronary artery disease; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
When this information will have an impact on diagnostic strategy or management.
3.1.6 Step 6: Assessment of event risk
Assessment of event risk is recommended in every patient being evaluated for suspected CAD or with a newly diagnosed CAD, as it has major impacts on therapy decisions. The process of risk stratification serves to identify patients at high event risk who will benefit from revascularization beyond the amelioration of symptoms. Event risk stratification is usually based on the assessments used to make a diagnosis of CAD. All patients should undergo cardiovascular event risk stratification using clinical evaluation, the assessment of LV function by resting echocardiography, and, in the majority of cases, non-invasive assessment of ischaemia or coronary anatomy. Although the diagnostic value of an exercise ECG is limited,73 the occurrence of ST-segment depression at a low workload combined with exertional symptoms (angina or dyspnoea), low exercise capacity, complex ventricular ectopy, or arrhythmias and abnormal BP response are markers of a high risk of cardiac mortality.97–100 Patients with typical angina and LV systolic dysfunction in a pattern that indicates CAD are also at high risk of cardiac mortality.101 ICA for risk stratification will only be required in a selected subgroup of patients and additional FFR may be required for event risk stratification as appropriate (Figure 4). Risk assessment in patients with HF and LV dysfunction, asymptomatic patients with known CAD, and patients with recurrent symptoms after previous coronary intervention is discussed in sections 4 and 5.
3.1.6.1 Definition of levels of risk
In patients with established CCS, the risk of annual cardiac mortality is used to describe the event risk. As in the previous version of the Guidelines,60 high event risk is defined as a cardiac mortality rate >3% per year and low event risk as a cardiac mortality rate <1% per year. The definitions of high event risk based on findings of diagnostic tests in symptomatic patients or in patients with established CCS are shown in Table 6.
| Exercise ECG | Cardiovascular mortality >3% per year according to Duke Treadmill Score |
| SPECT or PET perfusion imaging | Area of ischaemia ≥10% of the left ventricle myocardium |
| Stress echocardiography | ≥3 of 16 segments with stress-induced hypokinesia or akinesia |
| CMR | ≥2 of 16 segments with stress perfusion defects or ≥3 dobutamine-induced dysfunctional segments |
| Coronary CTA or ICA | Three-vessel disease with proximal stenoses, LM disease, or proximal anterior descending disease |
| Invasive functional testing | FFR ≤0.8, iwFR ≤0.89 |
| Exercise ECG | Cardiovascular mortality >3% per year according to Duke Treadmill Score |
| SPECT or PET perfusion imaging | Area of ischaemia ≥10% of the left ventricle myocardium |
| Stress echocardiography | ≥3 of 16 segments with stress-induced hypokinesia or akinesia |
| CMR | ≥2 of 16 segments with stress perfusion defects or ≥3 dobutamine-induced dysfunctional segments |
| Coronary CTA or ICA | Three-vessel disease with proximal stenoses, LM disease, or proximal anterior descending disease |
| Invasive functional testing | FFR ≤0.8, iwFR ≤0.89 |
CTA = computed tomography angiography; CMR = cardiac magnetic resonance; ECG = electrocardiogram; FFR = fractional flow reserve; ICA = invasive coronary angiography; iwFR = instantaneous wave-free ration (instant flow reserve); LM = left main; PET = positron emission tomography; SPECT; single-photon emission computed tomography.
For detailed explanations, refer to the Supplementary Data.
| Exercise ECG | Cardiovascular mortality >3% per year according to Duke Treadmill Score |
| SPECT or PET perfusion imaging | Area of ischaemia ≥10% of the left ventricle myocardium |
| Stress echocardiography | ≥3 of 16 segments with stress-induced hypokinesia or akinesia |
| CMR | ≥2 of 16 segments with stress perfusion defects or ≥3 dobutamine-induced dysfunctional segments |
| Coronary CTA or ICA | Three-vessel disease with proximal stenoses, LM disease, or proximal anterior descending disease |
| Invasive functional testing | FFR ≤0.8, iwFR ≤0.89 |
| Exercise ECG | Cardiovascular mortality >3% per year according to Duke Treadmill Score |
| SPECT or PET perfusion imaging | Area of ischaemia ≥10% of the left ventricle myocardium |
| Stress echocardiography | ≥3 of 16 segments with stress-induced hypokinesia or akinesia |
| CMR | ≥2 of 16 segments with stress perfusion defects or ≥3 dobutamine-induced dysfunctional segments |
| Coronary CTA or ICA | Three-vessel disease with proximal stenoses, LM disease, or proximal anterior descending disease |
| Invasive functional testing | FFR ≤0.8, iwFR ≤0.89 |
CTA = computed tomography angiography; CMR = cardiac magnetic resonance; ECG = electrocardiogram; FFR = fractional flow reserve; ICA = invasive coronary angiography; iwFR = instantaneous wave-free ration (instant flow reserve); LM = left main; PET = positron emission tomography; SPECT; single-photon emission computed tomography.
For detailed explanations, refer to the Supplementary Data.
Notably, the level of risk is different from the risk assessment based on SCORE in asymptomatic individuals without diabetes who are apparently healthy (see section 7). SCORE defines 10 year cardiovascular mortality in asymptomatic subjects. Differences in these risk-assessment tools and the scales are illustrated in Figure 6. The findings of different test modalities that correspond to high event risk are presented in Table 6 and are discussed in more detail in the Supplementary Data (sections 1.1 and 1.2).102–104 For all non-invasive tests presented in Table 6, a normal test result is associated with a low event risk.105
Comparison of risk assessments in asymptomatic apparently healthy subjects (primary prevention) and patients with established chronic coronary syndromes (secondary prevention). Note that in asymptomatic subjects (left panel), SCORE estimates 10 year cardiovascular mortality, while in symptomatic patients (right panel), annual cardiac mortality is estimated. CCS = chronic coronary syndromes; SCORE = Systematic COronary Risk Evaluation.
Recommendations on risk assessment
 |
 |
CAD = coronary artery disease; CTA = computed tomography angiography; ECG = electrocardiogram; FFR = fractional flow reserve; ICA = invasive coronary angiography; iwFR = instantaneous wave-free ratio; LM = left main; LV = left ventricular; LVEF = LV ejection fraction.
Class of recommendation.
Level of evidence.
Recommendations on risk assessment
 |
 |
CAD = coronary artery disease; CTA = computed tomography angiography; ECG = electrocardiogram; FFR = fractional flow reserve; ICA = invasive coronary angiography; iwFR = instantaneous wave-free ratio; LM = left main; LV = left ventricular; LVEF = LV ejection fraction.
Class of recommendation.
Level of evidence.
3.2 Lifestyle management
3.2.1 General management of patients with coronary artery disease
General management of CCS aims to reduce symptoms and improve prognosis through appropriate medications and interventions, and to control risk factors including lifestyle behaviours. Optimal medical therapy in the COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial included the promotion of medication adherence, behavioural counselling, and support for the managing lifestyle risk factors delivered by nurse case managers.117 Achievement of optimal management may be best accomplished through a multidisciplinary team approach that can provide tailored and flexible support to patients.
Patient-reported outcome measures can provide relevant and systematic information about patients’ symptoms, functioning, and concerns. Patient-reported outcome measures are increasingly being implemented sequentially in healthcare, and have been shown to improve clinical care and patient experiences, communication between providers and patients (including sensitive subjects), save time in consultations, and improve provider satisfaction.118
3.2.2 Lifestyle modification and control of risk factors
Implementing healthy lifestyle behaviours decreases the risk of subsequent cardiovascular events and mortality, and is additional to appropriate secondary prevention therapy. Lifestyle recommendations and interventions are described in more detail in the 2016 ESC Guidelines on CVD prevention in clinical practice.15 Lifestyle factors are important and the implementation of healthy behaviours (including smoking cessation, recommended physical activity, a healthy diet, and maintaining a healthy weight; see Table 7) significantly decreases the risk of future cardiovascular events and death, even when controlling for evidence-based secondary prevention therapy and interventions.119–122 Benefits are evident as early as 6 months after an index event.119
Lifestyle recommendations for patients with chronic coronary syndromes
| Lifestyle factor . | . |
|---|---|
| Smoking cessation | Use pharmacological and behavioural strategies to help patients quit smoking. Avoid passive smoking. |
| Healthy diet | Diet high in vegetables, fruit, and wholegrains. Limit saturated fat to <10% of total intake. Limit alcohol to <100 g/week or 15 g/day. |
| Physical activity | 30 − 60 min moderate physical activity most days, but even irregular activity is beneficial. |
| Healthy weight | Obtain and maintain a healthy weight (<25 kg/m2), or reduce weight through recommended energy intake and increased physical activity. |
| Other | Take medications as prescribed. Sexual activity is low risk for stable patients not symptomatic at low-to-moderate activity levels. |
| Lifestyle factor . | . |
|---|---|
| Smoking cessation | Use pharmacological and behavioural strategies to help patients quit smoking. Avoid passive smoking. |
| Healthy diet | Diet high in vegetables, fruit, and wholegrains. Limit saturated fat to <10% of total intake. Limit alcohol to <100 g/week or 15 g/day. |
| Physical activity | 30 − 60 min moderate physical activity most days, but even irregular activity is beneficial. |
| Healthy weight | Obtain and maintain a healthy weight (<25 kg/m2), or reduce weight through recommended energy intake and increased physical activity. |
| Other | Take medications as prescribed. Sexual activity is low risk for stable patients not symptomatic at low-to-moderate activity levels. |
Lifestyle recommendations for patients with chronic coronary syndromes
| Lifestyle factor . | . |
|---|---|
| Smoking cessation | Use pharmacological and behavioural strategies to help patients quit smoking. Avoid passive smoking. |
| Healthy diet | Diet high in vegetables, fruit, and wholegrains. Limit saturated fat to <10% of total intake. Limit alcohol to <100 g/week or 15 g/day. |
| Physical activity | 30 − 60 min moderate physical activity most days, but even irregular activity is beneficial. |
| Healthy weight | Obtain and maintain a healthy weight (<25 kg/m2), or reduce weight through recommended energy intake and increased physical activity. |
| Other | Take medications as prescribed. Sexual activity is low risk for stable patients not symptomatic at low-to-moderate activity levels. |
| Lifestyle factor . | . |
|---|---|
| Smoking cessation | Use pharmacological and behavioural strategies to help patients quit smoking. Avoid passive smoking. |
| Healthy diet | Diet high in vegetables, fruit, and wholegrains. Limit saturated fat to <10% of total intake. Limit alcohol to <100 g/week or 15 g/day. |
| Physical activity | 30 − 60 min moderate physical activity most days, but even irregular activity is beneficial. |
| Healthy weight | Obtain and maintain a healthy weight (<25 kg/m2), or reduce weight through recommended energy intake and increased physical activity. |
| Other | Take medications as prescribed. Sexual activity is low risk for stable patients not symptomatic at low-to-moderate activity levels. |
Primary care providers have an important role to play in prevention. The primary care arm of the EUROACTION cluster randomized trial demonstrated that a nurse-co-ordinated programme in primary care was more effective in helping patients achieve lifestyle and risk-factor goals than usual care.123 Practice nurses in the Netherlands were found to be as effective as general practitioners in decreasing cardiovascular risk in another randomized study.123
3.2.2.1 Smoking
Smoking cessation improves the prognosis in patients with CCS, including a 36% risk reduction in mortality for those who quit.124 Measures to promote smoking cessation include brief advice, counselling and behavioural interventions, and pharmacological therapy including nicotine replacement. Patients should also avoid passive smoking.
Brief advice, relative to no treatment, doubles the likelihood of smoking cessation in the short-term, but more intensive advice and support (behavioural interventions, telephone support, or self-help measures) is more effective than brief advice, especially if continued over 1 month.125,126 All forms of nicotine-replacement therapy, bupropion, and varenicline are more effective in increasing smoking cessation than control, and combining behavioural and pharmacological approaches is effective and highly recommended.125 A network meta-analysis of 63 clinical trials (including eight trials in CVD patients) found no increase in major adverse cardiovascular events linked to nicotine-replacement therapy, bupropion, or varenicline.127 Nicotine-replacement therapy was associated with minor events such as arrhythmias and angina, and bupropion appeared to have a protective effect against major adverse cardiovascular events.127 The use of e-cigarettes is considered to be a reduced-harm alternative to conventional cigarettes, but they are not harm-free. Newer devices can deliver higher nicotine contents, and e-cigarettes emit other constituents such as carbonyls and fine and ultrafine particulates.128 Although previous systematic reviews have found very limited and inconsistent evidence that e-cigarettes (primarily first-generation devices) are useful in improve smoking cessation compared with placebo or nicotine-replacement therapy, a recent large clinical trial found e-cigarettes to be more effective than nicotine-replacement therapy in smoking cessation.129–133 In this randomised trial of 886 smokers, those assigned to e-cigarettes had a sustained 1 year abstinence rate of 18% compared with 9.9% for nicotine-replacement therapy [relative risk, 1.83; 95% confidence interval (CI) 1.30 to 2.58; P < 0.001].133
In clinical encounters with smokers, clinicians should follow the ‘Five As’: ask about smoking, advise to quit, assess readiness to quit, assist with smoking cessation (pharmacological support and referral for behavioural counselling), and arrange follow-up (Figure 7).
3.2.2.2 Diet and alcohol
Unhealthy diets are a leading contributor to CAD and its progression, and changes to healthy eating patterns in patients with CCS have resulted in a reduction in mortality and cardiovascular events134 (recommended diet characteristics are detailed in Table 8).
| Characteristics . |
|---|
| Increase consumption of fruits and vegetables (≥200 g each per day). |
| 35–45 g of fibre per day, preferably from wholegrains. |
| Moderate consumption of nuts (30 g per day, unsalted). |
| 1–2 servings of fish per week (one to be oily fish). |
| Limited lean meat, low-fat dairy products, and liquid vegetable oils. |
| Saturated fats to account for <10% of total energy intake; replace with polyunsaturated fats. |
| As little intake of trans unsaturated fats as possible, preferably no intake from processed food, and <1% of total energy intake. |
| ≤5–6 g of salt per day. |
| If alcohol is consumed, limiting intake to ≤100 g/week or <15 g/day is recommended. |
| Avoid energy-dense foods such as sugar-sweetened soft drinks. |
| Characteristics . |
|---|
| Increase consumption of fruits and vegetables (≥200 g each per day). |
| 35–45 g of fibre per day, preferably from wholegrains. |
| Moderate consumption of nuts (30 g per day, unsalted). |
| 1–2 servings of fish per week (one to be oily fish). |
| Limited lean meat, low-fat dairy products, and liquid vegetable oils. |
| Saturated fats to account for <10% of total energy intake; replace with polyunsaturated fats. |
| As little intake of trans unsaturated fats as possible, preferably no intake from processed food, and <1% of total energy intake. |
| ≤5–6 g of salt per day. |
| If alcohol is consumed, limiting intake to ≤100 g/week or <15 g/day is recommended. |
| Avoid energy-dense foods such as sugar-sweetened soft drinks. |
| Characteristics . |
|---|
| Increase consumption of fruits and vegetables (≥200 g each per day). |
| 35–45 g of fibre per day, preferably from wholegrains. |
| Moderate consumption of nuts (30 g per day, unsalted). |
| 1–2 servings of fish per week (one to be oily fish). |
| Limited lean meat, low-fat dairy products, and liquid vegetable oils. |
| Saturated fats to account for <10% of total energy intake; replace with polyunsaturated fats. |
| As little intake of trans unsaturated fats as possible, preferably no intake from processed food, and <1% of total energy intake. |
| ≤5–6 g of salt per day. |
| If alcohol is consumed, limiting intake to ≤100 g/week or <15 g/day is recommended. |
| Avoid energy-dense foods such as sugar-sweetened soft drinks. |
| Characteristics . |
|---|
| Increase consumption of fruits and vegetables (≥200 g each per day). |
| 35–45 g of fibre per day, preferably from wholegrains. |
| Moderate consumption of nuts (30 g per day, unsalted). |
| 1–2 servings of fish per week (one to be oily fish). |
| Limited lean meat, low-fat dairy products, and liquid vegetable oils. |
| Saturated fats to account for <10% of total energy intake; replace with polyunsaturated fats. |
| As little intake of trans unsaturated fats as possible, preferably no intake from processed food, and <1% of total energy intake. |
| ≤5–6 g of salt per day. |
| If alcohol is consumed, limiting intake to ≤100 g/week or <15 g/day is recommended. |
| Avoid energy-dense foods such as sugar-sweetened soft drinks. |
A Mediterranean dietary pattern high in fruit, vegetables, legumes, fibre, polyunsaturated fats, nuts, and fish, avoiding or limiting refined carbohydrates, red meat, dairy, and saturated fat, is advocated.135–138 Although light-to-moderate alcohol intake (1–2 drinks per day) does not increase risk of MI, levels >100 g per week were associated with higher all-cause and other CVD mortality in a large individual-data meta-analysis.139 The Global Burden of Disease 1990–2016 analysis concluded that zero alcohol intake was the level at which risk for death and disability was minimized.140
3.2.2.3 Weight management
In a population-based study, lifetime risk of incident CVD, and cardiovascular morbidity and mortality, were higher in those who were overweight or obese compared with those with a normal BMI (20 − 25 kg/m2). Obesity was associated with a shorter overall lifespan, and overweight was associated with developing CVD at an earlier age.143 Waist circumference is a marker of central obesity and is strongly associated with developing CVD and diabetes. Waist circumference ≤94 cm for men (<90 cm for South Asian and Asian men) and ≤80 cm for women is recommended.
In subjects with CAD, intentional weight loss is associated with a significantly lower risk of adverse clinical outcomes.144 Although there has been much argument regarding the relative benefits of low-fat vs. low-carbohydrate diets, Gardner et al.145 found similar weight loss and benefits in patients randomized to either healthy low-fat or low-carbohydrate diets. This finding held, regardless of patients’ genotype patterns and baseline insulin secretion. Healthy diets with energy intake limited to the amount needed to obtain and maintain a healthy weight (BMI <25 kg/m2), and increasing physical activity, are recommended for weight management.
3.2.2.4 Physical activity
Exercise has been referred to as a ‘polypill’ due to its numerous beneficial effects on cardiovascular risk factors and cardiovascular system physiology.146,147 Exercise improves angina through enhanced oxygen delivery to the myocardium, and increasing exercise capacity is an independent predictor of increased survival among men and women with CCS, even among those with a regimen consistent with evidence-based management.122,147,148
Every 1 mL/kg/min increase in exercise peak oxygen consumption was associated with a 14–17% reduction of risk for cardiovascular and all-cause death in women and men.122
Physical activity recommendations for patients with CCS are 30–60 min of moderate-intensity aerobic activity ≥5 days per week.147 Even irregular leisure-time physical activity decreases mortality risk among previously sedentary patients,149 and increasing activity is associated with lower cardiovascular mortality.150 Previously sedentary patients will need support to work up to 30–60 min most days, reassurance that exercise is beneficial, and education regarding what to do if angina occurs while being active. Resistance exercises maintain muscle mass, strength, and function and, with aerobic activity, have benefits regarding insulin sensitivity and control of lipids and BP.
3.2.2.5 Cardiac rehabilitation
Exercise-based cardiac rehabilitation has consistently demonstrated its effectiveness in reducing cardiovascular mortality and hospitalizations compared with no exercise controls in patients with CAD, and this benefit persists into the modern era.151–153 Most patients participating in cardiac rehabilitation are referred following an acute MI or after revascularization, with 0 − 24% of patients found to be referred for CCS in 12 European countries.154 Importantly, the benefits of cardiac rehabilitation occur across diagnostic categories.151–153
3.2.2.6 Psychosocial factors
Patients with heart disease have a two-fold increased risk of mood and anxiety disorders compared with people without heart disease.155,156 Psychosocial stress, depression, and anxiety are associated with worse outcomes, and make it difficult for patients to make positive changes to their lifestyles or adhere to a therapeutic regimen. The ESC Prevention Guidelines recommend assessment for psychosocial risk factors.15 Clinical trials have shown that psychological (e.g. counselling and/or cognitive behavioural therapy) and pharmacological interventions have a beneficial effect on depression, anxiety, and stress, with some evidence of a reduction in cardiac mortality and events compared with placebo.157–159
3.2.2.7 Environmental factors
Air pollutants are estimated to be one of the 10 leading risk factors for global mortality. Exposure to air pollution increases risk of MI, as well as hospitalization and death from heart failure, stroke, and arrhythmia.160 Patients with CCS should avoid heavily traffic-congested areas. Air purifiers with high-efficiency particulate air (‘HEPA’) filters reduce indoor pollution, and wearing N95 respirator face masks in heavily polluted areas has been shown to be protective.160 Environmental noise also increases the risk of CVD.161 Policies and regulations that reduce air pollution and environmental noise should be supported, and patients should be advised about these risks.
3.2.2.8 Sexual activity
Patients with CCS often worry about the cardiovascular risk associated with sexual activity and/or experience sexual dysfunction.162 The risk of triggering sudden death or an acute MI is very low, especially when sexual activity is with a stable partner in a familiar environment without stress, or excessive intake of food or alcohol beforehand.163,164 Although sexual activity transiently increases the risk of MI, it is the cause of <1% of acute MIs and <1–1.7% of sudden deaths occur during sexual activity.164 The energy expenditure during sexual activity is generally low-to-moderate (3 − 5 metabolic equivalents) and climbing two flights of stairs is often used as an equivalent activity in terms of energy expended.163,164 Regular physical activity decreases the risk of adverse events during sexual activity.165 Sexual dysfunction in patients with CCS includes decreased libido and sexual activity, and a high prevalence of erectile dysfunction. Sexual dysfunction may be caused by underlying vascular conditions, psychosocial factors, specific medications, number of medications, and changes in relationships.166 Thiazide diuretics and beta-blockers (except nebivolol) may negatively influence erectile function, but studies published since 2011 have not found a consistent relationship between most contemporary cardiovascular medications and erectile dysfunction.162,164,165 Phosphodiesterase-5 inhibitors to treat erectile dysfunction are generally safe in CCS patients, but should not be used in those taking nitrates.164 Healthcare providers should ask patients about sexual activity, and offer advice and counselling.
3.2.2.9 Adherence and sustainability
Adherence to lifestyle modifications and to medications is a challenge. A systematic review of epidemiological studies indicated that a substantial proportion of patients do not adhere to cardiovascular medications, and that 9% of cardiovascular events in Europe were attributable to poor adherence.167 In older men with ischaemic heart disease, greater adherence to medication guidelines appears to be positively associated with better clinical outcomes, independent of other conditions.168 Polypharmacy plays a negative role in adherence to treatment,169 and complexity of drug regimen is associated with non-adherence and higher rates of hospitalizations.170 Drug prescriptions should prioritize medications that have proved their benefit with the highest level of evidence and those for whom the amplitude of benefit is largest. Simplifying medication regimens may help, and there is some evidence of benefits of cognitive educational strategies, electronically monitored feedback, and support by nurse case managers. Medication reviews by primary care providers may be helpful in patients with multiple comorbidities to minimize the risk of adverse interactions and to simplify medication regimens.117,171–173
Promoting behaviour change and medication adherence should be part of each clinical encounter in primary care and specialist follow-up, emphasising its importance, referring for support when needed, and congratulating patients for their achievements. Long-term support (intensive in the first 6 months, then every 6 months for 3 years) in the GOSPEL (Global secondary prevention strategies to limit event recurrence after myocardial infarction) trial resulted in significant improvements in risk factors, and decreases in several clinical mortality and morbidity endpoints.121 The Multicenter Lifestyle Demonstration Project showed that CCS patients could make intensive lifestyle changes and improve their risk factors and fitness, with changes sustained at 12 months.174
3.2.2.10 Influenza vaccination
An annual influenza vaccination can improve prevention of acute MI in patients with CCS,175,176 change HF prognosis,177 and decrease cardiovascular mortality in adults aged ≥65 years.178–180 Therefore, annual influenza vaccination is recommended for patients with CAD, especially in the elderly.
Recommendations on lifestyle management
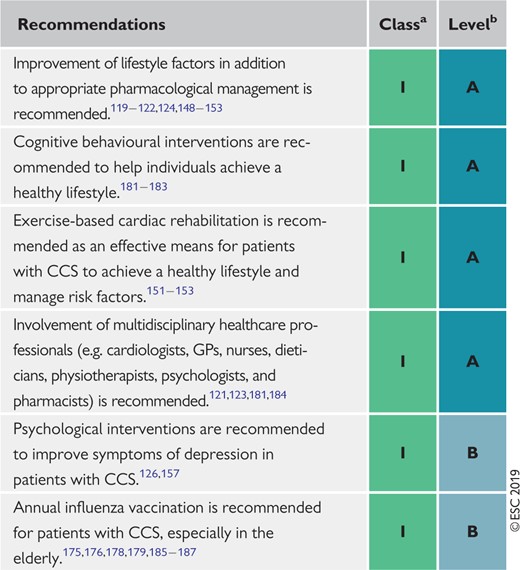 |
 |
CCS = chronic coronary syndrome; GPs = general practitioners.
Class of recommendation.
Level of evidence.
Recommendations on lifestyle management
 |
 |
CCS = chronic coronary syndrome; GPs = general practitioners.
Class of recommendation.
Level of evidence.
3.3 Pharmacological management
The aims of pharmacological management of CCS patients are to reduce angina symptoms and exercise-induced ischaemia, and to prevent cardiovascular events.
Immediate relief of anginal symptoms, or the prevention of symptoms under circumstances likely to elicit angina, is usually obtained with rapidly acting formulations of nitroglycerin. Anti-ischaemic drugs—but also lifestyle changes, regular exercise training, patient education, and revascularization—all play a role in minimizing or eradicating symptoms over the long-term (long-term prevention).
Prevention of cardiovascular events targets MI and death associated with CAD, and focuses primarily on reducing the incidence of acute thrombotic events and the development of ventricular dysfunction. Strategies include pharmacological and lifestyle interventions, as detailed in the 2016 European Guidelines on CVD prevention in clinical practice.15
3.3.1 Anti-ischaemic drugs
3.3.1.1 General strategy
Optimal treatment can be defined as the treatment that satisfactorily controls symptoms and prevents cardiac events associated with CCS, with maximal patient adherence and minimal adverse events.188–191 However, there is no universal definition of an optimal treatment in patients with CCS, and drug therapies must be adapted to each patient’s characteristics and preferences.192 Initial drug therapy usually consists of one or two antianginal drugs, as necessary, plus drugs for secondary prevention of CVD.193 The initial choice of antianginal drug(s) depends on the expected tolerance related to the individual patient’s profile and comorbidities, potential drug interactions with co-administered therapies, the patient’s preferences after being informed of potential adverse effects, and drug availability. Whether combination therapy with two antianginal drugs [e.g. a beta-blocker and a calcium channel blocker (CCB)] is superior to monotherapy with any class of antianginal drug in reducing clinical events remains unclear.194–197
Beta-adrenergic blockers or CCBs are recommended as the first choice, although no RCT to date has compared this strategy to an alternative strategy using initial prescription of other anti-ischaemic drugs, or the combination of a beta-blocker and a CCB.191,195 The results of a network meta-analysis of 46 studies and 71 treatment comparisons supported the initial combination of a beta-blocker and a CCB.198 The same meta-analysis suggested that several second-line add-on anti-ischaemic drugs (long-acting nitrates, ranolazine, trimetazidine, and, to a lesser extent, ivabradine) may prove beneficial in combination with a beta-blocker or a CCB as first-line therapy, while no data were available for nicorandil. However, it should be noted that the study pooled RCTs using endpoints of nitrate use, angina frequency, time to angina or to ST-segment depression, and total exercise time, and no study or meta-analysis has yet assessed with sufficient power the influence of combining a beta-blocker or a CCB with a second-line anti-ischaemic drug on morbidity or mortality events.198 Regardless of the initial strategy, response to initial antianginal therapy should be reassessed after 2–4 weeks of treatment initiation.
3.3.1.2 Available drugs
Anti-ischaemic drugs have proved benefits regarding symptoms associated with myocardial ischaemia but do not prevent cardiovascular events in most patients with CCS. Supplementary Table 3 in the Supplementary Data summarizes the principal major side effects, contraindications, drug–drug interactions, and precautions relating to anti-ischaemic drugs. Supplementary Table 2 summarizes the main mechanisms of action of anti-ischaemic drugs.
Nitrates
Short-acting nitrates for acute effort angina
Sublingual and spray nitroglycerin formulations provide immediate relief of effort angina. Spray nitroglycerin acts more rapidly than sublingual nitroglycerin.199 At the onset of angina symptoms, the patient should rest in a sitting position (standing promotes syncope, and lying down enhances venous return and preload) and take nitroglycerin (0.3–0.6 mg tablet sublingually and not swallowed, or 0.4 mg spray to the tongue and not swallowed or inhaled) every 5 min until the pain disappears, or a maximum of 1.2 mg has been taken within 15 min. During this time frame, if angina persists, immediate medical attention is needed. Nitroglycerin can be administered for prophylaxis before physical activities known to provoke angina. Isosorbide dinitrate (5 mg sublingually) has a slightly slower onset of action than nitroglycerin due to hepatic conversion to mononitrate. The effect of isosorbide dinitrate may last ≤1 h if the drug is taken sublingually or persist for several hours if the drug is taken by oral ingestion.
Long-acting nitrates for angina prophylaxis
Long-acting nitrate formulations (e.g. nitroglycerin, isosorbide dinitrate, and isosorbide mononitrate) should be considered as second-line therapy for angina relief when initial therapy with a beta-blocker or non-dihydropyridine (non-DHP) CCB is contraindicated, poorly tolerated, or insufficient to control symptoms. In fact, there is a paucity of data comparing nitrates with beta-blockers or CCB from which to draw firm conclusions about their relative efficacies.200 When taken over a prolonged period, long-acting nitrates provoke tolerance with loss of efficacy, which requires prescription of a nitrate-free or nitrate-low interval of ∼10–14 h.201 Nitroglycerin can be administered orally or transdermally through slow-release patch systems. Bioavailability of isosorbide dinitrate depends on the interindividual variability in hepatic conversion and is generally lower than the bioavailability of isosorbide mononitrate (its active metabolite), which is 100% bioavailable. Titration of dose is essential with all formulations to obtain the maximal control of symptoms at a tolerable dose. Discontinuation should be tapered and not abrupt to avoid a rebound increase in angina.202 The most common side effects are hypotension, headache, and flushing. Contraindications include hypertrophic obstructive cardiomyopathy, severe aortic valvular stenosis, and co-administration of phosphodiesterase inhibitors (e.g. sildenafil, tadalafil, or vardenafil) or riociguat.
Beta-blockers
The dose of beta-blockers should be adjusted to limit the heart rate to 55–60 b.p.m. (beats per minute) at rest.203,204 Discontinuation should be tapered and not abrupt. Beta-blockers can be combined with DHP CCBs to reduce DHP-induced tachycardia, but with uncertain incremental clinical value.205–208 Caution is warranted when a beta-blocker is combined with verapamil or diltiazem due to the potential for developing worsening of HF, excessive bradycardia, and/or atrioventricular block. Combination of a beta-blocker with a nitrate attenuates the reflex tachycardia of the latter. The principal side effects of beta-blockers are fatigue, depression, bradycardia, heart block, bronchospasm, peripheral vasoconstriction, postural hypotension, impotence, and masking of hypoglycaemia symptoms.
In certain patients with recent MI and those with chronic HF with reduced ejection fraction, beta-blockers have been associated with a significant reduction in mortality and/or cardiovascular events,209–215 but the protective benefit in patients with CAD without prior MI or HF is less well established and lacks placebo-controlled trials.216 A retrospective analysis of 21 860 matched patients from the REACH (REduction of Atherothrombosis for Continued Health) Registry showed no reduction in cardiovascular mortality with beta-blockers in patients with either CAD with risk factors only, known prior MI, or known CAD without MI.217 In a retrospective national registry of 755 215 patients aged ≥65 years with a history of CAD without prior MI or HF with reduced ejection fraction undergoing elective PCI, beta-blocker use at discharge was not associated with any reduction in cardiovascular morbidity or mortality at 30 day and 3 year follow-up.218 However, in patients with or without previous MI undergoing CABG, beta-blockers were associated with lower risk of long-term mortality and adverse cardiovascular events.219 Other observational studies and meta-analyses have questioned the benefit of long-term (>1 year) beta-blocker therapy in patients with a previous MI.216,220–224 This is still a matter for debate,225 and uncertainties remain on the comparative role of beta-blockers and angiotensin-converting enzyme (ACE) inhibitors.
Calcium channel blockers
While CCBs improve symptoms and myocardial ischaemia, they have not been shown to reduce major morbidity endpoints or mortality in patients with CCS.192,226–228
Non-dihydropyridine agents (heart rate-lowering calcium channel blockers)
Verapamil. Verapamil has a large range of approved indications, including all varieties of angina (effort, vasospastic, and unstable), supraventricular tachycardias, and hypertension. Indirect evidence suggests good safety but with risks of heart block, bradycardia, and HF. Compared with metoprolol, the antianginal activity was similar.229 Compared with atenonol in hypertension with CAD, verapamil is associated with fewer cases of diabetes, fewer anginal attacks,230 and less psychological depression.231 Beta-blockade combined with verapamil is not advised (due to risk of heart block).
Diltiazem. Diltiazem, with its low-side effect profile, has advantages compared with verapamil in the treatment of effort angina. Like verapamil, it acts by peripheral vasodilation, relief of exercise-induced coronary constriction, a modest negative inotropic effect, and sinus node inhibition. There have been no outcome studies comparing diltiazem and verapamil.
In some selected patients, non-DHP agents may be combined with beta-blockers for the treatment of angina. However, on such occasions they must be used under close monitoring of patients’ tolerance regarding excessive bradycardia or signs of HF. Use of non-DHP CCBs in patients with LV dysfunction is not advised.
Dihydropyridine agents
Long-acting nifedipine. This agent is a powerful arterial vasodilator with few serious side effects. Long-acting nifedipine has been especially well tested in hypertensive anginal patients when added to beta-blockade.232 In the large placebo-controlled ACTION (A Coronary disease Trial Investigating Outcome with Nifedipine gastrointestinal therapeutic system) trial, addition of long-acting nifedipine [60 mg o.d. (once a day)] to conventional treatment of angina had no effect on major cardiovascular event-free survival. Long-acting nifedipine proved to be safe, and reduced the need for coronary angiography and cardiovascular interventions.232 Relative contraindications to nifedipine are few (severe aortic stenosis, hypertrophic obstructive cardiomyopathy, or HF), and careful combination with beta-blockade is usually feasible and desirable. Vasodilatory side effects include headache and ankle oedema.
Amlodipine. The very long half-life of amlodipine and its good tolerability make it an effective once-a-day antianginal and antihypertensive agent, setting it apart from drugs that are taken either twice or three times daily. Side effects are few, mainly ankle oedema. In patients with CCS and normal BP (∼75% receiving a beta-blocker), amlodipine 10 mg/day reduced coronary revascularizations and hospitalizations for angina in a 24 month trial.233 Exercise-induced ischaemia is more effectively reduced by amlodipine, 5 mg titrated to 10 mg/day, than by the beta-blocker atenolol, 50 mg/day, and their combination is even better.234 However, the CCB–beta-blocker combination is often underused, even in some studies reporting ‘optimally treated’ stable effort angina.
Ivabradine
Ivabradine has been reported to be non-inferior to atenolol or amlodipine in the treatment of angina and ischaemia in patients with CCS.235,236 Adding ivabradine 7.5 mg b.i.d. [bis in die (twice a day)] to atenolol therapy gave better control of heart rate and anginal symptoms.237 In 10 917 patients with limiting previous angina enrolled in the morbidity–mortality evaluation of the BEAUTIFUL (If Inhibitor Ivabradine in Patients With Coronary Artery Disease and Left Ventricular Dysfunction) trial, ivabradine did not reduce the composite primary endpoint of cardiovascular death, hospitalization with MI, or HF.238 Also, in the SIGNIFY (Study Assessing the Morbidity–Mortality Benefits of the If Inhibitor Ivabradine in Patients with Coronary Artery Disease) study, consisting of 19 102 patients with CAD without clinical HF and a heart rate ≥70 b.p.m., there was no significant difference between the ivabradine group and the placebo group in the incidence of the primary composite endpoint of death from cardiovascular causes or non-fatal MI.239 Ivabradine was associated with an increase in the incidence of the primary endpoint among 12 049 patients with activity-limiting angina but not among those without activity-limiting angina (P=0.02 for interaction). In 2014, the European Medicines Agency issued recommendations to reduce the risk of bradycardia and placed ivabradine under additional monitoring.240 In aggregate, these results support the use of ivabradine as a second-line drug in patients with CCS.
Nicorandil
Nicorandil is a nitrate derivative of nicotinamide, with antianginal effects similar to those of nitrates or beta-blockers.241–244 Side effects include nausea, vomiting, and potentially severe oral, intestinal, and mucosal ulcerations.245
In the placebo-controlled IONA (Impact Of Nicorandil in Angina) trial (n = 5126), nicorandil significantly reduced the composite of coronary heart disease (CHD) death, non-fatal MI, or unplanned hospital admission for suspected anginal symptoms in patients with CCS, but there was no effect on death from ischaemic heart disease or non-fatal MI.246 These results support the use of nicorandil as a second-line drug in patients with CCS.
Ranolazine
Ranolazine is a selective inhibitor of the late inward sodium current. Side effects include dizziness, nausea, and constipation. In addition, ranolazine increases QTc, and should therefore be used carefully in patients with QT prolongation or on QT-prolonging drugs.
In a placebo-controlled trial of 6560 patients with non-ST-segment elevation ACS, the addition of ranolazine to standard treatment did not prove effective in reducing the primary efficacy endpoint of cardiovascular death, MI, or recurrent ischaemia.247 However, in the relatively large subgroup of patients with chronic angina (n = 3565), significant reductions in recurrent ischaemia, worsening angina, and intensification of antianginal therapy were observed.248 In another placebo-controlled trial of patients with diabetes and CAD receiving one or two antianginal drugs, ranolazine reduced angina and sublingual nitroglycerin use with good tolerability.249 In the RIVER-PCI (Ranolazine for Incomplete Vessel Revascularization Post‐Percutaneous Coronary Intervention) trial, ranolazine did not reduce the composite of ischaemia-driven revascularization or hospitalization without revascularization in 2651 patients with a history of chronic angina and incomplete revascularization after PCI, including those with and without PCI for a CAD indication, nor did it reduce angina symptoms at 1 year.250,251
These results support the use of ranolazine as a second-line drug in CCS patients with refractory angina despite commonly used antianginal agents such as beta-blockers, CCBs, and/or long-acting nitrates. Conversely, there is a lack of evidence to support the use of ranolazine in patients with CCS following PCI with incomplete revascularization.
Trimetazidine
Trimetazidine appears to have a haemodynamically neutral side effect profile.252 Trimetazidine (35 mg b.i.d.) added to beta-blockade (atenolol) improved effort-induced myocardial ischaemia, as reviewed by the European Medicines Agency in June 2012.253,254 It remains contraindicated in Parkinson’s disease and motion disorders, such as tremor (shaking), muscle rigidity, walking disorders, and restless leg syndrome. A 2014 meta-analysis of 13, mostly Chinese, studies consisting of 1628 patients showed that treatment with trimetazidine on top of other antianginal drugs was associated with a smaller weekly mean number of angina attacks, lower weekly nitroglycerin use, longer time to 1 mm ST-segment depression, higher total work, and longer exercise duration at peak exercise than treatment with the other antianginal drugs for stable angina pectoris.255 These results support the use of trimetazidine as a second-line drug in patients with CCS whose symptoms are not adequately controlled by, or who are intolerant to, other medicines for angina pectoris.
Allopurinol
In 2010, a randomized crossover study of 65 patients with CAD showed that allopurinol 600 mg/day increased times to ST-segment depression and to angina.256 An observational study of 29 298 episodes of incident allopurinol use found an association of allopurinol use with a reduction in the risk of incident MI in the elderly, particularly when used for >2 years.257 However, the role of allopurinol in reducing clinical events in CVD remains unclear.258
A stepwise strategy for anti-ischaemic drug therapy in CCS is proposed, depending on some baseline patient characteristics (Figure 8). Incomplete responses or poor tolerance at each step justify moving to the next step. The strategy must be adapted to each patient’s characteristics and preferences, and does not necessarily follow the steps indicated in the figure.
Suggested stepwise strategy for long term anti-ischaemic drug therapy in patients with chronic coronary syndromes and specific baseline characteristics. The proposed stepwise approach must be adapted to each patient's characteristics and preferences. Given the limited evidence on various combinations of drugs in different clinical conditions, the proposed options are only indicative of potential combinations and do not represent formal recommendations. BB = beta-blocker; bpm = beats per minute; CCB = [any class of] calcium channel blocker; DHP-CCB = dihydropyridine calcium channel blocker; HF = heart failure; LAN = long-acting nitrate; LV = left ventricular; non-DHP-CCB = non-dihydropyridine calcium channel blocker. aCombination of a BB with a DHP-CCB should be considered as first step; combination of a BB or a CCB with a second-line drug may be considered as a first step; bThe combination of a BB and non-DHP-CCB should initially use low doses of each drug under close monitoring of tolerance, particularly heart rate and blood pressure; cLow-dose BB or low-dose non-DHP-CCB should be used under close monitoring of tolerance, particularly heart rate and blood pressure; dIvabradine should not be combined with non-DHP-CCB; eConsider adding the drug chosen at step 2 to the drug tested at step 1 if blood pressure remains unchanged.
3.3.1.3 Patients with low blood pressure
In patients with low BP, it is recommended to start antianginal drugs at very low doses, with preferential use of drugs with no or limited effects on BP. A low-dose beta-blocker or low-dose non-DHP-CCB can be tested first under close monitoring of tolerance. Ivabradine (in patients with sinus rhythm), ranolazine, or trimetazidine can also be used.
3.3.1.4 Patients with low heart rate
Increased heart rate correlates linearly with cardiovascular events, and the benefit of heart-rate reduction as a treatment goal in subgroups of CCS patients has been demonstrated using various drugs.203,259–261 However, in patients with baseline bradycardia (e.g. heart rate <60 b.p.m.) heart rate-lowering drugs (beta-blockers, ivabradine, and heart-rate lowering CCBs) should be avoided or used with caution, and—if needed—started at very low doses. Antianginal drugs without heart rate-lowering effects should preferably be given.
Recommendations on anti-ischaemic drugs in patients with chronic coronary syndromes
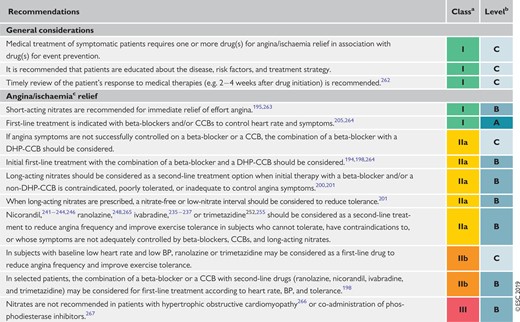 |
 |
BP = blood pressure; CCB = calcium channel blocker; CCS = chronic coronary syndromes; DHP-CCB = dihydropyridine calcium channel blocker.
Class of recommendation.
Level of evidence.
No demonstration of benefit on prognosis.
Recommendations on anti-ischaemic drugs in patients with chronic coronary syndromes
 |
 |
BP = blood pressure; CCB = calcium channel blocker; CCS = chronic coronary syndromes; DHP-CCB = dihydropyridine calcium channel blocker.
Class of recommendation.
Level of evidence.
No demonstration of benefit on prognosis.
3.3.2 Event prevention
3.3.2.1 Antiplatelet drugs
Platelet activation and aggregation is the driver for symptomatic coronary thrombosis, forming the basis for the use of antiplatelet drugs in patients with CCS in view of a favourable balance between the prevention of ischaemic events and increased risk of bleeding. Dual antiplatelet therapy (DAPT) with aspirin and an oral P2Y12 inhibitor is the mainstay of antithrombotic therapy after MI and/or PCI.
Low-dose aspirin
Aspirin acts via irreversible inhibition of platelet cyclooxygenase-1 and thus thromboxane production, which is normally complete with chronic dosing ≥75 mg/day. The gastrointestinal side effects of aspirin increase at higher doses, and current evidence supports a daily dose of 75–100 mg for the prevention of ischaemic events in CAD patients with or without a history of MI.268–270 As cyclooxygenase-1 inhibition by aspirin is consistent and predictable in adherent patients, no platelet function testing is required to monitor individual response.271 Although other non-selective non-steroidal anti-inflammatory drugs, such as ibuprofen, reversibly inhibit cyclooxygenase-1, their adverse effects on cardiovascular risk indicate that they cannot be recommended as an alternative treatment in patients with aspirin intolerance.272
Oral P2Y12 inhibitors
P2Y12 inhibitors block the platelet P2Y12 receptor, which plays a key role in platelet activation and the amplification of arterial thrombus formation. Clopidogrel and prasugrel are thienopyridine prodrugs that irreversibly block P2Y12 via active metabolites. Ticagrelor is a reversibly-binding P2Y12 inhibitor that does not require metabolic activation.
The CAPRIE (Clopidogrel vs. Aspirin in Patients at Risk of Ischaemic Events) trial showed an overall slight benefit of clopidogrel compared with aspirin, with a similar safety profile, in preventing cardiovascular events in patients with previous MI, previous stroke, or PAD.273 Subgroup analysis suggested greater benefit of clopidogrel in patients with PAD. Despite its lesser antiplatelet efficacy, clopidogrel demonstrated equivalent efficacy to ticagrelor in patients with PAD.274 Clopidogrel is limited by variable pharmacodynamic effects related to variable efficiency of conversion to its active metabolite, partly associated with loss-of-function variants in the CYP2C19 gene, leading to a lack of efficacy in some patients.271 Drugs that inhibit CYP2C19, such as omeprazole, may reduce the response to clopidogrel.275
Prasugrel has more rapid, more predictable, and, on average, greater antiplatelet effect compared with clopidogrel, and is not susceptible to drug interactions or the effect of CYP2C19 loss-of-function variants. Prasugrel has greater efficacy than clopidogrel in aspirin-treated patients with ACS undergoing PCI, but not in medically-managed patients with ACS.276,277 Prasugrel has been associated with more non-fatal and fatal bleeding events than clopidogrel in ACS patients undergoing PCI, leading to apparent harm in those with a history of ischaemic stroke, and a lack of apparent benefit in those aged >75 years or with body weight <60 kg.276
Ticagrelor has the most predictable and consistently high level of P2Y12 inhibition during maintenance therapy in adherent patients,219 and also has more rapid onset, as well as more rapid and predictable offset of action compared with clopidogrel.278–280 Ticagrelor as monotherapy appears to have similar efficacy and safety to aspirin monotherapy in patients with previous PCI.281 Ticagrelor, with a 180 mg loading dose followed by 90 mg b.i.d., achieved greater reduction of ischaemic events compared with clopidogrel in aspirin-treated ACS patients, regardless of revascularization strategy, at the expense of more non-fatal bleeding.282,283 Ticagrelor at doses of 90 or 60 mg b.i.d. reduced the 3 year combined incidence of MI, stroke, or cardiovascular death compared with placebo in stable aspirin-treated patients with a history of MI 1–3 years previously.284 Both ticagrelor doses increased non-fatal but not fatal bleeding. The equivalent efficacies and similar safeties of the two ticagrelor doses were explained by similar levels of platelet inhibition.285 Ticagrelor may cause dyspnoea, which is often transient and most often mild and tolerable, but occasionally necessitates switching to a thienopyridine.286,287 Ticagrelor is metabolized via CYP3A, and consequently should not be used with strong CYP3A inhibitors or inducers.
The optimal timing of initiation of P2Y12 inhibition before coronary angiography and possible PCI in patients with CCS is uncertain, but increasing use of a radial artery approach and clinical experience has allowed consideration of clopidogrel pre-treatment in patients who have a high chance of requiring PCI.284 Limited pharmacodynamic studies support the unlicensed use of prasugrel or ticagrelor in stable patients undergoing elective PCI who have a high risk of stent thrombosis, but the safety/efficacy balance of this approach, compared with clopidogrel, has not been established.288
Duration of dual antiplatelet therapy
After PCI for stable angina, 6 months of DAPT achieves the optimum balance of efficacy and safety in most patients.284 Premature discontinuation of a P2Y12 inhibitor is associated with an increased risk of stent thrombosis and is discouraged.284 However, a shorter duration of DAPT may be considered in those at high risk of life-threatening bleeding in view of the very low risk of stent thrombosis after 1–3 months.284 On the basis of phase III trials, 12 months is the recommended default duration of DAPT after ACS, but shorter duration may again be considered in those at high bleeding risk.11,284 A DAPT study of patients undergoing PCI showed that extended therapy beyond 12 months with clopidogrel or prasugrel reduced ischaemic events and stent thrombosis, but without mortality benefit and at the expense of increased bleeding.289 A greater benefit of extended clopidogrel or prasugrel was seen in patients who were treated for MI.290
The PEGASUS-TIMI 54 (Prevention of Cardiovascular Events in Patients with Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin–Thrombolysis In Myocardial Infarction 54) trial demonstrated that long-term therapy with ticagrelor 60 or 90 mg b.i.d., commenced in stable patients ≥1 year after MI, reduced ischaemic events at the expense of more non-fatal bleeding.284 The 60 mg dose appeared better tolerated and is approved in many countries for this indication. Subgroup analysis demonstrated greater absolute reductions in ischaemic events with long-term ticagrelor (60 mg b.i.d.) in higher-risk post-MI patients with diabetes, PAD, or multivessel CAD.291–293
3.3.2.2 Anticoagulant drugs in sinus rhythm
Anticoagulant drugs inhibit the action and/or formation of thrombin, which plays a pivotal role in both coagulation and platelet activation. Consequently, anticoagulants have been shown to reduce the risk of arterial thrombotic events. The superior efficacy and safety of DAPT, compared with aspirin and anticoagulation, in preventing stent thrombosis led to the latter strategy being abandoned in favour of DAPT following PCI.284 Combination of antiplatelet therapy and standard anticoagulant doses of warfarin or apixaban for secondary prevention after ACS was associated with an unfavourable balance of efficacy and bleeding.294,295 However, recently reported studies have renewed interest in combining lower anticoagulant doses with antiplatelet therapy.
Low-dose rivaroxaban
Rivaroxaban is a factor Xa inhibitor that has been studied at a low dose of 2.5 mg b.i.d. in several populations of patients in sinus rhythm, this dose being one-quarter of the standard dose used for anticoagulation in patients with AF. Rivaroxaban 2.5 mg b.i.d., compared with placebo, reduced the composite of MI, stroke, or cardiovascular death in stabilized patients treated predominantly with aspirin and clopidogrel following ACS, at the expense of increased bleeding but with evidence of a reduction in cardiovascular death.296 Subsequently, in the COMPASS (Cardiovascular Outcomes for People Using Anticoagulation Strategies) trial, the same regimen in combination with aspirin was compared with aspirin alone, as well as rivaroxaban 5 mg b.i.d. alone, in patients with CCS or PAD, and showed reduced ischaemic events at the expense of increased risk of predominantly non-fatal bleeding.297 Of note, the pre-specified significance thresholds for cardiovascular mortality and all-cause mortality were not met. Greater absolute risk reductions were seen in higher-risk patients with diabetes, PAD, or moderate chronic kidney disease (CKD), as well as current smokers. In GEMINI-ACS (A Study to Compare the Safety of Rivaroxaban Versus Acetylsalicylic Acid in Addition to Either Clopidogrel or Ticagrelor Therapy in Participants With Acute Coronary Syndrome), rivaroxaban 2.5 mg b.i.d. was compared with aspirin in patients treated with a P2Y12 inhibitor who were stable following PCI. The results suggested similar safety of rivaroxaban to aspirin in this setting, but larger studies are required to substantiate this finding.298 In addition, the safety of performing PCI without aspirin pre-treatment is unknown.
3.3.2.3 Anticoagulant drugs in atrial fibrillation
Anticoagulant therapy is recommended in patients with AF and CCS for reduction of ischaemic stroke and other ischaemic events. Anticoagulants in AF patients have demonstrated superiority over aspirin monotherapy or clopidogrel-based DAPT for stroke prevention, and are therefore recommended for this indication.299 When oral anticoagulation is initiated in a patient with AF who is eligible for a non-vitamin K antagonist oral anticoagulant (NOAC; apixaban, dabigatran, edoxaban, or rivaroxaban), a NOAC is recommended in preference to a vitamin K antagonist (VKA).299
Combination anticoagulant and antiplatelet therapy following percutaneous coronary intervention for patients with atrial fibrillation or another indication for oral anticoagulation
No studies to date have specifically focused on CCS patients with AF undergoing PCI and clinical decisions must be based on clinical trials that have included a large proportion of patients with ACS. For peri-procedural management, it is recommended that interruption of VKA is avoided, if feasible, whereas it is recommended that NOAC therapy is stopped for 12–48 h before elective PCI, depending on renal function and the particular NOAC regimen.300 Radial artery access is preferred along with intraprocedural unfractionated heparin either at a standard dose (70–100 U/kg) or, in those with uninterrupted VKA, at a lower dose of 30–50 U/kg.300 Pre-treatment with aspirin 75–100 mg daily is recommended, and clopidogrel (300–600 mg loading dose if not on long-term maintenance therapy) is recommended in preference to prasugrel or ticagrelor.300 VKA-treated patients receiving aspirin and clopidogrel post-PCI should have a target international normalized ratio in the range of 2.0–2.5, aiming for high time in therapeutic range (>70%).300 Subsequent to post-PCI trials of different antithrombotic regimens considered in previous Guidelines,88,284 the AUGUSTUS trial (An Open-label, 2 × 2 Factorial, Randomized Controlled, Clinical Trial to Evaluate the Safety of Apixaban vs. Vitamin K Antagonist and Aspirin vs. Aspirin Placebo in Patients With Atrial Fibrillation and Acute Coronary Syndrome or Percutaneous Coronary Intervention) showed, firstly, that apixaban 5 mg b.i.d. (i.e. the licensed dose for thromboprophylaxis in AF) was associated with significantly less major or clinically relevant non-major bleeding than VKA; and, secondly, that aspirin, compared with placebo, was associated with significantly more bleeding, with the safest combination being apixaban and placebo in addition to P2Y12 inhibitor (predominantly clopidogrel).301 However, there were numerically, but not statistically significantly, more stent thrombosis events with placebo than with aspirin, and the trial was not powered to assess differences in these events between groups.301 Consequently, when concerns about thrombotic risk prevail over concerns about bleeding risk, ≥1 month of triple therapy [oral anticoagulant (OAC), aspirin, and clopidogrel] is recommended to cover the period when the risk of stent thrombosis is presumed to exceed the risk of bleeding.300,301 There is currently limited evidence to support the use of OACs with ticagrelor or prasugrel as dual therapy after PCI as an alternative to triple therapy.300,301
Long-term combination therapy in patients with atrial fibrillation or another indication for anticoagulation
OAC monotherapy is generally recommended 6–12 months after PCI in patients with AF, as there is a lack of specific data supporting long-term treatment with an OAC and a single antiplatelet agent; however, in highly selected cases with high ischaemic risk, dual therapy with an OAC and aspirin or clopidogrel may be considered.300
3.3.2.4 Proton pump inhibitors
Proton pump inhibitors reduce the risk of gastrointestinal bleeding in patients treated with antiplatelet drugs and may be a useful adjunctive treatment for improving safety.275 Long-term proton pump inhibitor use is associated with hypomagnesaemia, but the role of monitoring serum magnesium levels is uncertain. Proton pump inhibitors that inhibit CYP2C19, particularly omeprazole and esomeprazole, may reduce the pharmacodynamic response to clopidogrel. Although this has not been shown to affect the risk of ischaemic events or stent thrombosis, co-administration of omeprazole or esomeprazole with clopidogrel is generally not recommended.
3.3.2.5 Cardiac surgery and antithrombotic therapy
Aspirin should normally be continued in patients with CCS undergoing elective cardiac surgery, and other antithrombotic drugs stopped at intervals according to their duration of action and indication (prasugrel stopped ≥7 days before; clopidogrel ≥5 days before; ticagrelor ≥3 days before; and rivaroxaban, apixaban, edoxaban, and dabigatran 1–2 days before depending on dose and renal function). Reloading of aspirin after CABG surgery may improve graft patency.302 The role of DAPT or dual therapy with aspirin and rivaroxaban after CABG surgery is uncertain as large prospective studies are lacking. However, RCT results have suggested higher graft patency rates with DAPT compared with aspirin monotherapy.284,303,304
3.3.2.6 Non-cardiac surgery and antithrombotic therapy
Non-cardiac surgery is associated with an increased risk of MI. Following PCI, whenever possible, it is recommended to postpone elective surgery until the recommended course of DAPT has been completed. Usually, this will mean delaying surgery until 6 months after PCI, but surgery between 3–6 months may be considered by a multidisciplinary team, including an interventional cardiologist, if clinically indicated. In most types of surgery, aspirin should be continued as the benefit outweighs the bleeding risk, but this may not be appropriate for procedures associated with extremely high bleeding risk (intracranial procedures, transurethral prostatectomy, intraocular procedures, etc.).284 The COMPASS study included CCS patients with a history of peripheral revascularization procedures, and demonstrated benefits of aspirin and rivaroxaban 2.5 mg b.i.d. compared with aspirin alone, including reductions in major adverse limb events and mortality, suggesting the need to risk-stratify patients after non-cardiac vascular surgery for atherosclerotic disease.305,306
Recommendations for event prevention I
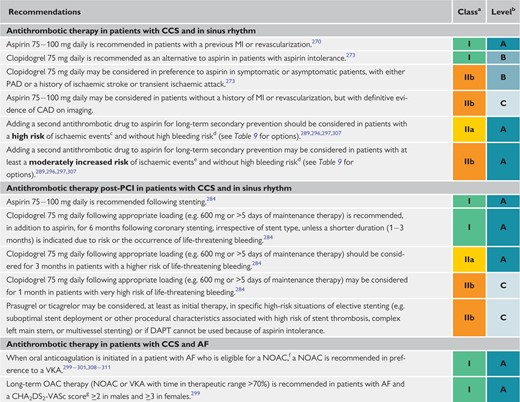 |
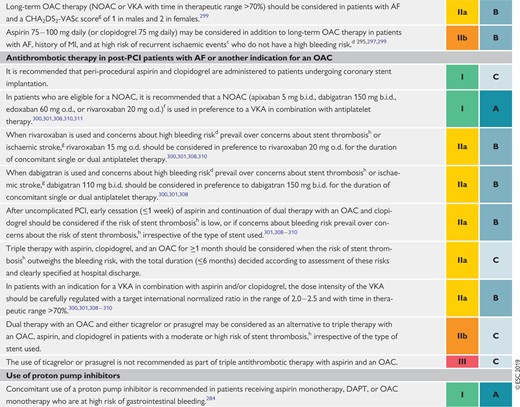 |
 |
 |
AF = atrial fibrillation; b.i.d. = bis in die (twice a day); CAD = coronary artery disease; CCS = chronic coronary syndromes; CHA2DS2-VASc = Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]; CKD = chronic kidney disease; DAPT = dual antiplatelet therapy; eGFR = estimated glomerular filtration rate; HF = heart failure; MI = myocardial infarction; NOAC = non-vitamin K antagonist oral anticoagulant; OAC = oral anticoagulant; o.d. = omni die (once a day); PAD = peripheral artery disease; PCI = percutaneous coronary intervention; VKA = vitamin K antagonist.
Class of recommendation.
Level of evidence.
Diffuse multivessel CAD with at least one of the following: diabetes mellitus requiring medication, recurrent MI, PAD, or CKD with eGFR 15–59 mL/min/1.73 m2.
Prior history of intracerebral haemorrhage or ischaemic stroke, history of other intracranial pathology, recent gastrointestinal bleeding or anaemia due to possible gastrointestinal blood loss, other gastrointestinal pathology associated with increased bleeding risk, liver failure, bleeding diathesis or coagulopathy, extreme old age or frailty, or renal failure requiring dialysis or with eGFR <15 mL/min/1.73 m2.
At least one of the following: multivessel/diffuse CAD, diabetes mellitus requiring medication, recurrent MI, PAD, HF, or CKD with eGFR 15–59 mL/min/1.73 m2.
See summary of product characteristics for reduced doses or contraindications for each NOAC in patients with CKD, body weight <60 kg, age >75–80 years, and/or drug interactions.
Congestive HF, hypertension, age ≥75 years (2 points), diabetes, prior stroke/transient ischaemic attack/embolus (2 points), vascular disease (CAD on imaging or angiography,312 prior MI, PAD, or aortic plaque), age 65–74 years, and female sex.
Risk of stent thrombosis encompasses (i) the risk of thrombosis occurring and (ii) the risk of death should stent thrombosis occur, both of which relate to anatomical, procedural, and clinical characteristics. Risk factors for CCS patients include stenting of left main stem, proximal LAD, or last remaining patent artery; suboptimal stent deployment; stent length >60 mm; diabetes mellitus; CKD; bifurcation with two stents implanted; treatment of chronic total occlusion; and previous stent thrombosis on adequate antithrombotic therapy.
Recommendations for event prevention I
 |
 |
 |
 |
AF = atrial fibrillation; b.i.d. = bis in die (twice a day); CAD = coronary artery disease; CCS = chronic coronary syndromes; CHA2DS2-VASc = Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]; CKD = chronic kidney disease; DAPT = dual antiplatelet therapy; eGFR = estimated glomerular filtration rate; HF = heart failure; MI = myocardial infarction; NOAC = non-vitamin K antagonist oral anticoagulant; OAC = oral anticoagulant; o.d. = omni die (once a day); PAD = peripheral artery disease; PCI = percutaneous coronary intervention; VKA = vitamin K antagonist.
Class of recommendation.
Level of evidence.
Diffuse multivessel CAD with at least one of the following: diabetes mellitus requiring medication, recurrent MI, PAD, or CKD with eGFR 15–59 mL/min/1.73 m2.
Prior history of intracerebral haemorrhage or ischaemic stroke, history of other intracranial pathology, recent gastrointestinal bleeding or anaemia due to possible gastrointestinal blood loss, other gastrointestinal pathology associated with increased bleeding risk, liver failure, bleeding diathesis or coagulopathy, extreme old age or frailty, or renal failure requiring dialysis or with eGFR <15 mL/min/1.73 m2.
At least one of the following: multivessel/diffuse CAD, diabetes mellitus requiring medication, recurrent MI, PAD, HF, or CKD with eGFR 15–59 mL/min/1.73 m2.
See summary of product characteristics for reduced doses or contraindications for each NOAC in patients with CKD, body weight <60 kg, age >75–80 years, and/or drug interactions.
Congestive HF, hypertension, age ≥75 years (2 points), diabetes, prior stroke/transient ischaemic attack/embolus (2 points), vascular disease (CAD on imaging or angiography,312 prior MI, PAD, or aortic plaque), age 65–74 years, and female sex.
Risk of stent thrombosis encompasses (i) the risk of thrombosis occurring and (ii) the risk of death should stent thrombosis occur, both of which relate to anatomical, procedural, and clinical characteristics. Risk factors for CCS patients include stenting of left main stem, proximal LAD, or last remaining patent artery; suboptimal stent deployment; stent length >60 mm; diabetes mellitus; CKD; bifurcation with two stents implanted; treatment of chronic total occlusion; and previous stent thrombosis on adequate antithrombotic therapy.
| Drug option . | Dose . | Indication . | Additional cautions . | References . |
|---|---|---|---|---|
| Clopidogrel | 75 mg o.d. | Post-MI in patients who have tolerated DAPT for 1 year | 289,290 | |
| Prasugrel | 10 mg o.d or 5 mg o.d.; if body weight <60 kg or age >75 years | Post-PCI for MI in patients who have tolerated DAPT for 1 year | Age >75 years | 289,290,313 |
| Rivaroxaban | 2.5 mg b.i.d. | Post-MI >1 year or multivessel CAD | Creatinine clearance 15 − 29 mL/min | 297 |
| Ticagrelor | 60 mg b.i.d. | Post-MI in patients who have tolerated DAPT for 1 year | 291–293,307,314 |
| Drug option . | Dose . | Indication . | Additional cautions . | References . |
|---|---|---|---|---|
| Clopidogrel | 75 mg o.d. | Post-MI in patients who have tolerated DAPT for 1 year | 289,290 | |
| Prasugrel | 10 mg o.d or 5 mg o.d.; if body weight <60 kg or age >75 years | Post-PCI for MI in patients who have tolerated DAPT for 1 year | Age >75 years | 289,290,313 |
| Rivaroxaban | 2.5 mg b.i.d. | Post-MI >1 year or multivessel CAD | Creatinine clearance 15 − 29 mL/min | 297 |
| Ticagrelor | 60 mg b.i.d. | Post-MI in patients who have tolerated DAPT for 1 year | 291–293,307,314 |
Treatment options are presented in alphabetical order.
b.i.d. = bis in die (twice a day); CAD = coronary artery disease; CKD = chronic kidney disease; DAPT = dual antiplatelet therapy; eGFR = estimated glomerular filtration rate; HF = heart failure; MI = myocardial infarction; o.d. = omni die (once a day); PAD = peripheral artery disease; PCI = percutaneous coronary intervention.
High risk of ischaemic events is defined as diffuse multivessel CAD with at least one of the following: diabetes mellitus requiring medication, recurrent MI, PAD, or CKD with eGFR 15 − 59 mL/min/1.73 m2.
Moderately increased risk of ischaemic events is defined as at least one of the following: multivessel/diffuse CAD, diabetes mellitus requiring medication, recurrent MI, PAD, HF, or CKD with eGFR 15 − 59 mL/min/1.73 m2.
High bleeding risk is defined as history of intracerebral haemorrhage or ischaemic stroke, history of other intracranial pathology, recent gastrointestinal bleeding or anaemia due to possible gastrointestinal blood loss, other gastrointestinal pathology associated with increased bleeding risk, liver failure, bleeding diathesis or coagulopathy, extreme old age or frailty, or renal failure requiring dialysis or with eGFR <15 mL/min/1.73 m2.
| Drug option . | Dose . | Indication . | Additional cautions . | References . |
|---|---|---|---|---|
| Clopidogrel | 75 mg o.d. | Post-MI in patients who have tolerated DAPT for 1 year | 289,290 | |
| Prasugrel | 10 mg o.d or 5 mg o.d.; if body weight <60 kg or age >75 years | Post-PCI for MI in patients who have tolerated DAPT for 1 year | Age >75 years | 289,290,313 |
| Rivaroxaban | 2.5 mg b.i.d. | Post-MI >1 year or multivessel CAD | Creatinine clearance 15 − 29 mL/min | 297 |
| Ticagrelor | 60 mg b.i.d. | Post-MI in patients who have tolerated DAPT for 1 year | 291–293,307,314 |
| Drug option . | Dose . | Indication . | Additional cautions . | References . |
|---|---|---|---|---|
| Clopidogrel | 75 mg o.d. | Post-MI in patients who have tolerated DAPT for 1 year | 289,290 | |
| Prasugrel | 10 mg o.d or 5 mg o.d.; if body weight <60 kg or age >75 years | Post-PCI for MI in patients who have tolerated DAPT for 1 year | Age >75 years | 289,290,313 |
| Rivaroxaban | 2.5 mg b.i.d. | Post-MI >1 year or multivessel CAD | Creatinine clearance 15 − 29 mL/min | 297 |
| Ticagrelor | 60 mg b.i.d. | Post-MI in patients who have tolerated DAPT for 1 year | 291–293,307,314 |
Treatment options are presented in alphabetical order.
b.i.d. = bis in die (twice a day); CAD = coronary artery disease; CKD = chronic kidney disease; DAPT = dual antiplatelet therapy; eGFR = estimated glomerular filtration rate; HF = heart failure; MI = myocardial infarction; o.d. = omni die (once a day); PAD = peripheral artery disease; PCI = percutaneous coronary intervention.
High risk of ischaemic events is defined as diffuse multivessel CAD with at least one of the following: diabetes mellitus requiring medication, recurrent MI, PAD, or CKD with eGFR 15 − 59 mL/min/1.73 m2.
Moderately increased risk of ischaemic events is defined as at least one of the following: multivessel/diffuse CAD, diabetes mellitus requiring medication, recurrent MI, PAD, HF, or CKD with eGFR 15 − 59 mL/min/1.73 m2.
High bleeding risk is defined as history of intracerebral haemorrhage or ischaemic stroke, history of other intracranial pathology, recent gastrointestinal bleeding or anaemia due to possible gastrointestinal blood loss, other gastrointestinal pathology associated with increased bleeding risk, liver failure, bleeding diathesis or coagulopathy, extreme old age or frailty, or renal failure requiring dialysis or with eGFR <15 mL/min/1.73 m2.
3.3.3 Statins and other lipid-lowering drugs
Dyslipidaemia should be managed according to lipid guidelines with pharmacological and lifestyle intervention.315 Patients with established CAD are regarded as being at very high risk for cardiovascular events and statin treatment must be considered, irrespective of LDL-C levels. The goal of treatment is to lower LDL-C by at least 50% from baseline and to <1.4 mmol/L (<55 mg/dL) although a lower target LDL-C of <1.0 mmol/L (<40 mg/dL) may be considered in patients who have experience a second vascular event within 2 years, not necessarily of the same type as the first event, whilst taking maximally tolerated statin-based therapy. When this level cannot be achieved, the addition of ezetimibe has been demonstrated to decrease cholesterol and cardiovascular events in post-ACS patients, and in those with diabetes,316 with no further effect on mortality.317 In addition to exercise, diet, and weight control, which should be recommended to all patients, dietary supplements including phytosterols may lower LDL-C to a lesser extent, but have not been shown to improve clinical outcomes.318 These are also used in patients with intolerance to statins who constitute a group at higher risk for cardiovascular events.319 Trials published since 2015 have demonstrated that proprotein convertase subtilisin-kexin type 9 (PCSK9) inhibitors (evolocumab320 and alirocumab321–323) are very effective at reducing cholesterol, lowering LDL-C in a stable fashion to ≤1.3 mmol/L (50 mg/dL). In outcomes trials, these agents have demonstrated a reduction of cardiovascular and mainly ischaemic events, with little or no impact on mortality.324 Very low levels of cholesterol are well tolerated and associated with fewer events,325 but the high cost of PCSK9 inhibitors, unaffordable for many health systems,326 and their unknown long-term safety have limited their use to date. Low-density lipoprotein apheresis and new therapies such as mipomersen and lomitapide need further research.
For patients undergoing PCI, high-dose atorvastatin has been shown to reduce the frequency of peri-procedural events in both statin-naïve patients and patients receiving chronic statin therapy.327
3.3.4 Renin−angiotensin−aldosterone system blockers
ACE inhibitors can reduce mortality, MI, stroke, and HF among patients with LV dysfunction,328–330 previous vascular disease,331–333 and high-risk diabetes.334 It is recommended that ACE inhibitors [or angiotensin receptor blockers (ARBs) in cases of intolerance] be considered for the treatment of patients with CCS with coexisting hypertension, LVEF ≤40%, diabetes, or CKD, unless contraindicated (e.g. severe renal impairment, hyperkalaemia, etc.). However, not all trials have demonstrated that ACE inhibitors reduce all-cause death, cardiovascular death, non-fatal MI, stroke, or HF in patients with atherosclerosis and without impaired LV function.331,332,335 A meta-analysis, including 24 trials and 61 961 patients, documented that, in CCS patients without HF, renin−angiotensin system (RAS) inhibitors reduced cardiovascular events and death only when compared with placebo, but not when compared with active controls.336 Hence, ACE inhibitor therapy in CCS patients without HF or high cardiovascular risk is not generally recommended, unless required to meet BP targets.
Neprilysin is an endogenous enzyme that degrades vasoactive peptides such as bradykinin and natriuretic peptides. Pharmacological inhibition of neprilysin raises the levels of these peptides, enhancing diuresis, natriuresis, myocardial relaxation, and antiremodelling, and reducing renin and aldosterone secretion. The first in class is LCZ696, which combines valsartan and sacubitril (neprilysin inhibitor) in a single pill. In patients with HF (LVEF ≤35%) who remain symptomatic despite optimal treatment with an ACE inhibitor, a beta-blocker, and a mineralocorticoid receptor antagonist (MRA), sacubitril/valsartan is recommended as a replacement for an ACE inhibitor to further reduce the risk of HF hospitalization and death in ambulatory patients.337
Aldosterone blockade with spironolactone or eplerenone is recommended for use in post-MI patients who are already receiving therapeutic doses of an ACE inhibitor and a beta-blocker, have an LVEF ≤35%, and have either diabetes or HF.338,339 Caution should be exercised when MRAs are used in patients with impaired renal function [estimated GFR (eGFR) <45 mL/min/1.73 m2] and in those with serum potassium levels ≥5.0 mmol/L.340
Recommendations for event prevention II
 |
 |
ACE = angiotensin-converting enzyme; ARB = angiotensin-receptor blocker; CCS = chronic coronary syndrome; HF = heart failure; LV = left ventricular; PCSK9 = proprotein convertase subtilisin-kexin type 9; STEMI = ST-elevation myocardial infarction.
Class of recommendation.
Level of evidence.
The treatment goals are shown in the European Society of Cardiology/European Atherosclerosis Society Guidelines for the management of dyslipidaemias.315
Recommendations for event prevention II
 |
 |
ACE = angiotensin-converting enzyme; ARB = angiotensin-receptor blocker; CCS = chronic coronary syndrome; HF = heart failure; LV = left ventricular; PCSK9 = proprotein convertase subtilisin-kexin type 9; STEMI = ST-elevation myocardial infarction.
Class of recommendation.
Level of evidence.
The treatment goals are shown in the European Society of Cardiology/European Atherosclerosis Society Guidelines for the management of dyslipidaemias.315
3.3.5 Hormone replacement therapy
The results from large randomized trials have shown that hormone replacement therapy provides no prognostic benefit and increases the risk of CVD in women aged >60 years.344
3.4 Revascularization
In patients with CCS, optimal medical therapy is key for reducing symptoms, halting the progression of atherosclerosis, and preventing atherothrombotic events. Myocardial revascularization plays a central role in the management of CCS on top of medical treatment, but always as an adjunct to medical therapy without supplanting it. The two objectives of revascularization are symptom relief in patients with angina and/or improvement of prognosis.
Previous Guidelines support indications for revascularization mainly in patients with CCS who receive Guideline-recommended optimal medical therapy and continue to be symptomatic, and/or in whom revascularization may ameliorate prognosis.88 These recommendations suggested that revascularization in patients with angina and significant stenosis was often a second-line therapy after medical therapy had been unsuccessful. However, angina is associated with impaired quality of life, reduced physical endurance, mental depression, and recurrent hospitalizations and office visits, with impaired clinical outcomes.345,346
Revascularization by PCI or CABG may effectively relieve angina, reduce the use of antianginal drugs, and improve exercise capacity and quality of life compared with a strategy of medical therapy alone. In the 5 year follow-up of the FAME 2 (Fractional Flow Reserve versus Angiography for Multivessel Evaluation 2) trial, revascularization improved quality of life, and reduced the use of antianginal drugs and associated side effects.347 The ORBITA (Objective Randomised Blinded Investigation with optimal medical Therapy or Angioplasty in stable angina) study, entailing a sham procedure in the control group, found no significant improvement in exercise capacity with PCI.262 The study highlights a significant placebo component to the clinical effects, and alerts us to the pitfalls of interpreting endpoints subject to bias in the absence of sham control and blinding. However, the ORBITA results cannot inform Guidelines due to the limited trial size, short-term observation time until crossover, and insufficient power to assess clinical endpoints.
Revascularization by either PCI or CABG also aims to effectively eliminate myocardial ischaemia and its adverse clinical manifestations among patients with significant coronary stenosis, and to reduce the risk of major acute cardiovascular events including MI and cardiovascular death. Numerous meta-analyses comparing a strategy of PCI with initial medical therapy among patients with CCS have found no348,349 or a modest104,350,351 benefit, in terms of survival or MI for an invasive strategy. In this regard, previous Guidelines identified specific subgroups of patients (based on the anatomy of the coronary tree, LV function, risk factors, etc.) in whom revascularization may improve prognosis, indicating that in other groups it may not.88
A meta-analysis by Windecker et al. reported an incremental reduction of death and MI by revascularization vs. medical therapy only in CCS patients when revascularization was performed with CABG or new-generation drug-eluting stents (DES), as opposed to balloon angioplasty, bare-metal stents, or early DES.351 Data reported in 2018 indicate a potentially broader prognostic impact of revascularization strategies. The 5 year follow-up of the FAME 2 trial confirmed a sustained clinical benefit in patients treated with PCI specifically targeting the ischaemia-producing stenoses (i.e. FFR <0.80) plus optimal medical therapy vs. optimal medical therapy alone in terms of a significantly lower rate of urgent revascularization (hazard ratio 0.27, 95% CI 0.18–0.41), and a lower rate of spontaneous MI (hazard ratio 0.62, 95% CI 0.39–0.99).347 In contrast to some of the earlier meta-analyses, this signal was confirmed in a patient-level meta-analysis including 2400 subjects, all of whom underwent invasive physiological guidance, showing a significant reduction in cardiac death and MI after a median follow-up of 33 months with FFR-guided PCI vs. medical therapy (hazard ratio 0.74, 95% CI 0.56–0.989; P=0.041).352 Together, these new data support a less restrictive indication for revascularization in CCS, in addition to specific anatomy [e.g. left main (LM)] or extended ischaemia (>10%), when PCI is restricted to angiographic stenoses on large vessels causing a significant intracoronary pressure gradient. Figure 9 summarizes a practical approach to the indications of revascularization in CCS according to the presence or absence of symptoms, and documentation of non-invasive ischaemia. However, the individual risk−benefit ratio should always be evaluated and revascularization considered only if its expected benefit outweighs its potential risk. Also, the aspect of shared decision-making is key, with full information given to the patient about the anticipated advantages and disadvantages of the two strategies, including the DAPT-related bleeding risks in cases of revascularization by PCI. For the discussion of the best choice between revascularization modalities–PCI or CABG–for individual patients, we refer readers to the 2018 ESC myocardial revascularization Guidelines.88
Decision tree for patients undergoing invasive coronary angiography. Decisions for revascularization by percutaneous coronary intervention or coronary artery bypass grafting are based on clinical presentation (symptoms present or absent), and prior documentation of ischaemia (present or absent). In the absence of prior documentation of ischaemia, indications for revascularization depend on invasive evaluation of stenosis severity or prognostic indications. Patients with no symptoms and ischaemia include candidates for transcatheter aortic valve implantation, valve, and other surgery. CAD = coronary artery disease; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio; LV = left ventricle; LVEF = left ventricular ejection fraction; MVD = multivessel disease.
4 Patients with new onset of heart failure or reduced left ventricular function
CAD is the most common cause of HF in Europe, and most of the trial evidence supporting management recommendations is based on research conducted in patients with ischaemic cardiomyopathy. The pathophysiology results in systolic dysfunction due to myocardial injury and ischaemia, and most patients with symptomatic HF have reduced ejection fraction (<40%), although patients with CCS may also have symptomatic HF and a preserved ejection fraction (≥50%). Patients with symptomatic HF should be managed clinically according to the 2016 ESC heart failure Guidelines.340
History should include the assessment of symptoms suggestive of HF, especially exercise intolerance and dyspnoea on exertion. All major past events related to CAD including MI and revascularization procedures are recorded, as well as all major cardiovascular comorbidity requiring treatment such as AF, hypertension, or valvular dysfunction, and non-cardiovascular comorbidity such as CKD, diabetes, anaemia, or cancer. Current medical therapy, adherence, and tolerance should be reviewed.
Physical examination should assess the nutritional status of patients, and estimate biological age and cognitive ability. Recorded physical signs include heart rate, heart rhythm, supine BP, murmurs suggestive of aortic stenosis or mitral insufficiency, signs of pulmonary congestion with basal rales or pleural effusion, signs of systemic congestion with dependant oedema, hepatomegaly, and elevated jugular venous pressure.
A routine ECG provides information on heart rate and rhythm, extrasystole, signs of ischaemia, pathological Q waves, hypertrophy, conduction abnormalities, and bundle branch block.
Imaging should include an echocardiographic examination with Doppler to evaluate evidence of ischaemic cardiomyopathy with HF with reduced ejection fraction, HF with mid-range ejection fraction, or HF with preserved ejection fraction, focal/diffuse LV or right ventricular systolic dysfunction, evidence of diastolic dysfunction, hypertrophy, chamber volumes, valvular function, and evidence of pulmonary hypertension. Chest X-ray can detect signs of pulmonary congestion, interstitial oedema, infiltration, or pleural effusion. If not known, coronary angiography (or coronary CTA) should be performed to establish the presence and extent of CAD, and evaluate the potential for revascularization.52,53
Laboratory investigations should measure natriuretic peptide levels to rule-out the diagnosis of suspected HF. When present, the severity of HF can be assessed.25,49 Renal function along with serum electrolytes should be measured routinely to detect the development of renal insufficiency, hyponatraemia, or hyperkalaemia, especially at the initiation and during up-titration of pharmacological therapy.
The management of patients with symptomatic HF requires adequate diuretic therapy, preferably with a loop diuretic, to relieve signs and symptoms of pulmonary and systemic congestion. Inhibitors of both the RAS system (ACE inhibitors, ARBs, angiotensin receptor−neprilysin inhibitor) and the adrenergic nervous system (beta-blockers) are indicated in all symptomatic patients with HF.340 In patients with persistent symptoms, an MRA is also indicated. Up-titration of these drugs should be gradual to avoid symptomatic systolic hypotension, renal insufficiency, or hyperkalaemia.
Patients who remain symptomatic, with LV systolic dysfunction and evidence of ventricular dysrhythmia or bundle branch block, may be eligible for a device [cardiac resynchronization therapy (CRT)/implantable cardioverter-defibrillator]. Such devices may provide symptomatic relief, reduce morbidity, and improve survival.353–356 Patients with HF may decompensate rapidly following the onset of atrial or ventricular dysrhythmia, and should be treated according to current Guidelines. Patients with HF, and haemodynamically significant aortic stenosis or mitral insufficiency, may require percutaneous or surgical intervention.
Myocardial revascularization should be considered in eligible patients with HF based on their symptoms, coronary anatomy, and risk profile. Successful revascularization in patients with HF due to ischaemic cardiomyopathy may improve LV dysfunction and prognosis by reducing ischaemia to viable, hibernating myocardium. If available, cooperation with a multidisciplinary HF team is strongly advised.348,357,358
General recommendations for the management of patients with chronic coronary syndromes and symptomatic heart failure due to ischaemic cardiomyopathy and left ventricular systolic dysfunction
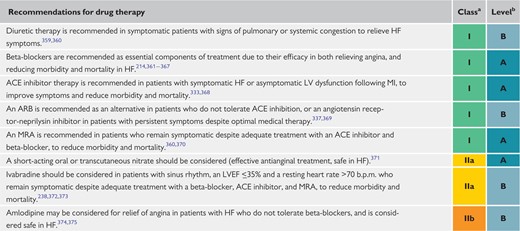 |
 |
 |
 |
ACE inhibitor = angiotensin-converting enzyme; ARB = angiotensin receptor blocker; b.p.m. = beats per minute; CCS = chronic coronary syndromes; CRT = cardiac resynchronization therapy; HF = heart failure; LBBB = left bundle branch block; LV = left ventricular; LVEF = left ventricular ejection fraction; MI = myocardial infarction; MRA = mineralocorticoid receptor antagonist.
Class of recommendation.
Level of evidence.
General recommendations for the management of patients with chronic coronary syndromes and symptomatic heart failure due to ischaemic cardiomyopathy and left ventricular systolic dysfunction
 |
 |
 |
 |
ACE inhibitor = angiotensin-converting enzyme; ARB = angiotensin receptor blocker; b.p.m. = beats per minute; CCS = chronic coronary syndromes; CRT = cardiac resynchronization therapy; HF = heart failure; LBBB = left bundle branch block; LV = left ventricular; LVEF = left ventricular ejection fraction; MI = myocardial infarction; MRA = mineralocorticoid receptor antagonist.
Class of recommendation.
Level of evidence.
5 Patients with a long-standing diagnosis of chronic coronary syndromes
In patients with a long-standing diagnosis of CCS, lifelong treatment and surveillance are required (Figure 10). The clinical course of patients with CCS may be benign over the course of time. However, patients with CCS may develop a variety of cardiovascular complications or undergo therapeutic measures, some directly related to the underlying CAD, and some having therapeutic or prognostic interactions with the underlying disease. Risk for complications may occur in an otherwise asymptomatic patient, and thus the assessment of risk status applies to symptomatic and asymptomatic patients.
Proposed algorithm according to patient types commonly observed at chronic coronary syndrome outpatient clinics. The frequency of follow-up may be subject to variation based on clinical judgement. ACS = acute coronary syndromes; CCS = chronic coronary syndromes; DAPT = dual antiplatelet therapy; ECG = electrocardiogram; LV = left ventricular; MI = myocardial infarction; PCI = percutaneous coronary intervention. aCardiologist, internist, general practitioner, or cardiovascular nurse.
Periodic assessment of the patient's individual risk may be considered (Figure 10). Scores that apply clinical parameters have been shown to predict outcomes among patients with CCS. Moreover, if the clinical parameters are complemented by biomarkers, such a risk score may be even more accurate. In 2017, a biomarker-based risk model to predict cardiovascular mortality in patients with CCS was developed and externally validated.398
5.1 Patients with stabilized symptoms <1 year after an acute coronary syndrome or patients with recent revascularization
After revascularization and/or after stabilized ACS (<1 year), patients should be monitored more vigilantly, because they are at greater risk for complications and because they are subject to changes in pharmacological treatment.45 Thus, we recommend at least two visits in the first year of follow-up. In a patient who had LV systolic dysfunction before the revascularization procedure or after the ACS, a reassessment of LV function must be considered 8–12 weeks after the intervention. Cardiac function may have improved, owing to mechanisms such as recovery from myocardial stunning or hibernation, which may be reversed by revascularization.52,53 Conversely, cardiac function may have deteriorated given other concomitant CVD (e.g. valvular disease, infection or inflammation, arrhythmia, etc.). In such cases, these other damaging factors need to be identified and treated. Likewise, non-invasive assessment of myocardial ischaemia may be considered after revascularization to rule-out residual ischaemia or to document the residual ischaemia as reference for subsequent assessments over time.
5.2 Patients >1 year after initial diagnosis or revascularization
To assess a patient's risk, an annual evaluation by a cardiovascular practitioner (cardiologist, general physician, or nurse) is warranted, even if the patient is asymptomatic. It is recommended that the annual evaluation should assess the patient's overall clinical status and medication compliance, as well as the risk profile (as reflected by risk scores). Laboratory tests—which include a lipid profile, renal function, a complete blood count, and possibly biomarkers—should be performed every 2 years.45 A patient with a worsening risk score over time may warrant more intense therapy or diagnostic measures, although risk score-guided therapy has not yet been proved to ameliorate outcomes.
A 12 lead ECG should be a part of every such visit to delineate heart rate and heart rhythm, to detect changes suggestive of silent ischaemia/infarction, and to discern abnormalities in the specific electrocardiographic segments (e.g. PR, QRS, and QT intervals). It may be beneficial to assess LV function (diastolic and systolic), valvular status, and cardiac dimensions in apparently asymptomatic patients every 3–5 years.52,53 In cases of unexplained reduction in systolic LV function, especially if regional, imaging of coronary artery anatomy is recommended. Likewise, it may be beneficial to assess non-invasively for silent ischaemia in an apparently asymptomatic patient every 3–5 years, preferably applying stress imaging. Coronary CTA should not be used for follow-up of patients with established CAD given its strength on morphological insight, but lack of functional information related to ischaemia. However, coronary CTA may be used for unique cases, such as delineation of patency of coronary artery bypass grafts.
The lipid profile and glycaemia status should be reassessed periodically to determine efficacy of treatment and, in patients without diabetes, to detect new development of diabetes. There is no evidence to support recommendations for the frequency of reassessment of these risk factors, but consensus suggests annual evaluation.
Elevated inflammatory markers, particularly of high-sensitivity C-reactive protein, have also been associated with an increased event risk in patients with and without CAD in multiple studies,25 although the robustness of the association has been questioned because of reporting and publication bias.399 In addition, von Willebrand factor, interleukin-6, and N-terminal pro-B-type natriuretic peptide (NT-proBNP) have identified as been predictors of outcome.25 Other readily available biomarkers shown to predict prognosis in patients with CCS include heart rate, haemoglobin, and white cell count.400 Scores based on aggregated biomarkers may have greater success than individual biomarkers. A multiple biomarker score combining high-sensitivity C-reactive protein, heat shock protein 70, and fibrin degradation products significantly improved C-statistics and the net reclassification index compared with a basic model using clinical data.401 Similar results were reported for a combination of high-sensitivity cardiac troponin T, NT-proBNP, and LDL-C.398 In several studies, genetic risk scores have been shown to improve risk prediction above traditional risk factors in general population samples402,403 and to predict recurrent events in populations with known CCS.404–407 Although there is additional prognostic value in using several individual and aggregated biomarkers, there is currently no evidence that routine use leads to improved care. Nevertheless, these measurements may have a role in selected patients (e.g. when testing for haemostatic abnormalities in those with previous MI without conventional risk factors or a strong family history of CAD).
Patients with unequivocal symptoms suggestive of ACS should be expeditiously referred for evaluation, applying current Guidelines for diagnosis and management. Among patients with more equivocal symptoms, stress imaging is recommended408 and, if not available and the ECG is amenable to identification of ischaemia, exercise stress electrocardiography can be used as an alternative. In patients with severe angina and a high-risk clinical profile, direct referral for ICA is recommended, provided that ad hoc physiological assessment of haemodynamic stenosis significance is readily available in the catheterization laboratory [e.g. instantaneous wave-free ratio (iwFR) or FFR]. Likewise, ICA is recommended for patients with evidence of significant ischaemia obtained by non-invasive testing.
Recommendations for patients with a long-standing diagnosis of chronic coronary syndromes
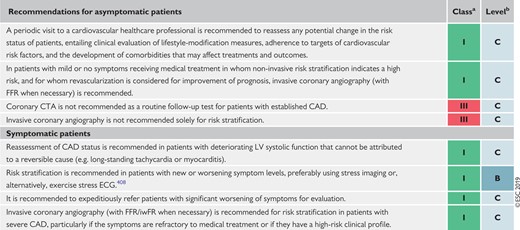 |
 |
CAD = coronary artery disease; CTA = computed tomography angiography; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio; LV = left ventricular.
Class of recommendation.
Level of evidence.
Recommendations for patients with a long-standing diagnosis of chronic coronary syndromes
 |
 |
CAD = coronary artery disease; CTA = computed tomography angiography; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio; LV = left ventricular.
Class of recommendation.
Level of evidence.
6 Angina without obstructive disease in the epicardial coronary arteries
In clinical practice, a marked discrepancy between findings regarding coronary anatomy, the presence of symptoms, and the results of non-invasive tests frequently occurs.13 These patients deserve attention, as angina and non-obstructive disease are associated with an increased risk of adverse clinical events.14 Low diagnostic yield of ICA can be explained by the presence of: (i) stenoses with mild or moderate angiographic severity, or diffuse coronary narrowing, with underestimated functional significance identified by ICA; (ii) disorders affecting the microcirculatory domain that escape the resolution of angiographic techniques; and (iii) dynamic stenoses of epicardial vessels caused by coronary spasm or intramyocardial bridges that are not evident during CTA or ICA. Intracoronary pressure measurements are useful in circumventing the first of these scenarios. For diagnostic workup, patients with angina and/or myocardial ischaemia showing coronary stenoses with non-ischaemic FFR or iwFR values may also be labelled as having non-obstructive epicardial disease.
The presence of clear-cut anginal symptoms and abnormal non-invasive tests in patients with non-obstructed epicardial vessels should lead to the suspicion of a non-obstructive cause of ischaemia. Quite often, and mainly as a result of persistence of symptoms, patients with angina and no obstructive disease undergo multiple diagnostic tests, including repeated coronary CTA or ICA, that contribute to increased healthcare costs.409 Because diagnostic pathways to investigate microcirculatory or vasomotor coronary disorders are often not implemented, a final diagnosis supported by objective evidence is seldom reached. Owing to this, patient dismay and depression are not rare in this clinical population.410,411 Of note, the use of a structured, systematic approach to explore microcirculatory and vasomotor disorders in patients with non-obstructive CAD, as delineated below, has been shown to increase diagnostic yield.412,413 Furthermore, an RCT, which reported in 2018, found that in patients with non-obstructive coronary disease, tailored treatment guided by the results of intracoronary testing [coronary flow reserve (CFR), microcirculatory resistance, and acetylcholine testing] resulted in a significant reduction of anginal symptoms, compared with conventional, non-guided medical treatment.414
6.1 Microvascular angina
Patients with microvascular angina typically have exercise-related angina, evidence of ischaemia in non-invasive tests, and either no stenoses or mild-to-moderate stenoses (40–60%), revealed by ICA or CTA, that are deemed functionally non-relevant.415 Given the similarity of angina symptoms, a microvascular origin of angina is typically suspected, after excluding obstructive epicardial coronary stenoses, during diagnostic workup of patients with suspected myocardial ischaemia. Regional LV wall motion abnormalities rarely develop during exercise or stress in patients with microvascular angina.412,416 Some patients may also have a mixed pattern of angina, with occasional episodes at rest, particularly associated with exposure to cold.
Secondary microvascular angina, in the absence of epicardial obstruction, may result from cardiac or systemic conditions, including those that cause LV hypertrophy (such as hypertrophic cardiomyopathy, aortic stenosis, and hypertensive heart disease) or inflammation (such as myocarditis or vasculitis).417
6.1.1 Risk stratification
The presence of microcirculatory dysfunction in patients with CCS entails a worse prognosis than originally thought, probably because most recent evidence has been based on follow-up of patients in whom abnormalities in the microcirculation have been objectively documented with invasive or non-invasive techniques.418–423
Microcirculatory dysfunction precedes the development of epicardial lesions, particularly in women,419 and is associated with impaired outcomes. Among patients with diabetes undergoing diagnostic workup, those without obstructive epicardial disease but with an abnormal CFR have similarly poor long-term prognosis as those with obstructive epicardial disease.421 In patients with non-significant coronary stenoses by FFR, the presence of abnormal CFR is associated with an excess of events in the long-term,418,422,423 particularly when the index of microcirculatory resistance (IMR) is also abnormal.422
6.1.2 Diagnosis
The possibility of a microcirculatory origin of angina should be considered in patients with clear-cut angina, abnormal non-invasive functional tests, and coronary vessels that are either normal or have mild stenosis deemed functionally non-significant on ICA or CTA. One of the challenges in performing a comprehensive assessment of microvascular function is testing the two main mechanisms of dysfunction separately: impaired microcirculatory conductance and arteriolar dysregulation.424–426 Yet, outlining which of these two pathways is affected is critically relevant in setting medical treatment to relieve patient symptoms.414
Impaired microcirculatory conductance can be diagnosed by measuring CFR or minimal microcirculatory resistance (the inverse of conductance). CFR can be measured non-invasively with transthoracic Doppler echocardiography [by imaging left anterior descending (LAD) flow],427 magnetic resonance imaging (myocardial perfusion index),428–430 or PET.431 Microcirculatory resistance can be measured in the catheterization laboratory by combining intracoronary pressure with thermodilution-based data (to calculate the IMR) or Doppler flow velocity (to calculate hyperaemic microvascular resistance or HMR).432,433 Both intracoronary thermodilution and Doppler allow the calculation of CFR. For decision-making purposes, values of IMR ≥25 units or CFR <2.0 are indicative of abnormal microcirculatory function.414 Both CFR and IMR are typically measured while using intravenous vasodilators, such as adenosine or regadenoson.
In contrast, the diagnosis of arteriolar dysregulation requires the assessment of endothelial function in the coronary microcirculation by selective intracoronary acetylcholine infusion (see section 6.5). In the presence of dysfunctional vascular endothelium or abnormal function of smooth muscle cells, acetylcholine (an endothelium-dependent vasodilator that also acts directly on smooth muscle cells) triggers paradoxical arteriolar vasoconstriction.434 Thus, in patients with microvascular angina and arteriolar dysregulation, acetylcholine challenge is likely to trigger microvascular spasm. This arteriolar response to acetylcholine causes anginal symptoms with or without concomitant ischaemic ECG changes, and a decrease in coronary blood flow velocity if concomitant Doppler measurements are performed. Peripheral pulse tonometry during reactive hyperaemia may also reveal abnormal systemic endothelial function in patients with angina and non-obstructive CAD.435
6.1.3 Treatment
Treatment of microvascular angina should address the dominant mechanism of microcirculatory dysfunction. In patients with abnormal CFR <2.0 or IMR ≥25 units, and a negative acetylcholine provocation test, beta-blockers, ACE inhibitors, and statins, along with lifestyle changes and weight loss, are indicated.436,437 Patients developing ECG changes and angina in response to acetylcholine testing but without severe epicardial vasoconstriction (all suggestive of microvascular spasm) may be treated like vasospastic angina patients. The effectiveness of a tailored treatment strategy was investigated in the CorMiCa trial, which randomized 151 patients to a stratified medical treatment (based on the results of CFR, IMR, and acetylcholine testing) vs. a standard-care group (including a sham interventional diagnostic procedure). At 1 year, there was a significant difference in angina scores favouring patients assigned to the stratified medical treatment arm.414
Investigations in patients with suspected coronary microvascular angina
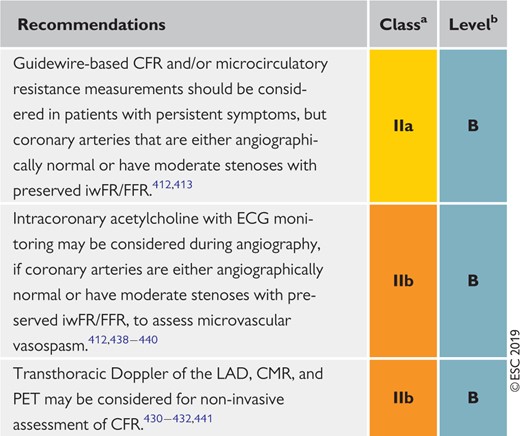 |
 |
CFR = coronary flow reserve; CMR = cardiac magnetic resonance; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio; LAD = left anterior descending; PET = positron emission tomography.
Class of recommendation.
Level of evidence.
Investigations in patients with suspected coronary microvascular angina
 |
 |
CFR = coronary flow reserve; CMR = cardiac magnetic resonance; ECG = electrocardiogram; FFR = fractional flow reserve; iwFR = instantaneous wave-free ratio; LAD = left anterior descending; PET = positron emission tomography.
Class of recommendation.
Level of evidence.
6.2 Vasospastic angina
Vasospastic angina should be suspected in patients with anginal symptoms occurring predominantly at rest, with maintained effort tolerance. The likelihood of vasospastic angina increases when attacks follow a circadian pattern, with more episodes at night and in the early morning hours. Patients are frequently younger and have fewer cardiovascular risk factors than patients with effort angina, except for cigarette smoking.442 Coronary vasospasm should be also suspected in patients with patent coronary stents and persistent angina.443,444
6.2.1 Diagnosis
The diagnosis of vasospastic angina is based on detecting transient ischaemic ST-segment changes during an angina attack (usually at rest). Patients with Prinzmetal angina represent a special subset in whom resting angina is accompanied by transient ST-segment elevation.442,445 These ECG changes correlate with proximal vessel occlusion and diffuse, distal subocclusive narrowing of epicardial vessels. As most attacks of vasospastic angina are self-limiting, documentation of these ECG changes is challenging. Ambulatory ECG monitoring, preferably with 12 lead recording, may be helpful in patients in whom vasospastic angina is suspected. The occurrence of ST-segment shifts at normal heart rate supports the likelihood of myocardial ischaemia caused by spasm. Extended Holter monitoring (for >1 week) may be required for successful documentation of transient ST-segment changes in these patients. Ambulatory ECG monitoring may also be used to assess the results of medical therapy in controlling the frequency of vasospastic events.
In patients with suspected vasospastic angina and documented ECG changes, CTA or ICA is indicated to rule-out the presence of fixed coronary stenosis. Angiographic documentation of coronary spasm requires the use of a provocation test in the catheterization laboratory. Given the low sensitivity of hyperventilation and the cold pressor test, intracoronary administration of acetylcholine or ergonovine during ICA are the preferred provocation tests.442 Both pharmacological agents are safe, provided that they are selectively infused into the left or right coronary artery, and that triggered spasm is readily controlled with intracoronary nitrates. A low percentage of patients may develop ventricular tachycardia/ventricular fibrillation or bradyarrhythmias during the provocation test (3.2 and 2.7%, respectively), similar to that reported during spontaneous spasm attacks (7%).446 Intravenous administration of ergonovine for non-invasive tests should be discouraged due to the risk of triggering prolonged spasm in multiple vessels, which may be very difficult to manage and can be fatal.447
A provocation test for coronary spasm is considered positive when it triggers: (i) anginal symptoms, (ii) ischaemic ECG changes, and (iii) severe vasoconstriction of the epicardial vessel. Should the test fail in triggering all three components, it should be considered equivocal.442 The development of angina in response to acetylcholine injections in the absence of angiographically evident spasm, with or without accompanying ST-segment changes, may indicate microvascular spasm and is seen frequently in patients presenting with microvascular angina.445
6.2.2 Treatment
In patients with epicardial or microcirculatory vasomotor disorders, CCBs and long-acting nitrates constitute the treatment of choice, in addition to the control of cardiovascular risk factors and lifestyle changes.437,445 Nifedipine has been shown to be effective in reducing coronary spasm associated with stent implantation.444
Recommendations for investigations in patients with suspected vasospastic angina
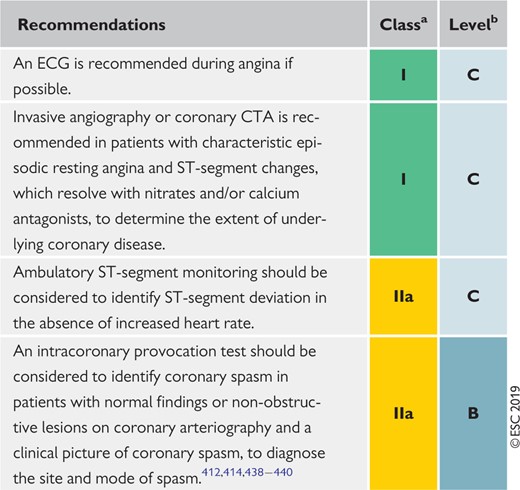 |
 |
CTA = computed tomography angiography; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
Recommendations for investigations in patients with suspected vasospastic angina
 |
 |
CTA = computed tomography angiography; ECG = electrocardiogram.
Class of recommendation.
Level of evidence.
7 Screening for coronary artery disease in asymptomatic subjects
In an effort to lower the high burden of coronary deaths in asymptomatic adults, numerous measurements of risk factors and risk markers, as well as stress tests, are often performed as screening investigations. The 2016 European Guidelines on CVD prevention in clinical practice have focused on these issues in detail.15 These recommendations have been adapted for the purpose of these Guidelines.
In general, the use of risk-estimation systems such as SCORE is recommended (see also Figure 6). Subjects with a family history of premature CAD should be screened for familial hypercholesterolaemia. Coronary calcium score, ankle−brachial index, and carotid ultrasound for plaque detection may provide useful information about the atherosclerotic risk in selected patients, but routine use of biomarkers or other imaging tests for CAD are not recommended. The new biomarkers have incremental predictive value over classical ones,448 but the net reclassification improvement is still only modest (7–18%) compared, for example, with the coronary calcium score, which has a net reclassification improvement of 66%.449
Only subjects at high event risk should be considered for further non-invasive or invasive testing. There are no data on how to manage asymptomatic subjects who receive testing and have a positive test result beyond the recommendations listed in these Guidelines. However, the principles of risk stratification, as described above for symptomatic patients, also apply to these individuals.450 It is important to remember that data demonstrating improved prognosis following appropriate management based on new biomarkers are still lacking.
It is important to note that patients with cancer and undergoing cancer treatment, or chronic inflammatory diseases such as inflammatory bowel diseases, rheumatoid arthritis, and systemic lupus erythematosus, may deserve more intensive risk screening, counselling, and management.451–454
Persons whose occupations involve public safety (e.g. airline pilots, or lorry or bus drivers), or who are professional or high-profile athletes, commonly undergo periodic testing for the assessment of exercise capacity and evaluation of possible heart disease, including CAD. Although there are insufficient data to justify this approach, these evaluations may be done for medicolegal reasons. The threshold for performing an imaging test in such persons may be lower than in the average patient. Otherwise, the same considerations as discussed above for other asymptomatic persons apply to these individuals.
Recommendations for screening for coronary artery disease in asymptomatic subjects
 |
 |
ABI = ankle−brachial index; CAD = coronary artery disease; CKD = chronic kidney disease; CTA = computed tomography angiography; CVD = cardiovascular disease; ECG = electrocardiogram; IMT = intima−media thickness; SCORE = Systematic COronary Risk Evaluation.
Class of recommendation.
Level of evidence.
Reclassifies patients better into low- or high-risk groups.
Recommendations for screening for coronary artery disease in asymptomatic subjects
 |
 |
ABI = ankle−brachial index; CAD = coronary artery disease; CKD = chronic kidney disease; CTA = computed tomography angiography; CVD = cardiovascular disease; ECG = electrocardiogram; IMT = intima−media thickness; SCORE = Systematic COronary Risk Evaluation.
Class of recommendation.
Level of evidence.
Reclassifies patients better into low- or high-risk groups.
8 Chronic coronary syndromes in specific circumstances
8.1 Cardiovascular comorbidities
8.1.1 Hypertension
Hypertension is the most prevalent cardiovascular risk factor and is closely associated with CCS. Thresholds for the definition of hypertension are provided in Table 10. BP lowering can significantly reduce major cardiovascular risk, including CHD. Meta-analysis suggests that for every 10 mmHg reduction in systolic BP, CAD can be reduced by 17%.463 More intensive BP targets (office BP <130 mmHg) have been associated with favourable outcomes and are endorsed by the 2018 ESC/ESH Guidelines for the management of arterial hypertension.464 It is recommended treat hypertensive patients with CCS are treated to office targets of 130/80 mmHg, because an increased systolic BP of ≥140 mmHg and diastolic BP of ≥80 mmHg, but also a systolic BP of <120 mmHg and diastolic BP of <70 mmHg, are associated with increased risk465,466 (Table 10). Whether the J-curve phenomenon exists in patients with revascularized CAD remains uncertain. In hypertensive patients with CHD, beta-blockers and RAS blockers may improve post-MI outcomes.467 In patients with symptomatic angina, beta-blockers and calcium antagonists are the preferred components of the drug-treatment strategy. The combination of ACE inhibitors and ARBs is not recommended for the treatment of hypertension because of increased renal adverse events without beneficially influencing outcome.468,469
| Category . | Systolic BP (mmHg) . | . | Diastolic BP (mmHg) . |
|---|---|---|---|
| Office BP | ≥140 | and/or | ≥90 |
| ≥80 years of age | ≥160 | and/or | ≥90 |
| Ambulatory BP | |||
| Daytime (or awake) | ≥135 | and/or | ≥85 |
| Night-time (or asleep) | ≥120 | and/or | ≥70 |
| 24 h | ≥130 | and/or | ≥80 |
| Home BP | ≥135 | and/or | ≥85 |
| Category . | Systolic BP (mmHg) . | . | Diastolic BP (mmHg) . |
|---|---|---|---|
| Office BP | ≥140 | and/or | ≥90 |
| ≥80 years of age | ≥160 | and/or | ≥90 |
| Ambulatory BP | |||
| Daytime (or awake) | ≥135 | and/or | ≥85 |
| Night-time (or asleep) | ≥120 | and/or | ≥70 |
| 24 h | ≥130 | and/or | ≥80 |
| Home BP | ≥135 | and/or | ≥85 |
BP = blood pressure.
| Category . | Systolic BP (mmHg) . | . | Diastolic BP (mmHg) . |
|---|---|---|---|
| Office BP | ≥140 | and/or | ≥90 |
| ≥80 years of age | ≥160 | and/or | ≥90 |
| Ambulatory BP | |||
| Daytime (or awake) | ≥135 | and/or | ≥85 |
| Night-time (or asleep) | ≥120 | and/or | ≥70 |
| 24 h | ≥130 | and/or | ≥80 |
| Home BP | ≥135 | and/or | ≥85 |
| Category . | Systolic BP (mmHg) . | . | Diastolic BP (mmHg) . |
|---|---|---|---|
| Office BP | ≥140 | and/or | ≥90 |
| ≥80 years of age | ≥160 | and/or | ≥90 |
| Ambulatory BP | |||
| Daytime (or awake) | ≥135 | and/or | ≥85 |
| Night-time (or asleep) | ≥120 | and/or | ≥70 |
| 24 h | ≥130 | and/or | ≥80 |
| Home BP | ≥135 | and/or | ≥85 |
BP = blood pressure.
Recommendations for hypertension treatment in chronic coronary syndromes
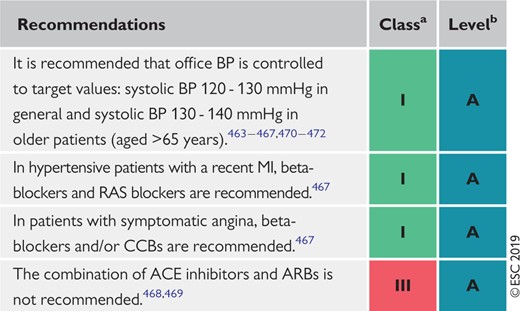 |
 |
ACE = angiotensin converting enzyme; ARB = angiotensin receptor blocker; BP = blood pressure; CCB = calcium channel blocker; RAS = renin−angiotensin system.
Class of recommendation.
Level of evidence.
Recommendations for hypertension treatment in chronic coronary syndromes
 |
 |
ACE = angiotensin converting enzyme; ARB = angiotensin receptor blocker; BP = blood pressure; CCB = calcium channel blocker; RAS = renin−angiotensin system.
Class of recommendation.
Level of evidence.
8.1.2 Valvular heart disease (including planned transcatheter aortic valve implantation)
Coronary angiography for the assessment of CAD is recommended before valve surgery or when percutaneous valvular intervention is planned, to determine if revascularization is required. Coronary CTA may be considered in patients with low risk for CAD, or in patients in whom conventional ICA is technically not feasible or associated with increased risk. The combination of PCI and transcatheter aortic valve implantation appears feasible and safe, but more data are needed before definite recommendations can be provided.473,474 The routine use of stress testing to detect CAD associated with severe symptomatic valvular disease is not recommended because of low diagnostic value and potential risk. Symptom-limited stress testing in patients with valvular heart disease appears safe, and may be useful to unmask symptoms in asymptomatic patients or in patients with equivocal symptoms.475
Recommendations for valvular disease in chronic coronary syndromes476
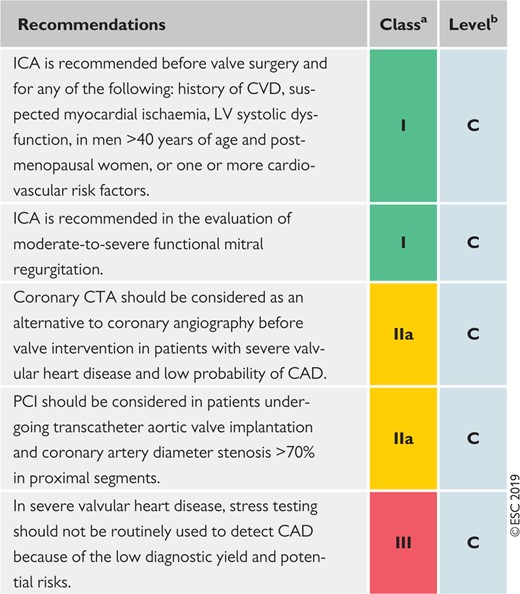 |
 |
CAD = coronary artery disease; CTA = computed tomography angiography; CVD = cardiovascular disease; ICA = invasive coronary angiography; LV = left ventricular; PCI = percutaneous coronary intervention.
Class of recommendation.
Level of evidence.
Recommendations for valvular disease in chronic coronary syndromes476
 |
 |
CAD = coronary artery disease; CTA = computed tomography angiography; CVD = cardiovascular disease; ICA = invasive coronary angiography; LV = left ventricular; PCI = percutaneous coronary intervention.
Class of recommendation.
Level of evidence.
8.1.3 After heart transplantation
The follow-up and assessment of long-term heart transplant survivors requires specific know-how. Transplant CAD is largely an immunological phenomenon, and remains a significant cause of morbidity and mortality.477 ICA is recommended for the assessment of transplant CAD and should be performed annually for 5 years after transplantation. If there are no significant abnormalities, angiograms can be performed biannually thereafter. Intravascular ultrasound examinations may be useful in assessing cardiac allograft vasculopathy and plaque stability.478 Treatment options for CAD in transplant recipients include pharmacotherapy and revascularization. PCI in the transplanted heart has become an established therapy.479
8.2 Non-cardiovascular comorbidities
8.2.1 Cancer
Occurrence of CAD in patients with active cancer is increasing451,452 as a side effect of cancer therapy (i.e. radiotherapy to the thorax/mediastinum, cardiotoxic chemotherapy, or immunotherapies) or a result of extended cancer therapies in elderly individuals. CAD in patients with active cancer is associated with challenges for clinicians as treatment decisions should be the subject of individualized discussions based on life expectancy, additional comorbidities such as thrombocytopenia, increased thrombosis and bleeding propensity, and potential interactions between drugs used in CCS management and antineoplastic drugs. In cancer patients with increased frailty, the least invasive revascularization procedures are recommended. For further information, see the ESC position paper on cancer treatments and cardiovascular toxicity.480
Recommendations for active cancer in chronic coronary syndromes
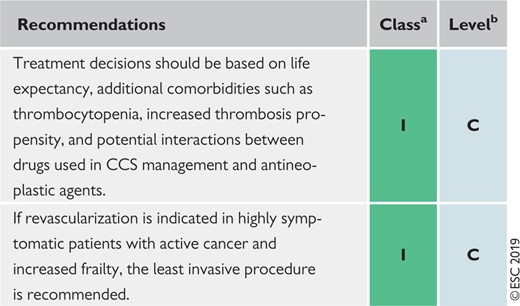 |
 |
CCS = chronic coronary syndromes.
Class of recommendation.
Level of evidence.
Recommendations for active cancer in chronic coronary syndromes
 |
 |
CCS = chronic coronary syndromes.
Class of recommendation.
Level of evidence.
8.2.2 Diabetes mellitus
Diabetes mellitus confers about a two-fold increased risk for CAD481 and, consequently, control of risk factors is recommended for the prevention of CVD. Systolic BP in patients with diabetes should be targeted to ≤130 mmHg, if tolerated, but not <120 mmHg, and diastolic BP to <80 mmHg, but not <70 mmHg.482 Initial antihypertension treatment should consist of a combination of a RAS blocker with a CCB or thiazide/thiazide-like diuretic. ACE inhibitors reduce albuminuria, and the appearance or progression of diabetic nephropathy, more effectively than other drug classes.482 Patients with diabetes and CAD are considered to be at very high risk; consequently, LDL-C should be lowered to <1.8 mmol/L (<70 mg/dL) or reduced by ≥50% if the baseline LCL-C is between 1.8 and 3.5 mmol/L (70 and 135 mg/dL).15 For the majority of patients with diabetes and CAD, a target glycated HbA1c level of <7% (<53 mmol/L) is recommended.483,484 Large safety studies on new glucose-lowering drugs, namely sodium-glucose co-transporter-2 and glucagon-like peptide-1 receptor agonists, have demonstrated significant reductions in cardiovascular events. Indications for their clinical use are described in the 2019 ESC/European Association for the Study of Diabetes Guidelines on diabetes mellitus, pre-diabetes, and cardiovascular diseases.16
A 12 lead ECG is recommended as part of the routine assessment for screening for conduction abnormalities, LV hypertrophy, and arrhythmias. The high prevalence of significant CAD and prohibitively high cardiovascular mortality may suggest the usefulness of routine screening for CAD (with functional imaging testing or coronary CTA) in asymptomatic patients with diabetes, but no data have shown an improvement in outcomes so far. Routine use of CTA in asymptomatic patients with diabetes is therefore not recommended.
Recommendations for diabetes mellitus in chronic coronary syndromes
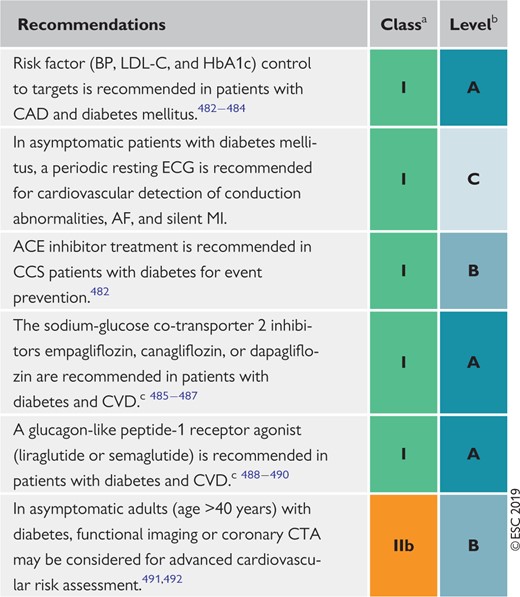 |
 |
ACE = angiotensin-converting enzyme; AF = atrial fibrillation; BP = blood pressure; CAD = coronary artery disease; CCS = chronic coronary syndromes; CTA = computed tomography angiography; CVD = cardiovascular disease; ECG = electrocardiogram; HbA1c = glycated haemoglobin; LDL-C = low-density lipoprotein cholesterol; MI = myocardial infarction.
Class of recommendation.
Level of evidence.
Treatment algorithm is available in the 2019 European Society of Cardiology/European Association for the Study of Diabetes Guidelines on diabetes mellitus, pre-diabetes, and cardiovascular diseases.16
Recommendations for diabetes mellitus in chronic coronary syndromes
 |
 |
ACE = angiotensin-converting enzyme; AF = atrial fibrillation; BP = blood pressure; CAD = coronary artery disease; CCS = chronic coronary syndromes; CTA = computed tomography angiography; CVD = cardiovascular disease; ECG = electrocardiogram; HbA1c = glycated haemoglobin; LDL-C = low-density lipoprotein cholesterol; MI = myocardial infarction.
Class of recommendation.
Level of evidence.
Treatment algorithm is available in the 2019 European Society of Cardiology/European Association for the Study of Diabetes Guidelines on diabetes mellitus, pre-diabetes, and cardiovascular diseases.16
8.2.3 Chronic kidney disease
CAD is highly prevalent in patients with CKD and a growing number of patients undergoing PCI have concomitant CKD.493 There is a linear increase in the risk of cardiovascular mortality with decreasing GFR.494 Medical treatment for risk-factor control (lipids, BP, and glucose) can improve outcomes. Special attention during the workup for CKD patients with suspected obstructive CAD should be paid to the fact that angina is less common and silent ischaemia more common.495 Additionally, non-invasive stress testing shows reduced accuracy in patients with CKD.496 The use of an iodinated contrast agent should be minimized to prevent further deterioration of renal function. Decisions regarding diagnostic and treatment modalities should be made accordingly. Interestingly, patients with CKD are less likely to receive invasive management for treatment of CAD compared with those without, although benefits of invasive management have been reported.497 Revascularization options in patients with CKD include CABG and PCI. Meta-analyses suggest that CABG is associated with higher short-term risk of death, stroke, and repeat revascularization, whereas PCI with a new-generation DES is associated with a higher long-term risk of repeat revascularization.498,499 Data on patients on haemodialysis are very limited, making generalizable treatment recommendations difficult.
Recommendations for chronic kidney disease in chronic coronary syndromes
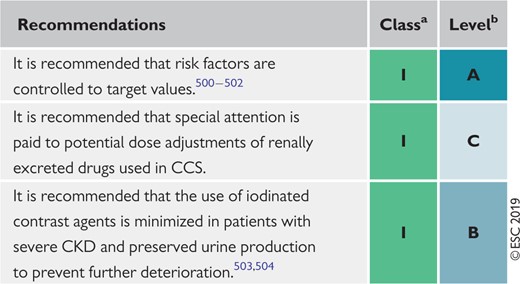 |
 |
CKD = chronic coronary disease; CCS = chronic coronary syndromes.
Class of recommendation.
Level of evidence.
Recommendations for chronic kidney disease in chronic coronary syndromes
 |
 |
CKD = chronic coronary disease; CCS = chronic coronary syndromes.
Class of recommendation.
Level of evidence.
8.2.4 Elderly
Ageing predisposes patients to a high incidence and prevalence of CAD, in both men and women. Elderly patients (age >75 years) have the greatest mortality and morbidity risk attributable to CCS, which is enriched by the high prevalence of comorbidities (e.g. hypertension, diabetes mellitus, CKD, etc.).505 Although the prevalence of elderly patients with CAD is increasing, this population is usually undertreated, underdiagnosed, and under-represented in clinical trials. Elderly patients often present with atypical symptoms, which may delay proper diagnosis. The treatment of CCS in the elderly is complicated by a higher vulnerability to complications for both conservative and invasive strategies, such as bleeding, renal failure, and neurological impairments, all of which require special attention. It is recommended that radial access is used whenever possible to reduce access-site complications, when choosing an invasive strategy for patient management.506,507 The use of DES, compared with bare-metal stents, in combination with a short duration of DAPT is associated with significant safety and efficacy benefits in elderly patients.508,509
Recommendations for elderly patients with chronic coronary syndromes
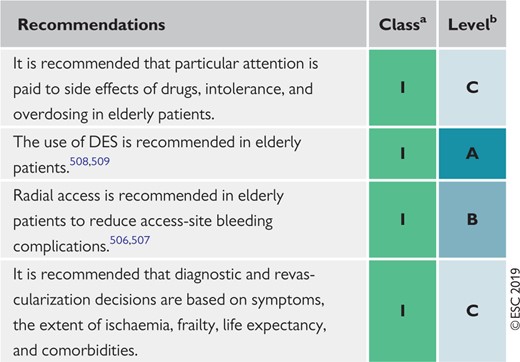 |
 |
DES = drug-eluting stents.
Class of recommendation.
Level of evidence.
Recommendations for elderly patients with chronic coronary syndromes
 |
 |
DES = drug-eluting stents.
Class of recommendation.
Level of evidence.
8.3 Sex
Making up ≤30% of study populations, women are widely under-represented in cardiovascular studies.510 This recruitment bias causes an evidence gap, as sex-based randomized controlled studies are lacking, and most data are extracted from meta-analyses and post hoc analyses of trials in ACS patients. Differences in symptom presentation, the accuracy of diagnostic tests for CAD, and other factors that lead to differential triage, evaluation, or early treatment of women with myocardial ischaemia compared with men could contribute to their worse outcomes.511–514 Whether there are true sex-related differences in mortality after myocardial ischaemia, or whether they owing to older age or a higher prevalence of coexisting diseases in women, remains incompletely understood. It has become evident that sex-related mortality differences are particularly apparent in younger patients, typically those aged <60 years.511,512,515 The reasons for this age-dependent disparity in mortality remain unclear. Women tend to be treated less aggressively than men.515 However, patient characteristics and treatments do not entirely account for sex differences in outcomes, even after PCI.512 It is therefore recommended that women who present with signs suggestive of cardiac ischaemia undergo careful investigation, as clinical symptoms might be atypical. The diagnostic accuracy of the exercise ECG is even lower in women than in men, which is in part related to functional impairment, precluding some women from achieving an adequate workload. Stress echocardiography with exercise or dobutamine stress is an accurate, non-invasive technique for the detection of obstructive CAD and risk among women with suspected CCS.516 Both women and men have experienced improvements in mortality when new-generation DES were used.517–519 The mortality reductions were similar among women and men leaving sex disparities in outcomes unchanged.512 Women have higher complication rates following CABG and may also have higher mortality risk,520,521 especially in elderly patients. Hormone replacement therapy in post-menopausal women does not reduce the risk of ischaemic myocardial disease (see section 3.3.5), and is therefore not recommended for primary and secondary prevention.344,522,523
Recommendation for sex issues and chronic coronary syndromes
 |
 |
Class of recommendation.
Level of evidence.
Recommendation for sex issues and chronic coronary syndromes
 |
 |
Class of recommendation.
Level of evidence.
8.4 Patients with refractory angina
Refractory angina refers to long-lasting symptoms (for ≥3 months) due to established reversible ischaemia in the presence of obstructive CAD, which cannot be controlled by escalating medical therapy with the use of second- and third-line pharmacological agents, bypass grafting, or stenting including PCI of chronic total coronary occlusion. Incidence is growing with more advanced CAD, multiple comorbidities, and ageing of the population. The quality of life of patients with refractory angina is poor, with frequent hospitalization and a high level of resource utilization. The number of potential treatment options is increasing, but the level of evidence in support of their safety and efficacy varies from non-existent (in the case of transmyocardial laser application) to promising. RCTs with endpoints such as the severity and frequency of angina, as well as quality of life, are obviously needed, along with safety metrics. To confirm treatment efficacy, trials with a sham-controlled design are desirable, a significant placebo effect being part of the therapeutic effect. Patients with refractory angina are best treated in dedicated ‘angina clinics’ by multidisciplinary teams experienced in selecting the most suitable therapeutic approach in the individual patient based on an accurate diagnosis of the mechanisms of the pain syndrome. Once conventional anti-ischaemic targets have been exhausted (through an increase in nutrient blood flow delivery and/or reduction in oxygen consumption), novel therapies can be ranked by mechanism of action: promotion of collateral growth, transmural redistribution of blood flow, and neuromodulation of the cardiac pain syndrome (Table 11).
Potential treatment options for refractory angina and summary of trial data
| Therapy . | Type of therapy . | RCT . | Type of control group . | Number of patients enrolled . |
|---|---|---|---|---|
| External counterpulsation | Enhanced external counterpulsation | MUST524 | Sham | 139 |
| Extracorporeal shockwave | Low-energy extracorporeal shockwave therapy | Not available | Not available | — |
| Coronary sinus constriction | Reducer device | COSIRA525 | Sham | 104 |
| Neuromodulation | Spinal cord stimulation | STARTSTIM526 | Not available | 68 |
| Transcutaneous electrical neural stimulation | Not available | Not available | — | |
| Subcutaneous electrical neural stimulation | Not available | Not available | — | |
| Sympathectomy | Denby et al.527 | Placebo | 65 | |
| Gene therapy | Adenovirus fibroblast growth factor 5 | Not available | Not available | — |
| Autologous cell therapy | Mononuclear bone marrow-derived haematopoietic progenitor cells | RENEW528 | Placebo | 112 |
| Therapy . | Type of therapy . | RCT . | Type of control group . | Number of patients enrolled . |
|---|---|---|---|---|
| External counterpulsation | Enhanced external counterpulsation | MUST524 | Sham | 139 |
| Extracorporeal shockwave | Low-energy extracorporeal shockwave therapy | Not available | Not available | — |
| Coronary sinus constriction | Reducer device | COSIRA525 | Sham | 104 |
| Neuromodulation | Spinal cord stimulation | STARTSTIM526 | Not available | 68 |
| Transcutaneous electrical neural stimulation | Not available | Not available | — | |
| Subcutaneous electrical neural stimulation | Not available | Not available | — | |
| Sympathectomy | Denby et al.527 | Placebo | 65 | |
| Gene therapy | Adenovirus fibroblast growth factor 5 | Not available | Not available | — |
| Autologous cell therapy | Mononuclear bone marrow-derived haematopoietic progenitor cells | RENEW528 | Placebo | 112 |
RCT = randomized clinical trial.
Potential treatment options for refractory angina and summary of trial data
| Therapy . | Type of therapy . | RCT . | Type of control group . | Number of patients enrolled . |
|---|---|---|---|---|
| External counterpulsation | Enhanced external counterpulsation | MUST524 | Sham | 139 |
| Extracorporeal shockwave | Low-energy extracorporeal shockwave therapy | Not available | Not available | — |
| Coronary sinus constriction | Reducer device | COSIRA525 | Sham | 104 |
| Neuromodulation | Spinal cord stimulation | STARTSTIM526 | Not available | 68 |
| Transcutaneous electrical neural stimulation | Not available | Not available | — | |
| Subcutaneous electrical neural stimulation | Not available | Not available | — | |
| Sympathectomy | Denby et al.527 | Placebo | 65 | |
| Gene therapy | Adenovirus fibroblast growth factor 5 | Not available | Not available | — |
| Autologous cell therapy | Mononuclear bone marrow-derived haematopoietic progenitor cells | RENEW528 | Placebo | 112 |
| Therapy . | Type of therapy . | RCT . | Type of control group . | Number of patients enrolled . |
|---|---|---|---|---|
| External counterpulsation | Enhanced external counterpulsation | MUST524 | Sham | 139 |
| Extracorporeal shockwave | Low-energy extracorporeal shockwave therapy | Not available | Not available | — |
| Coronary sinus constriction | Reducer device | COSIRA525 | Sham | 104 |
| Neuromodulation | Spinal cord stimulation | STARTSTIM526 | Not available | 68 |
| Transcutaneous electrical neural stimulation | Not available | Not available | — | |
| Subcutaneous electrical neural stimulation | Not available | Not available | — | |
| Sympathectomy | Denby et al.527 | Placebo | 65 | |
| Gene therapy | Adenovirus fibroblast growth factor 5 | Not available | Not available | — |
| Autologous cell therapy | Mononuclear bone marrow-derived haematopoietic progenitor cells | RENEW528 | Placebo | 112 |
RCT = randomized clinical trial.
Both the STARTSTIM and RENEW (Efficacy and Safety of Targeted Intramyocardial Delivery of Auto CD34+ Stem Cells for Improving Exercise Capacity in Subjects With Refractory Angina) trials were underpowered due to premature study termination. Of note, a patient-level pooled analysis of 304 patients included in three double-blind, cell therapy, placebo-controlled trials, among which was the RENEW trial, showed that active treatment with autologous haematopoietic cells had significant effects on exercise time and angina frequency.528
Based on positive results from two RCTs in small groups of patients, both enhanced external counterpulsation and the coronary sinus reducer device represent alternative options in patients with refractory angina, which is resistant after having exhausted all options for medical therapy and mechanical revascularization. Controlled coronary sinus narrowing with the implantation of a large stainless-steel device increases coronary sinus pressure, leading to improved perfusion in the LAD territory.
Total reported experience with all novel therapeutic options remains limited, both regarding the number of treated patients and the duration of follow-up. Larger RCTs are required to define the role of each treatment modality for specific subgroups, to decrease non-responder rates and ascertain benefit beyond potential placebo effects.
Recommendations for treatment options for refractory angina
 |
 |
Class of recommendation.
Level of evidence.
Recommendations for treatment options for refractory angina
 |
 |
Class of recommendation.
Level of evidence.
9 Key messages
Careful evaluation of patient history, including the characterization of anginal symptoms, and evaluation of risk factors and manifestations of CVD, as well as proper physical examination and basic testing, are crucial for the diagnosis and management of CCS.
Unless obstructive CAD can be excluded based on clinical evaluation alone, either non-invasive functional imaging or anatomical imaging using coronary CTA may be used as the initial test to rule-out or establish the diagnosis of CCS.
Selection of the initial non-invasive diagnostic test is based on the PTP, the test’s performance in ruling-in or ruling-out obstructive CAD, patient characteristics, local expertise, and the availability of the test.
For revascularization decisions, both anatomy and functional evaluation are to be considered. Either non-invasive or invasive functional evaluation is required for the assessment of myocardial ischaemia associated with angiographic stenosis, unless very high grade (>90% diameter stenosis).
Assessment of risk serves to identify CCS patients at high event risk who are projected to derive prognostic benefit from revascularization. Risk stratification includes the assessment of LV function.
Patients at high event risk should undergo invasive investigation for consideration of revascularization, even if they have mild or no symptoms.
Implementation of healthy lifestyle behaviours decreases the risk of subsequent cardiovascular events and mortality, and is additional to appropriate secondary prevention therapy. Clinicians should advise on and encourage necessary lifestyle changes in every clinical encounter.
Cognitive behavioural interventions such as supporting patients to set realistic goals, self-monitor, plan how to implement changes and deal with difficult situations, set environmental cues, and engage social support are effective interventions for behaviour change.
Multidisciplinary teams can provide patients with support to make healthy lifestyle changes, and address challenging aspects of behaviour and risk.
Anti-ischaemic treatment must be adapted to the individual patient based on comorbidities, co-administered therapies, expected tolerance and adherence, and patient preferences. The choice of anti-ischaemic drugs to treat CCS should be adapted to the patient’s heart rate, BP, and LV function.
Beta-blockers and/or CCBs remain the first-line drugs in patients with CCS. Beta-blockers are recommended in patients with LV dysfunction or HF with reduced ejection fraction.
Long-acting nitrates provoke tolerance with loss of efficacy. This requires prescription of a daily nitrate-free or nitrate-low interval of ∼10–14 h.
Antithrombotic therapy is a key part of secondary prevention in patients with CCS and warrants careful consideration. Patients with a previous MI, who are at high risk of ischaemic events and low risk of fatal bleeding, should be considered for long-term DAPT with aspirin and either a P2Y12 inhibitor or very low-dose rivaroxaban, unless they have an indication for an OAC such as AF.
Statins are recommended in all patients with CCS. ACE inhibitors (or ARBs) are recommended in the presence of HF, diabetes, or hypertension and should be considered in high-risk patients.
Proton pump inhibitors are recommended in patients receiving aspirin or combination antithrombotic therapy who are at high risk of gastrointestinal bleeding.
Efforts should be made to explain to patients the importance of evidence-based prescriptions to increase adherence to treatment, and repeated therapeutic education is essential in every clinical encounter.
Patients with a long-standing diagnosis of CCS should undergo periodic visits to assess potential changes in risk status, adherence to treatment targets, and the development of comorbidities. Repeat stress imaging or ICA with functional testing is recommended in the presence of worsening symptoms and/or increased risk status.
Assessment of myocardial and valvular function and dimensions, as well as a functional test to rule-out significant myocardial silent ischaemia, may be contemplated every 3–5 years in asymptomatic patients with a long-standing diagnosis of CCS.
An assessment of coronary vasomotor function should be considered in patients with non-significant epicardial CAD and objective evidence of ischaemia.
10 Gaps in the evidence
10.1 Diagnosis and assessment
More information on the effects of various risk factors, biomarkers, and comorbidities on the PTP of obstructive CAD is needed. Adequately powered RCTs are needed to compare the effectiveness of different diagnostic strategies, and to evaluate how to best integrate diagnostic tests in patient care in terms of clinical outcomes and the use of healthcare resources.
10.2 Assessment of risk
Studies should address whether an initial invasive strategy, in addition to optimal medical therapy in patients with CCS and inducible ischaemia by non-invasive testing, improves outcomes. Larger trials are needed to verify the utility of systematic assessment of biomarkers in patients with suspected obstructive CAD.
10.3 Lifestyle management
Research regarding the most effective methods to support healthy lifestyle behaviours in brief or very brief clinical encounters, and sustain medication and lifestyle behaviour adherence over time, is needed. The cardiovascular effects of newer e-cigarettes over the long-term remain unknown, as does their effectiveness in smoking cessation.
The relative benefits of high-intensity interval training vs. moderate-intensity exercise in patients with CCS should be further evaluated. The benefits of decreasing sedentary behaviour, and the most appropriate ‘dose’ and type of physical activity in patients with CCS, are unknown, as are the effectiveness and cost-effectiveness of increasing cardiac rehabilitation participation among patients with CCS.
10.4 Pharmacological management
The need for and duration of beta-blocker therapy following MI to maintain a protective effect on cardiac events in the absence of LV systolic dysfunction are unknown.
In patients with CCS and without a previous MI, it remains to be determined whether current anti-ischaemic drugs improve prognosis.
Whether the initial use of second-line anti-ischaemic therapy (i.e. long-acting nitrates, ranolazine, nicorandil, ivabradine, or trimetazidine) alone or in combination with a first-line drug (i.e. beta-blocker or CCB) is superior to the combination of a beta-blocker with a CCB to control anginal symptoms and myocardial ischaemia in patients with CCS remains to be proven.
The efficacy and safety of aspirin or an alternative antithrombotic therapy in patients with a mild extent of atherosclerotic disease, such as that discovered by coronary CTA, requires further assessment, including the effect on cancer rates as well as cardiovascular events. The optimal long-term antithrombotic therapy, and strategies for individualizing this, in patients at high risk of ischaemic events is uncertain. Consequently, clinical studies comparing the efficacy and safety of aspirin + P2Y12 inhibitor with aspirin + factor Xa inhibitor are warranted to determine which subgroups may be preferentially treated with one or other strategy. The potential clinical benefit of ticagrelor monotherapy, while stopping aspirin, remains unproved at present.
The role of biomarkers in stratifying patients' risk of ischaemic events and bleeding requires clarification, including the role of growth differentiation factor-15 in guiding the risk of bleeding with DAPT. It is uncertain what effect novel lipid-lowering strategies will have on the net clinical benefit of DAPT, with similar implications of other strategies such as intensive BP lowering and, potentially in the future, selective anti-inflammatory therapies.
10.5 Revascularization
Further studies, including RCTs, are needed to assess the value of functional vs. anatomical guidance for CABG. The concept of complete revascularization and its effect on prognosis needs to be re-evaluated by prospective comparisons of functional vs. anatomical guidance for stenting on the one hand, and bypass on the other. Of note, none of the RCTs comparing PCI with CABG to date have used combined anatomical and functional guidance for PCI, a strategy that is suggested to significantly improve outcomes of PCI (Syntax II registry).
10.6 Heart failure and left ventricular dysfunction
Most of the evidence from RCTs supporting the recommendations for the use of drugs and devices in patients with chronic heart failure is based on cohorts with stable ischaemic heart disease and reduced LV function. However, patients with CCS requiring acute or chronic mechanical support are largely excluded from clinical trials, and the optimal management of such patients with drugs and devices during episodes of acute decompensation has not been adequately addressed.
10.7 Patients with long-standing diagnosis of chronic coronary syndromes
The incremental value of using risk scores to serially evaluate patients' risks, and more importantly to adjust the intensity of treatment, remains to be determined.
The optimal time intervals for serial visits remain to be determined.
10.8 Angina without obstructive coronary artery disease
Development of safe and efficacious novel pharmacological agents for this indication remains an unmet need.
10.9 Screening in asymptomatic subjects
Further studies on biomarkers and imaging tests for screening of CAD in asymptomatic subjects are needed. Furthermore, there are limited data on how to manage asymptomatic subjects who receive testing and have a positive test result, as evidence demonstrating improved prognosis following appropriate management is still lacking.
10.10 Comorbidities
The role of PCI in patients with aortic stenosis remains undetermined with respect to the indication for coronary revascularization and timing vs. valve intervention. Further information is needed on how to adapt cardiovascular therapies in patients with chronic inflammatory diseases.
10.11 Patients with refractory angina
Larger RCTs and registries are required to define the role of additional treatment modalities for specific subgroups, to decrease non-responder rates and ascertain benefit beyond potential placebo effects.
11 ‘What to do’ and ‘what not to do’ messages from the Guidelines
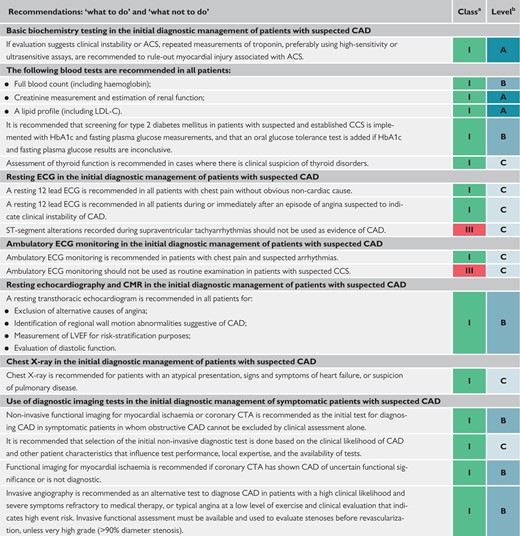 |
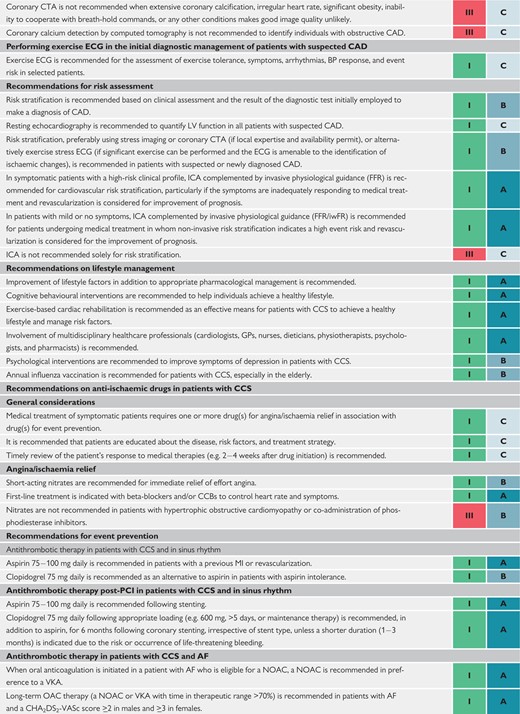 |
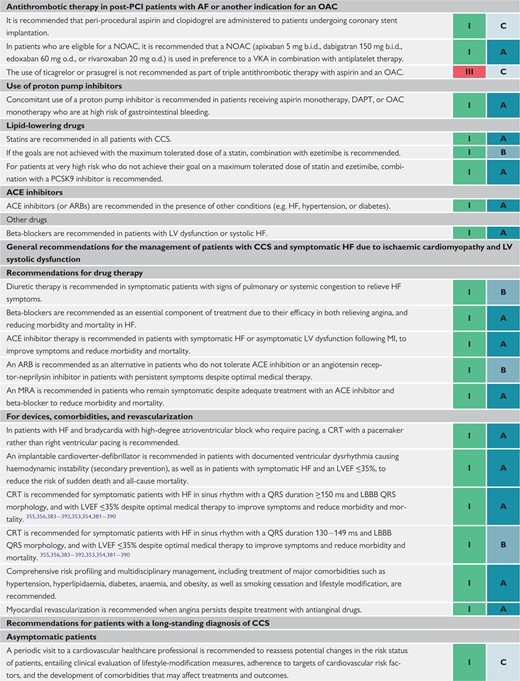 |
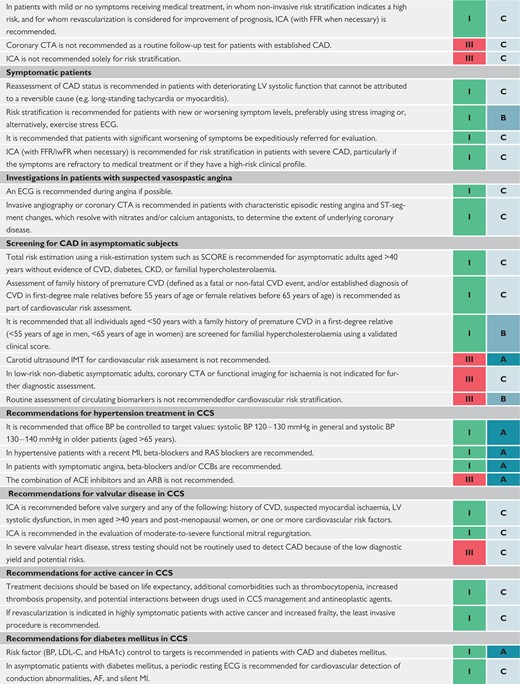 |
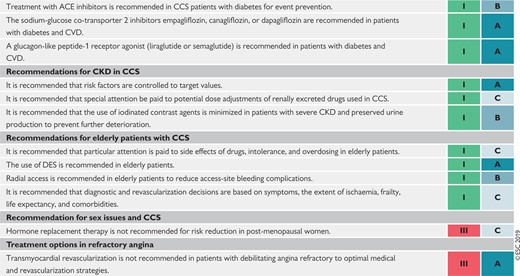 |
 |
 |
 |
 |
 |
ACE = angiotensin-converting enzyme; ACS = acute coronary syndromes; AF = atrial fibrillation; ARB = angiotensin receptor blocker; b.i.d. = bis in die (twice a day); BP = blood pressure; CHA2DS2-VASc = Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]; CAD = coronary artery disease; CCB = calcium channel blocker; CCS = chronic coronary syndromes; CKD = chronic kidney disease; CMR = cardiac magnetic resonance; CRT = cardiac resynchronization therapy; CTA = computed tomography angiography; CVD = cardiovascular disease; DAPT = dual antiplatelet therapy; DES = drug-eluting stent; ECG = electrocardiogram; FFR = fractional flow reserve; GPs = general practitioners; HbA1C = glycated haemoglobin; HF = heart failure; ICA = invasive coronary angiography; IMT = intima−media thickness; iwFR = instantaneous wave-free ratio (instant flow reserve); LBBB = left bundle branch block; LDL-C = low-density lipoprotein cholesterol; LV = left ventricular; LVEF = left ventricular ejection fraction; MI = myocardial infarction; MRA = mineralocorticoid receptor antagonist; NOAC = non-vitamin K antagonist oral anticoagulant; OAC = oral anticoagulant; o.d. = omni die (once a day); PCI = percutaneous coronary intervention; PCSK9 = proprotein convertase subtilisin-kexin type 9; RAS = renin−angiotensin system; VKA = vitamin K antagonist.
Class of recommendation.
Level of evidence.
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
ACE = angiotensin-converting enzyme; ACS = acute coronary syndromes; AF = atrial fibrillation; ARB = angiotensin receptor blocker; b.i.d. = bis in die (twice a day); BP = blood pressure; CHA2DS2-VASc = Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65–74 and Sex category [Female]; CAD = coronary artery disease; CCB = calcium channel blocker; CCS = chronic coronary syndromes; CKD = chronic kidney disease; CMR = cardiac magnetic resonance; CRT = cardiac resynchronization therapy; CTA = computed tomography angiography; CVD = cardiovascular disease; DAPT = dual antiplatelet therapy; DES = drug-eluting stent; ECG = electrocardiogram; FFR = fractional flow reserve; GPs = general practitioners; HbA1C = glycated haemoglobin; HF = heart failure; ICA = invasive coronary angiography; IMT = intima−media thickness; iwFR = instantaneous wave-free ratio (instant flow reserve); LBBB = left bundle branch block; LDL-C = low-density lipoprotein cholesterol; LV = left ventricular; LVEF = left ventricular ejection fraction; MI = myocardial infarction; MRA = mineralocorticoid receptor antagonist; NOAC = non-vitamin K antagonist oral anticoagulant; OAC = oral anticoagulant; o.d. = omni die (once a day); PCI = percutaneous coronary intervention; PCSK9 = proprotein convertase subtilisin-kexin type 9; RAS = renin−angiotensin system; VKA = vitamin K antagonist.
Class of recommendation.
Level of evidence.
12 Supplementary data
Supplementary Data with additional Supplementary Tables and Figures complementing the full text—as well as section 3 on patients with angina and/or dyspnoea, and suspected coronary artery disease—are available on the European Heart Journal website and via the ESC website at www.escardio.org/guidelines.
13 Appendix
Author/Task Force Member affiliations:
Stephan Achenbach, Department of Cardiology, Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, Germany; Stefan Agewall, Department of Medicine, Clinical Science, Oslo, Norway; Emanuele Barbato, Advanced Biomedical Sciences, University Federico II, Naples, Italy; Jeroen J. Bax, Cardiology, Leiden University Medical Center, Leiden, Netherlands; Davide Capodanno, CardioThoracic-Vascular and Transplant Department, A.O.U. ‘Policlinico-Vittorio Emanuele’, University of Catania, Catania, Italy; Thomas Cuisset, Cardiology, CHU Timone, Marseille, France; Christi Deaton, Public Health and Primary Care, University of Cambridge School of Clinical Medicine, Cambridge, United Kingdom; Kenneth Dickstein, Cardiology, Stavanger University Hospital, University of Bergen, Stavanger, Norway; Thor Edvardsen, Cardiology, Oslo University Hospital, Oslo, Norway; Javier Escaned, Interventional Cardiology Unit, Hospital Clinico San Carlos, Madrid, Spain; Christian Funck-Brentano, Department of Clinical Pharmacology, Sorbonne Université, AP-HP, ICAN and INSERM CIC Paris-Est, Paris, France; Bernard J. Gersh, Department of Cardiovascular Medicine, Mayo Clinic, Rochester, MN, United States of America; Martine Gilard, Cardiology, Brest University, Brest, France; David Hasdai, Cardiology, Rabin Medical Center Petah Tikva, Israel; Robert Hatala, Department of Cardiology and Angiology, Slovak Cardiovascular Institute, Slovak Medical University, Bratislava, Slovakia; Felix Mahfoud, Internal Medicine III, Saarland University, Homburg, Germany; Josep Masip, Cardiology Department /Intensive Care Department, Hospital CIMA-Sanitas/Consorci Sanitari Integral/University of Barcelona, Barcelona, Spain; Claudio Muneretto, Cardiovascular Surgery, University of Brescia Medical School, Brescia, Italy; Eva Prescott, Department of Cardiology, Bispebjerg University Hospital, Copenhagen, Denmark; Antti Saraste, Heart Center, Turku University Hospital, Turku, Finland; Robert F. Storey, Department of Infection, Immunity and Cardiovascular Disease, University of Sheffield, Sheffield, United Kingdom; Pavel Svitil, Cardiologic Practice, Practice of General Cardiology, Jihlava, Czech Republic; Marco Valgimigli, Inselspital, University of Bern, Bern, Switzerland.
ESC Committee for Practice Guidelines (CPG): Stephan Windecker (Chairperson) (Switzerland), Victor Aboyans (France), Colin Baigent (United Kingdom), Jean-Philippe Collet (France), Veronica Dean (France), Victoria Delgado (Netherlands), Donna Fitzsimons (United Kingdom), Christopher P. Gale (United Kingdom), Diederick E. Grobbee (Netherlands), Sigrun Halvorsen (Norway), Gerhard Hindricks (Germany), Bernard Iung (France), Peter Jüni (Canada), Hugo A. Katus (Germany), Ulf Landmesser (Germany), Christophe Leclercq (France), Maddalena Lettino (Italy), Basil S. Lewis (Israel), Bela Merkely (Hungary), Christian Mueller (Switzerland), Steffen Petersen (United Kingdom), Anna Sonia Petronio (Italy), Dimitrios J. Richter (Greece), Marco Roffi (Switzerland), Evgeny Shlyakhto (Russian Federation), Iain A. Simpson (United Kingdom), Miguel Sousa-Uva (Portugal), Rhian M. Touyz (United Kingdom).
ESC National Cardiac Societies actively involved in the review process of the 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes.
Algeria: Algerian Society of Cardiology, Salim Benkhedda; Austria: Austrian Society of Cardiology, Bernhard Metzler; Belarus: Belorussian Scientific Society of Cardiologists, Volha Sujayeva; Belgium: Belgian Society of Cardiology, Bernard Cosyns; Bosnia and Herzegovina: Association of Cardiologists of Bosnia and Herzegovina, Zumreta Kusljugic; Bulgaria: Bulgarian Society of Cardiology, Vasil Velchev; Cyprus: Cyprus Society of Cardiology, Georgios Panayi; Czech Republic: Czech Society of Cardiology, Petr Kala; Denmark: Danish Society of Cardiology, Sune Ammentorp Haahr-Pedersen; Egypt: Egyptian Society of Cardiology, Hamza Kabil; Estonia: Estonian Society of Cardiology, Tiia Ainla; Finland: Finnish Cardiac Society, Tomi Kaukonen; France: French Society of Cardiology, Guillaume Cayla; Georgia: Georgian Society of Cardiology, Zurab Pagava; Germany: German Cardiac Society, Jochen Woehrle; Greece: Hellenic Society of Cardiology, John Kanakakis; Hungary: Hungarian Society of Cardiology, Kálmán Tóth; Iceland: Icelandic Society of Cardiology, Thorarinn Gudnason; Ireland: Irish Cardiac Society, Aaron Peace; Israel: Israel Heart Society, Doron Aronson; Italy: Italian Federation of Cardiology, Carmine Riccio; Kosovo (Republic of): Kosovo Society of Cardiology, Shpend Elezi; Kyrgyzstan: Kyrgyz Society of Cardiology, Erkin Mirrakhimov; Latvia: Latvian Society of Cardiology, Silvija Hansone; Lebanon: Lebanese Society of Cardiology, Antoine Sarkis; Lithuania: Lithuanian Society of Cardiology, Ruta Babarskiene; Luxembourg: Luxembourg Society of Cardiology, Jean Beissel; Malta: Maltese Cardiac Society, Andrew J. Cassar Maempel; Moldova (Republic of): Moldavian Society of Cardiology, Valeriu Revenco; Netherlands: Netherlands Society of Cardiology, G.J. de Grooth; North Macedonia: Macedonian FYR Society of Cardiology, Hristo Pejkov; Norway: Norwegian Society of Cardiology, Vibeke Juliebø; Poland: Polish Cardiac Society, Piotr Lipiec; Portugal: Portuguese Society of Cardiology, José Santos; Romania: Romanian Society of Cardiology, Ovidiu Chioncel; Russian Federation: Russian Society of Cardiology, Dmitry Duplyakov; San Marino: San Marino Society of Cardiology, Luca Bertelli; Serbia: Cardiology Society of Serbia, Ana Djordjevic Dikic; Slovakia: Slovak Society of Cardiology, Martin Studenčan; Slovenia: Slovenian Society of Cardiology, Matjaz Bunc; Spain: Spanish Society of Cardiology, Fernando Alfonso; Sweden: Swedish Society of Cardiology, Magnus Bäck; Switzerland: Swiss Society of Cardiology, Michael Zellweger; Tunisia: Tunisian Society of Cardiology and Cardio-Vascular Surgery, Faouzi Addad; Turkey: Turkish Society of Cardiology, Aylin Yildirir; Ukraine: Ukrainian Association of Cardiology, Yuriy Sirenko; United Kingdom of Great Britain and Northern Ireland: British Cardiovascular Society, Brian Clapp.
The disclosure forms of all experts involved in the development of these Guidelines are available on the ESC website www.escardio.org/guidelines
Author/Task Force Member Affiliations: listed in the Appendix.
ESC Committee for Practice Guidelines (CPG) and National Cardiac Societies document reviewers: listed in the Appendix.
ESC entities having participated in the development of this document:
Associations: Acute Cardiovascular Care Association (ACCA), Association of Cardiovascular Nursing & Allied Professions (ACNAP), European Association of Cardiovascular Imaging (EACVI), European Association of Preventive Cardiology (EAPC), European Association of Percutaneous Cardiovascular Interventions (EAPCI), European Heart Rhythm Association (EHRA), Heart Failure Association (HFA).
Councils: Council for Cardiology Practice.
Working Groups: Atherosclerosis and Vascular Biology, Cardiovascular Pharmacotherapy, Cardiovascular Surgery, Coronary Pathophysiology and Microcirculation, Thrombosis.
The content of these ESC Guidelines has been published for personal and educational use only. No commercial use is authorized. No part of the ESC Guidelines may be translated or reproduced in any form without written permission from the ESC. Permission can be obtained upon submission of a written request to Oxford University Press, the publisher of the European Heart Journal and the party authorized to handle such permissions on behalf of the ESC (journals.permissions@oxfordjournals.org).
Disclaimer. The ESC Guidelines represent the views of the ESC and were produced after careful consideration of the scientific and medical knowledge, and the evidence available at the time of their publication. The ESC is not responsible in the event of any contradiction, discrepancy, and/or ambiguity between the ESC Guidelines and any other official recommendations or guidelines issued by the relevant public health authorities, in particular in relation to good use of healthcare or therapeutic strategies. Health professionals are encouraged to take the ESC Guidelines fully into account when exercising their clinical judgment, as well as in the determination and the implementation of preventive, diagnostic, or therapeutic medical strategies; however, the ESC Guidelines do not override, in any way whatsoever, the individual responsibility of health professionals to make appropriate and accurate decisions in consideration of each patient’s health condition and in consultation with that patient and, where appropriate and/or necessary, the patient’s caregiver. Nor do the ESC Guidelines exempt health professionals from taking into full and careful consideration the relevant official updated recommendations or guidelines issued by the competent public health authorities, in order to manage each patient’s case in light of the scientifically accepted data pursuant to their respective ethical and professional obligations. It is also the health professional’s responsibility to verify the applicable rules and regulations relating to drugs and medical devices at the time of prescription.
14 References
SCOT-HEART investigators.
SCOT-HEART investigators,
2019 ESC/EASD Guidelines on diabetes, pre-diabetes, and cardiovascular diseases. Eur Hear J
Cholesterol Treatment Trialists’ (CTT) Collaboration,
GBD 2016 Alcohol Collaborators.
National Institute for Health and Care Excellence (NICE). Stable angina: management. Clinical guideline [CG126]. https://www.nice.org.uk/guidance/cg126 (
European Medicines Agency. European Medicines Agency recommends measures to reduce risk of heart problems with Corlentor/Procoralan (ivabradine). https://www.ema.europa.eu/en/news/european-medicines-agency-recommends-measures-reduce-risk-heart-problems-corlentorprocoralan (28 March 2019).
Medicines & Healthcare products Regulatory Agency (MHRA). Public Assessment Report. UKPAR. Nicorandil 10 mg Tablets. Nicorandil 20 mg Tablets. http://www.mhra.gov.uk/home/groups/par/documents/websiteresources/con786861.pdf (28 March 2019).
IONA Study Group.
European Medicines Agency. Questions and answers on the review of medicines containing trimetazidine (20 mg tablets, 35 mg modified release tablet and 20 mg/ml oral solution). https://www.ema.europa.eu/en/documents/referral/questions-answers-review-medicines-containing-trimetazidine-20-mg-tablets-35-mg-modified-release/ml-oral-solution_en.pdf (28 March 2019).
European Medicines Agency. Assessment Report for trimetazidine containing medicinal products. https://www.ema.europa.eu/en/documents/referral/trimetazidine-article-31-referral-assessment-report_en.pdf (28 March 2019).
CURRENT-OASIS 7 Investigators,
Antithrombotic Trialists' Collaboration.
Antithrombotic Trialists' (ATT) Collaboration,
Coxib and traditional NSAID Trialists' (CNT) Collaboration,
CAPRIE Steering Committee.
Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, Chapman MJ, De Backer GG, Delgado V, Ference BA, Graham IM, Halliday A, Landmesser U, Mihaylova B, Pedersen TR, Riccardi G, Richter DJ, Sabatine MS, Taskinen M-R, Tokgozoglu L, Wiklund O. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 2020;41:111–188.
SOLVD Investigators,
Cholesterol Treatment Trialists’ (CTT) Collaboration,
CIBIS-II Investigators and Committees.
SOLVD Investigators,
Antiarrhythmics versus Implantable Defibrillators (AVID) Investigators.
JCS Joint Working Group.
Scientific Steering Committee on behalf of the Simon Broome Register Group.
Ankle Brachial Index Collaboration,
ONTARGET Investigators,
Emerging Risk Factors Collaboration,
ADVANCE Collaborative Group,





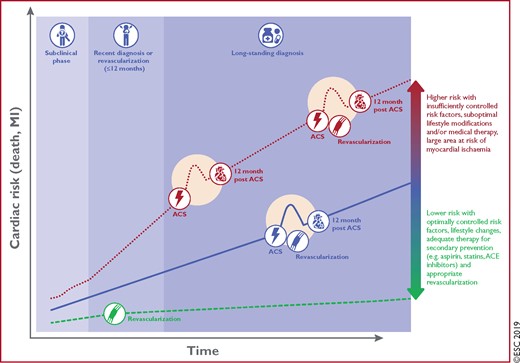
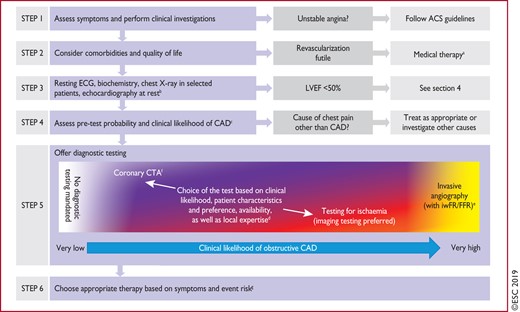
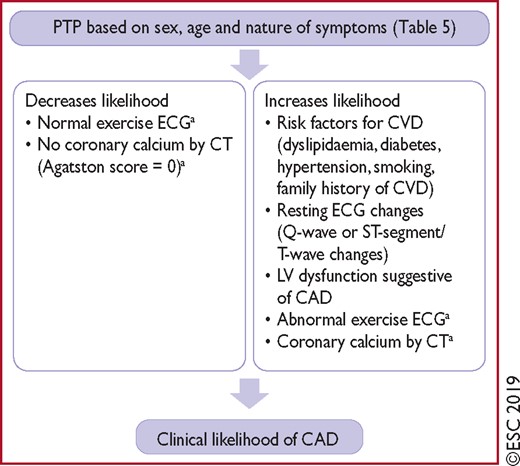
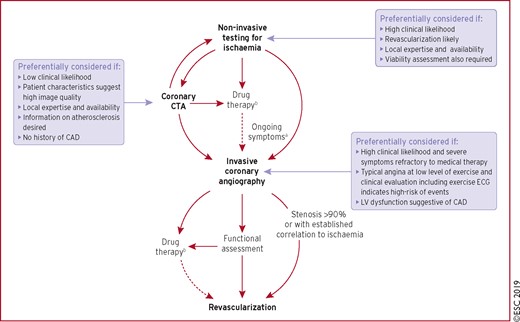
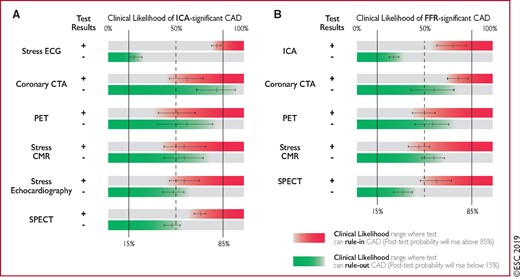
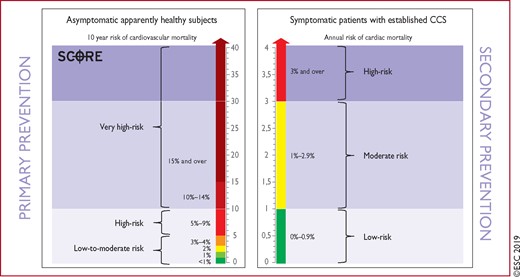
![Suggested stepwise strategy for long term anti-ischaemic drug therapy in patients with chronic coronary syndromes and specific baseline characteristics. The proposed stepwise approach must be adapted to each patient's characteristics and preferences. Given the limited evidence on various combinations of drugs in different clinical conditions, the proposed options are only indicative of potential combinations and do not represent formal recommendations. BB = beta-blocker; bpm = beats per minute; CCB = [any class of] calcium channel blocker; DHP-CCB = dihydropyridine calcium channel blocker; HF = heart failure; LAN = long-acting nitrate; LV = left ventricular; non-DHP-CCB = non-dihydropyridine calcium channel blocker. aCombination of a BB with a DHP-CCB should be considered as first step; combination of a BB or a CCB with a second-line drug may be considered as a first step; bThe combination of a BB and non-DHP-CCB should initially use low doses of each drug under close monitoring of tolerance, particularly heart rate and blood pressure; cLow-dose BB or low-dose non-DHP-CCB should be used under close monitoring of tolerance, particularly heart rate and blood pressure; dIvabradine should not be combined with non-DHP-CCB; eConsider adding the drug chosen at step 2 to the drug tested at step 1 if blood pressure remains unchanged.](https://oup.silverchair-cdn.com/oup/backfile/Content_public/Journal/eurheartj/41/3/10.1093_eurheartj_ehz425/2/m_ehz425f8.jpeg?Expires=1716876022&Signature=kAC0ka-ExqK2vNSdXGtsxlbvGNMwprUqvHC6ZY98E8UVN4lk3-XADO6oHGeydMsGj12fvmGMXKqyqcrvD6kw8uECtRh0kwqA5lZlKgKtrOLtYmFwy2ONnSvrkUgft8NfdADocGlxtSL8aYwuN47Q9aNajz0PVzHPaHnNW2gsB09TphUJQpO7p07gn~FRLSt3UZOw7~rIa4jD4xUzKHgq1m-6LjdqW~7tK0VkrqUsmfze7HIp4i-roTfM5vD7N7RP0w1jBi7vTV3bWDT60TMx236wFJiHP31zUARokKdZyWqC3MZUJt5MIfXUzoP3n-Cy1J9dl794dxYfpP8LpL~Izw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)