-
PDF
- Split View
-
Views
-
Cite
Cite
Stelios Katsanevakis, Christos D. Maravelias, Laurie T. Kell, Landings profiles and potential métiers in Greek set longliners, ICES Journal of Marine Science, Volume 67, Issue 4, May 2010, Pages 646–656, https://doi.org/10.1093/icesjms/fsp279
Close - Share Icon Share
Abstract
A very large number (>14 000) of generally small vessels operate as longliners in Greek seas. The aim of this study was to identify potential set longline métiers, based on a large sample of landings records from all over Greece. Landings data from set longliners between 2002 and 2006, collected from several ports in the Aegean and East Ionian Sea, were used. The landings profiles were grouped using a two-step procedure, the first involving factorial analysis of the log-transformed landing profiles, and the second a classification of the factorial coordinates (hierarchical agglomerative clustering). In all, 13 métiers were identified in the Aegean Sea and 7 in the Ionian Sea. The most important métiers identified were those targeting white sea bream (Diplodus sargus), hake (Merluccius merluccius), common sea bream (Pagrus pagrus), and common pandora (Pagellus erythrinus), and mixed métiers. Varying spatial (within the Aegean and Ionian Seas) and seasonal patterns were evident for the métiers identified, indicating that fisher motivation to engage in a specific métier varies both spatially and temporally.Katsanevakis, S., Maravelias, C. D., and Kell, L. T. 2010. Landings profiles and potential métiers in Greek set longliners. – ICES Journal of Marine Science, 67: 646–656.
Introduction
A fleet of some 17 920 vessels operates in Greek seas (based on 2007 data), representing some 45% of the total number of EU fishing vessels in the Mediterranean Sea. Of these, 14 347 (of which 10 882 are registered in Aegean ports and 3465 in the Ionian Sea) are licensed to fish with set longlines. Most of these vessels (97.1%) are <12 m, and most of them (85.1%) are also licensed to fish with trammelnets or gillnets. Vessels operating as longliners have an average length of 6.8 m, a gross registered tonnage of 2.3, an engine power of 20.1 kW, and a mean age of 24.6 years (IMAS–FISH, 2008). In many areas, especially on the smaller islands, small-scale fisheries (which include the longline fisheries) have significant socio-economic value, because they offer job opportunities and vital support to local economies. Their catches represent 47% of Greek fisheries production and 54% of the market income (Tzanatos et al., 2005).
Longliners target a wide variety of species in multispecies fisheries, and there is extensive interaction with other gear and fleet segments, because many of the target species of longliners are also targeted by trawlers and seiners (Stergiou and Erzini, 2002; Tzanatos et al., 2005). In such multispecies, multifleet fisheries, where more than one species is caught in the same area and different fleets exploit the same stocks, conventional single-species fisheries management has long been recognized as problematic (Pelletier and Ferraris, 2000). As species are not exploited independently, management of one stock influences the management of all other target (and non-target) stocks, so to give management advice in mixed fisheries, it is more practical and effective if fleet- or fishery-based approaches are used instead.
However, to provide the necessary multispecies, multi-fisheries advice, fisheries scientists have to understand the behaviour of fishers and assess the flexibility of fishing practices and the response of fisheries to management. This may vary depending on market conditions, season, management regulations, and skipper's empirical knowledge (Hilborn and Ledbetter, 1985; Pelletier and Ferraris, 2000; Marchal et al., 2006). Each fishing practice is likely to impact exploited stocks in a particular manner, and to assess the relationship between total fishing effort of the fleet and the resulting fishing mortalities of the exploited stocks, a separate evaluation for each fishing practice is necessary (Pelletier and Ferraris, 2000). A first step in a fishery as diverse as the Greek longliners is to define the fishing practices of each fleet segment by reducing the description of the variety of fishing trips to a single categorical variable that summarizes its main characteristics, i.e. the gear used, the fishing ground, and the target species (Pelletier and Ferraris, 2000; Ulrich and Andersen, 2004). This has been referred to in the literature by a wide variety of terms, such as métier, fishery, directed fishery, fishery management unit, fishing trip type, fishing strategy, or fishing tactic (Pelletier and Ferraris, 2000, and references therein; Pech et al., 2001; Ulrich et al., 2001; Silva et al., 2002; Maynou et al., 2003; Jiménez et al., 2004; Ulrich and Andersen, 2004). The term métier is used here.
A first attempt at identifying métiers for small-scale fisheries (including longliners), based on questionnaires, was carried out by Tzanatos et al. (2005) in 9 of the 40 coastal prefectures of Greece. Tzanatos et al. (2006) also identified the main small-scale métiers of the Patraikos Gulf, using a sample of landings data in five ports. However, despite the importance of small-scale fisheries to Greece, the identification of métiers at a national level is lacking. The aim of this study was to group landings profiles and to identify potential set longline métiers, based on a large sample of landings from all over Greece. The identification of métiers is important in mixed fisheries management, to understand the spatio-temporal patterns of fishing allocation and the response of fishers to management, and to improve the design of stratified data collection to achieve better performance in the estimates of species-specific production.
Material and methods
Study area
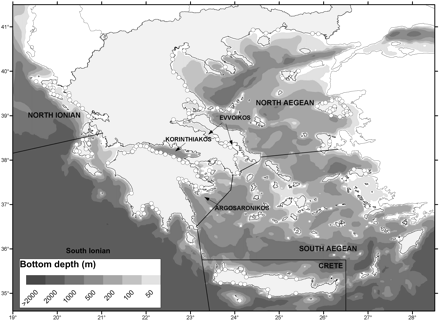
The study area included all Greek territorial coastal waters, i.e. most of the Aegean Sea (GFSM 37.3.1, GSAs 22 and 23) and the eastern Ionian Sea (GFCM 37.2.2, GSA 20; Figure 1). The seabed of Greek seas displays a complex geomorphology, reflecting a complexity of geological and geodynamic processes (Sakellariou and Alexandri, 2007). The Aegean Sea has a long coastline (∼16 000 km), a complex bathymetry, a generally narrow continental shelf (except the northern Aegean and the Kyklades Plateau), and many small islands (∼2000). The Greek part of the Ionian Sea is characterized by a narrower continental shelf than in the Aegean Sea, and the existence of the deep Hellenic Trench, lying along the western and southwestern Hellenic coast and the islands of the Cretan Arc. Both Aegean and Ionian offshore waters are oligotrophic, whereas most coastal areas are mesotrophic (Gotsis-Skretas and Ignatiades, 2005; Siokou-Frangou et al., 2005). The Aegean Sea may be subdivided into five subareas (North Aegean, South Aegean, Evoikos Gulf, Argosaronikos Gulf, and Crete), and the Ionian Sea into three (Central–South Ionian, North Ionian, and Korinthiakos Gulf), based on their distinctive geomorphology (Figure 1).
Map of Greece, with the main areas mentioned in text. The sampling ports are shown as open circles.
Data
According to EU legislation, logbooks in the Mediterranean are not compulsory for vessels of <10 m total length (EC, 1993) or for landed net weight of fish <15 kg per species (EC, 2006). For Greek set longliners, most vessels are <10 m (93.5%) and the landed net weight of fish is usually <15 kg per species, so usually there is no record of landings in logbooks. Moreover, because of the very large number of small vessels and landing ports, complete recording of landings from small-scale fisheries is impractical. Therefore, contrary to the data-rich demersal fisheries of much of the ICES Area, the Mediterranean has a shortage of landings data, and assessment of fisheries is based on a small sample of total landings. Under the Data Collection Regulation framework (EC, 2000, 2001), data on effort and landings have been collected in Greece since 2002, from 30 major sites including 209 landing ports (Figure 1). From each site, species-specific landings data were gathered by local correspondents (mainly Prefecture Fisheries Inspectors) monthly, according to a systematic sampling procedure (details are given in Bazigos and Kavadas, 2007). Specifically, local correspondents visit a predefined number of landings ports in their site of responsibility every month and collect landings data from the vessels arriving. From this dataset (2002–2006), records of set longliner landings were used to identify landings profiles and potential métiers in the Aegean and Ionian Seas.
Only fishing trips with non-zero landings were considered, i.e. 4390 trips for the Aegean Sea and 1386 trips for the Ionian Sea. Rare species, i.e. caught in <0.5% of the trips, were excluded from the analysis.
Identification and description of fisheries
Separate analyses were conducted for the Aegean and the Ionian Seas. First, a data matrix A with fishing trips as individuals (n rows) and landings per species as variables (p columns) was constructed for each area. For each trip, the absolute weight of the landings was transformed into a landings profile, i.e. a relative species composition, by dividing the weight of the landings per species by the total weight of the landings derived from their fishing trip. This removed the differences in the level of the landings, which are often linked to factors such as total effort, time of year, and weather conditions. Values of landings were not used rather than weight because values are not collected on a trip-by-trip basis. Using average annual or seasonal values would not be a good idea either, because values of the same species vary temporally and spatially.
Data were then log-transformed to render their distribution symmetrical. A modification of the multivariate approach proposed by Pelletier and Ferraris (2000) was used to identify potential métiers. The first step involved factorial analysis of the log-transformed landings profiles, and the second step was a classification of the factorial coordinates. Specifically, a non-normalized principal components analysis based on the covariance matrix was performed to produce a convenient lower dimensional summary of the original variables which accounts for a substantial proportion of the total variation in the initial data. The correlation matrix is useful, if variables in the analysis are measured at different scales. In our case, the variables are the log-transformed relative species composition, so they all have the same scale. We also made trials with correlation matrices, but the dendrograms produced were improved when based on covariance matrices.
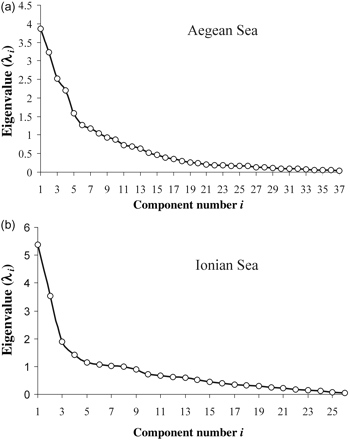
The number of principal components selected was based on a scree diagram, which is a plot of the eigenvalues (λi) of the covariance matrix against the rank (i) of the eigenvalues. The number of components selected was the value of i corresponding to where large eigenvalues (i.e. accounting for a large proportion of the total variation of the original data) cease and small eigenvalues begin (Everitt, 2005).
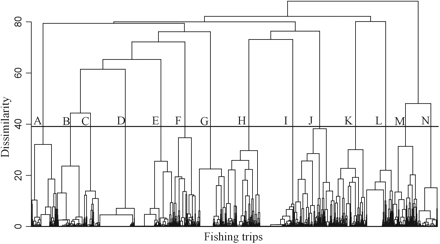
A hierarchical agglomerative cluster (HAC) analysis, based on Euclidean distances and applying Ward's minimum variance criterion (Ward, 1963), was carried out using the retained principal components. The HAC analysis of the fishing trips led to homogenous groups (clusters) being identified, representing different landings profiles. The choice of the number of clusters was based on expert knowledge and on several trials with different choices of dissimilarity threshold in the resulting dendrogram. Each cluster was considered to represent a potential métier.
Results
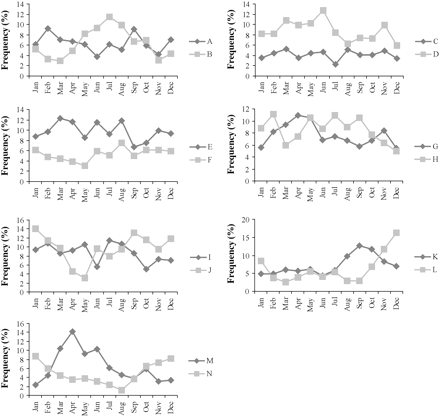
A total of 69 species was recorded in the Aegean Sea samples, of which 37 were retained in the analysis after removing rare species. For the fishing trips in the Aegean Sea, 11 principal components were retained based on the scree diagram and on the contribution of each component to the total variance (Figure 2a). These 11 components accounted for 77% of the total variation in the original data. The HAC analysis of the fishing trips based on these 11 principal components led to the identification of 14 clusters (A–N), representing different landings profiles (Figure 3). The 14 clusters had different average landing profiles (Table 1) and different geographical distribution among the five main subareas of the Aegean Sea (Table 2). Additionally, there was a temporal pattern in the frequency of most landings profiles and a succession of landings profiles throughout the year (Figure 4).
Scree diagrams for the landings profiles data matrices for set longline fishing trips in (a) the Aegean Sea and (b) the Ionian Sea.
Dendrogram of set longline fishing trips in the Aegean Sea, based on log-transformed landings profiles. In all, 14 clusters (A–N) were identified, representing different landings profiles.
Average landings profiles of the 14 clusters identified in the Aegean Sea (Figure 3), given as a proportion (%) of the landings of each species to the total landings of each cluster.
| . | Landings profile . | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxon . | A . | B . | C . | D . | E . | F . | G . | H . | I . | J . | K . | L . | M . | N . | Total . |
| Pagrus pagrus | 9.9 | 0.1 | 4.9 | 93.4 | 12.4 | 13.2 | 13.0 | 8.7 | 0.0 | 24.9 | 13.3 | 2.3 | 16.8 | 0.1 | 17.1 |
| Dilplodus sargus | 2.9 | 0.1 | 0.0 | 0.0 | 72.8 | 8.2 | 5.2 | 14.0 | 0.0 | 11.8 | 14.7 | 8.1 | 2.7 | 0.0 | 12.2 |
| Merluccius merluccius | 0.1 | 0.0 | 3.5 | 0.3 | 0.0 | 0.3 | 0.0 | 2.1 | 84.3 | 0.0 | 1.0 | 0.1 | 0.5 | 65.2 | 10.9 |
| Dentex dentex | 17.0 | 0.0 | 0.4 | 0.0 | 0.0 | 6.2 | 68.4 | 0.6 | 0.4 | 0.0 | 3.2 | 1.6 | 1.1 | 0.0 | 7.0 |
| Pagellus erythrinus | 0.7 | 0.0 | 0.3 | 0.0 | 0.0 | 0.4 | 0.3 | 50.7 | 0.0 | 12.5 | 0.0 | 1.6 | 16.5 | 0.0 | 6.7 |
| Sparus auratus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 5.7 | 0.0 | 0.6 | 5.8 | 72.5 | 0.0 | 0.0 | 5.8 |
| Epinephelus marginatus | 62.6 | 0.0 | 0.0 | 0.0 | 0.0 | 14.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.6 | 0.2 | 0.7 | 0.0 | 4.9 |
| Spondyliosoma cantharus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.5 | 0.1 | 2.6 | 0.0 | 2.5 | 41.5 | 0.1 | 0.0 | 1.5 | 3.8 |
| Serranus cabrilla | 0.2 | 0.0 | 0.4 | 0.1 | 0.0 | 0.0 | 0.8 | 1.3 | 0.0 | 22.0 | 0.2 | 2.8 | 17.5 | 0.0 | 3.6 |
| Scorpaena scrofa | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.4 | 0.1 | 0.7 | 30.3 | 28.3 | 3.4 |
| Epinephelus costae | 0.0 | 0.6 | 0.0 | 0.0 | 0.2 | 47.7 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.3 | 0.7 | 0.0 | 2.8 |
| Dicentrarchus labrax | 0.0 | 36.9 | 0.3 | 0.0 | 0.1 | 0.0 | 0.1 | 0.1 | 0.2 | 0.0 | 0.1 | 3.6 | 0.0 | 0.1 | 2.7 |
| Dentex macrophthalmus | 0.8 | 0.2 | 37.1 | 0.0 | 1.8 | 0.1 | 0.1 | 4.7 | 0.0 | 0.2 | 1.0 | 0.2 | 4.2 | 0.0 | 2.5 |
| Oblada melanura | 0.1 | 0.0 | 29.4 | 0.0 | 3.8 | 0.0 | 0.3 | 1.3 | 0.0 | 0.0 | 7.1 | 1.0 | 1.1 | 0.1 | 2.4 |
| Conger conger | 0.2 | 2.6 | 0.0 | 0.0 | 0.0 | 0.0 | 0.8 | 0.1 | 0.5 | 16.4 | 1.2 | 0.0 | 2.5 | 0.0 | 2.2 |
| Epinephelus aeneus | 2.5 | 8.6 | 1.1 | 1.2 | 1.0 | 2.6 | 4.7 | 0.4 | 0.4 | 0.0 | 0.7 | 0.0 | 0.2 | 0.1 | 1.6 |
| Sharks | 0.7 | 7.7 | 1.6 | 0.4 | 0.2 | 0.2 | 0.6 | 0.3 | 3.5 | 0.1 | 0.4 | 0.0 | 0.9 | 1.4 | 1.2 |
| Rays | 0.5 | 7.1 | 0.4 | 0.0 | 0.1 | 1.0 | 1.3 | 0.2 | 2.4 | 1.0 | 0.9 | 0.1 | 0.1 | 0.4 | 1.1 |
| Mullus barbatus | 0.0 | 4.3 | 6.9 | 0.1 | 0.1 | 0.0 | 0.2 | 1.4 | 0.5 | 0.0 | 1.4 | 0.0 | 0.6 | 0.0 | 0.9 |
| Lithognathus mormyrus | 0.0 | 6.1 | 0.5 | 0.2 | 0.8 | 0.1 | 0.0 | 1.0 | 0.0 | 0.2 | 0.0 | 1.4 | 0.0 | 0.0 | 0.7 |
| Pomatomus saltatrix | 0.0 | 7.2 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 | 0.5 |
| Other species | 1.8 | 18.4 | 13.2 | 4.1 | 6.6 | 4.5 | 4.2 | 4.5 | 7.7 | 7.2 | 5.8 | 3.3 | 3.6 | 2.8 | 6.2 |
| . | Landings profile . | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxon . | A . | B . | C . | D . | E . | F . | G . | H . | I . | J . | K . | L . | M . | N . | Total . |
| Pagrus pagrus | 9.9 | 0.1 | 4.9 | 93.4 | 12.4 | 13.2 | 13.0 | 8.7 | 0.0 | 24.9 | 13.3 | 2.3 | 16.8 | 0.1 | 17.1 |
| Dilplodus sargus | 2.9 | 0.1 | 0.0 | 0.0 | 72.8 | 8.2 | 5.2 | 14.0 | 0.0 | 11.8 | 14.7 | 8.1 | 2.7 | 0.0 | 12.2 |
| Merluccius merluccius | 0.1 | 0.0 | 3.5 | 0.3 | 0.0 | 0.3 | 0.0 | 2.1 | 84.3 | 0.0 | 1.0 | 0.1 | 0.5 | 65.2 | 10.9 |
| Dentex dentex | 17.0 | 0.0 | 0.4 | 0.0 | 0.0 | 6.2 | 68.4 | 0.6 | 0.4 | 0.0 | 3.2 | 1.6 | 1.1 | 0.0 | 7.0 |
| Pagellus erythrinus | 0.7 | 0.0 | 0.3 | 0.0 | 0.0 | 0.4 | 0.3 | 50.7 | 0.0 | 12.5 | 0.0 | 1.6 | 16.5 | 0.0 | 6.7 |
| Sparus auratus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 5.7 | 0.0 | 0.6 | 5.8 | 72.5 | 0.0 | 0.0 | 5.8 |
| Epinephelus marginatus | 62.6 | 0.0 | 0.0 | 0.0 | 0.0 | 14.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.6 | 0.2 | 0.7 | 0.0 | 4.9 |
| Spondyliosoma cantharus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.5 | 0.1 | 2.6 | 0.0 | 2.5 | 41.5 | 0.1 | 0.0 | 1.5 | 3.8 |
| Serranus cabrilla | 0.2 | 0.0 | 0.4 | 0.1 | 0.0 | 0.0 | 0.8 | 1.3 | 0.0 | 22.0 | 0.2 | 2.8 | 17.5 | 0.0 | 3.6 |
| Scorpaena scrofa | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.4 | 0.1 | 0.7 | 30.3 | 28.3 | 3.4 |
| Epinephelus costae | 0.0 | 0.6 | 0.0 | 0.0 | 0.2 | 47.7 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.3 | 0.7 | 0.0 | 2.8 |
| Dicentrarchus labrax | 0.0 | 36.9 | 0.3 | 0.0 | 0.1 | 0.0 | 0.1 | 0.1 | 0.2 | 0.0 | 0.1 | 3.6 | 0.0 | 0.1 | 2.7 |
| Dentex macrophthalmus | 0.8 | 0.2 | 37.1 | 0.0 | 1.8 | 0.1 | 0.1 | 4.7 | 0.0 | 0.2 | 1.0 | 0.2 | 4.2 | 0.0 | 2.5 |
| Oblada melanura | 0.1 | 0.0 | 29.4 | 0.0 | 3.8 | 0.0 | 0.3 | 1.3 | 0.0 | 0.0 | 7.1 | 1.0 | 1.1 | 0.1 | 2.4 |
| Conger conger | 0.2 | 2.6 | 0.0 | 0.0 | 0.0 | 0.0 | 0.8 | 0.1 | 0.5 | 16.4 | 1.2 | 0.0 | 2.5 | 0.0 | 2.2 |
| Epinephelus aeneus | 2.5 | 8.6 | 1.1 | 1.2 | 1.0 | 2.6 | 4.7 | 0.4 | 0.4 | 0.0 | 0.7 | 0.0 | 0.2 | 0.1 | 1.6 |
| Sharks | 0.7 | 7.7 | 1.6 | 0.4 | 0.2 | 0.2 | 0.6 | 0.3 | 3.5 | 0.1 | 0.4 | 0.0 | 0.9 | 1.4 | 1.2 |
| Rays | 0.5 | 7.1 | 0.4 | 0.0 | 0.1 | 1.0 | 1.3 | 0.2 | 2.4 | 1.0 | 0.9 | 0.1 | 0.1 | 0.4 | 1.1 |
| Mullus barbatus | 0.0 | 4.3 | 6.9 | 0.1 | 0.1 | 0.0 | 0.2 | 1.4 | 0.5 | 0.0 | 1.4 | 0.0 | 0.6 | 0.0 | 0.9 |
| Lithognathus mormyrus | 0.0 | 6.1 | 0.5 | 0.2 | 0.8 | 0.1 | 0.0 | 1.0 | 0.0 | 0.2 | 0.0 | 1.4 | 0.0 | 0.0 | 0.7 |
| Pomatomus saltatrix | 0.0 | 7.2 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 | 0.5 |
| Other species | 1.8 | 18.4 | 13.2 | 4.1 | 6.6 | 4.5 | 4.2 | 4.5 | 7.7 | 7.2 | 5.8 | 3.3 | 3.6 | 2.8 | 6.2 |
The most important species of each profile (with a proportion in landings >10%) are shown emboldened.
Average landings profiles of the 14 clusters identified in the Aegean Sea (Figure 3), given as a proportion (%) of the landings of each species to the total landings of each cluster.
| . | Landings profile . | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxon . | A . | B . | C . | D . | E . | F . | G . | H . | I . | J . | K . | L . | M . | N . | Total . |
| Pagrus pagrus | 9.9 | 0.1 | 4.9 | 93.4 | 12.4 | 13.2 | 13.0 | 8.7 | 0.0 | 24.9 | 13.3 | 2.3 | 16.8 | 0.1 | 17.1 |
| Dilplodus sargus | 2.9 | 0.1 | 0.0 | 0.0 | 72.8 | 8.2 | 5.2 | 14.0 | 0.0 | 11.8 | 14.7 | 8.1 | 2.7 | 0.0 | 12.2 |
| Merluccius merluccius | 0.1 | 0.0 | 3.5 | 0.3 | 0.0 | 0.3 | 0.0 | 2.1 | 84.3 | 0.0 | 1.0 | 0.1 | 0.5 | 65.2 | 10.9 |
| Dentex dentex | 17.0 | 0.0 | 0.4 | 0.0 | 0.0 | 6.2 | 68.4 | 0.6 | 0.4 | 0.0 | 3.2 | 1.6 | 1.1 | 0.0 | 7.0 |
| Pagellus erythrinus | 0.7 | 0.0 | 0.3 | 0.0 | 0.0 | 0.4 | 0.3 | 50.7 | 0.0 | 12.5 | 0.0 | 1.6 | 16.5 | 0.0 | 6.7 |
| Sparus auratus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 5.7 | 0.0 | 0.6 | 5.8 | 72.5 | 0.0 | 0.0 | 5.8 |
| Epinephelus marginatus | 62.6 | 0.0 | 0.0 | 0.0 | 0.0 | 14.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.6 | 0.2 | 0.7 | 0.0 | 4.9 |
| Spondyliosoma cantharus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.5 | 0.1 | 2.6 | 0.0 | 2.5 | 41.5 | 0.1 | 0.0 | 1.5 | 3.8 |
| Serranus cabrilla | 0.2 | 0.0 | 0.4 | 0.1 | 0.0 | 0.0 | 0.8 | 1.3 | 0.0 | 22.0 | 0.2 | 2.8 | 17.5 | 0.0 | 3.6 |
| Scorpaena scrofa | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.4 | 0.1 | 0.7 | 30.3 | 28.3 | 3.4 |
| Epinephelus costae | 0.0 | 0.6 | 0.0 | 0.0 | 0.2 | 47.7 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.3 | 0.7 | 0.0 | 2.8 |
| Dicentrarchus labrax | 0.0 | 36.9 | 0.3 | 0.0 | 0.1 | 0.0 | 0.1 | 0.1 | 0.2 | 0.0 | 0.1 | 3.6 | 0.0 | 0.1 | 2.7 |
| Dentex macrophthalmus | 0.8 | 0.2 | 37.1 | 0.0 | 1.8 | 0.1 | 0.1 | 4.7 | 0.0 | 0.2 | 1.0 | 0.2 | 4.2 | 0.0 | 2.5 |
| Oblada melanura | 0.1 | 0.0 | 29.4 | 0.0 | 3.8 | 0.0 | 0.3 | 1.3 | 0.0 | 0.0 | 7.1 | 1.0 | 1.1 | 0.1 | 2.4 |
| Conger conger | 0.2 | 2.6 | 0.0 | 0.0 | 0.0 | 0.0 | 0.8 | 0.1 | 0.5 | 16.4 | 1.2 | 0.0 | 2.5 | 0.0 | 2.2 |
| Epinephelus aeneus | 2.5 | 8.6 | 1.1 | 1.2 | 1.0 | 2.6 | 4.7 | 0.4 | 0.4 | 0.0 | 0.7 | 0.0 | 0.2 | 0.1 | 1.6 |
| Sharks | 0.7 | 7.7 | 1.6 | 0.4 | 0.2 | 0.2 | 0.6 | 0.3 | 3.5 | 0.1 | 0.4 | 0.0 | 0.9 | 1.4 | 1.2 |
| Rays | 0.5 | 7.1 | 0.4 | 0.0 | 0.1 | 1.0 | 1.3 | 0.2 | 2.4 | 1.0 | 0.9 | 0.1 | 0.1 | 0.4 | 1.1 |
| Mullus barbatus | 0.0 | 4.3 | 6.9 | 0.1 | 0.1 | 0.0 | 0.2 | 1.4 | 0.5 | 0.0 | 1.4 | 0.0 | 0.6 | 0.0 | 0.9 |
| Lithognathus mormyrus | 0.0 | 6.1 | 0.5 | 0.2 | 0.8 | 0.1 | 0.0 | 1.0 | 0.0 | 0.2 | 0.0 | 1.4 | 0.0 | 0.0 | 0.7 |
| Pomatomus saltatrix | 0.0 | 7.2 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 | 0.5 |
| Other species | 1.8 | 18.4 | 13.2 | 4.1 | 6.6 | 4.5 | 4.2 | 4.5 | 7.7 | 7.2 | 5.8 | 3.3 | 3.6 | 2.8 | 6.2 |
| . | Landings profile . | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Taxon . | A . | B . | C . | D . | E . | F . | G . | H . | I . | J . | K . | L . | M . | N . | Total . |
| Pagrus pagrus | 9.9 | 0.1 | 4.9 | 93.4 | 12.4 | 13.2 | 13.0 | 8.7 | 0.0 | 24.9 | 13.3 | 2.3 | 16.8 | 0.1 | 17.1 |
| Dilplodus sargus | 2.9 | 0.1 | 0.0 | 0.0 | 72.8 | 8.2 | 5.2 | 14.0 | 0.0 | 11.8 | 14.7 | 8.1 | 2.7 | 0.0 | 12.2 |
| Merluccius merluccius | 0.1 | 0.0 | 3.5 | 0.3 | 0.0 | 0.3 | 0.0 | 2.1 | 84.3 | 0.0 | 1.0 | 0.1 | 0.5 | 65.2 | 10.9 |
| Dentex dentex | 17.0 | 0.0 | 0.4 | 0.0 | 0.0 | 6.2 | 68.4 | 0.6 | 0.4 | 0.0 | 3.2 | 1.6 | 1.1 | 0.0 | 7.0 |
| Pagellus erythrinus | 0.7 | 0.0 | 0.3 | 0.0 | 0.0 | 0.4 | 0.3 | 50.7 | 0.0 | 12.5 | 0.0 | 1.6 | 16.5 | 0.0 | 6.7 |
| Sparus auratus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 5.7 | 0.0 | 0.6 | 5.8 | 72.5 | 0.0 | 0.0 | 5.8 |
| Epinephelus marginatus | 62.6 | 0.0 | 0.0 | 0.0 | 0.0 | 14.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.6 | 0.2 | 0.7 | 0.0 | 4.9 |
| Spondyliosoma cantharus | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 1.5 | 0.1 | 2.6 | 0.0 | 2.5 | 41.5 | 0.1 | 0.0 | 1.5 | 3.8 |
| Serranus cabrilla | 0.2 | 0.0 | 0.4 | 0.1 | 0.0 | 0.0 | 0.8 | 1.3 | 0.0 | 22.0 | 0.2 | 2.8 | 17.5 | 0.0 | 3.6 |
| Scorpaena scrofa | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.4 | 0.1 | 0.7 | 30.3 | 28.3 | 3.4 |
| Epinephelus costae | 0.0 | 0.6 | 0.0 | 0.0 | 0.2 | 47.7 | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.3 | 0.7 | 0.0 | 2.8 |
| Dicentrarchus labrax | 0.0 | 36.9 | 0.3 | 0.0 | 0.1 | 0.0 | 0.1 | 0.1 | 0.2 | 0.0 | 0.1 | 3.6 | 0.0 | 0.1 | 2.7 |
| Dentex macrophthalmus | 0.8 | 0.2 | 37.1 | 0.0 | 1.8 | 0.1 | 0.1 | 4.7 | 0.0 | 0.2 | 1.0 | 0.2 | 4.2 | 0.0 | 2.5 |
| Oblada melanura | 0.1 | 0.0 | 29.4 | 0.0 | 3.8 | 0.0 | 0.3 | 1.3 | 0.0 | 0.0 | 7.1 | 1.0 | 1.1 | 0.1 | 2.4 |
| Conger conger | 0.2 | 2.6 | 0.0 | 0.0 | 0.0 | 0.0 | 0.8 | 0.1 | 0.5 | 16.4 | 1.2 | 0.0 | 2.5 | 0.0 | 2.2 |
| Epinephelus aeneus | 2.5 | 8.6 | 1.1 | 1.2 | 1.0 | 2.6 | 4.7 | 0.4 | 0.4 | 0.0 | 0.7 | 0.0 | 0.2 | 0.1 | 1.6 |
| Sharks | 0.7 | 7.7 | 1.6 | 0.4 | 0.2 | 0.2 | 0.6 | 0.3 | 3.5 | 0.1 | 0.4 | 0.0 | 0.9 | 1.4 | 1.2 |
| Rays | 0.5 | 7.1 | 0.4 | 0.0 | 0.1 | 1.0 | 1.3 | 0.2 | 2.4 | 1.0 | 0.9 | 0.1 | 0.1 | 0.4 | 1.1 |
| Mullus barbatus | 0.0 | 4.3 | 6.9 | 0.1 | 0.1 | 0.0 | 0.2 | 1.4 | 0.5 | 0.0 | 1.4 | 0.0 | 0.6 | 0.0 | 0.9 |
| Lithognathus mormyrus | 0.0 | 6.1 | 0.5 | 0.2 | 0.8 | 0.1 | 0.0 | 1.0 | 0.0 | 0.2 | 0.0 | 1.4 | 0.0 | 0.0 | 0.7 |
| Pomatomus saltatrix | 0.0 | 7.2 | 0.0 | 0.2 | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | 0.0 | 0.0 | 0.2 | 0.0 | 0.0 | 0.5 |
| Other species | 1.8 | 18.4 | 13.2 | 4.1 | 6.6 | 4.5 | 4.2 | 4.5 | 7.7 | 7.2 | 5.8 | 3.3 | 3.6 | 2.8 | 6.2 |
The most important species of each profile (with a proportion in landings >10%) are shown emboldened.
Percentage distribution of the recorded fishing trips of the 14 set-longline-identified landing profiles in each main geographic area of the Aegean Sea.
| Cluster . | Sampled trips . | S Aegean . | N Aegean . | Evvoikos–Maliakos . | Argosaronikos . | Crete . |
|---|---|---|---|---|---|---|
| A | 276 | 8 | 3 | 1 | 2 | 9 |
| B | 284 | 3 | 5 | 63 | 7 | 4 |
| C | 180 | 5 | 1 | 0 | 0 | 7 |
| D | 375 | 11 | 5 | 0 | 5 | 11 |
| E | 420 | 9 | 4 | 1 | 14 | 19 |
| F | 241 | 5 | 1 | 0 | 2 | 16 |
| G | 326 | 5 | 11 | 3 | 8 | 11 |
| H | 369 | 8 | 10 | 13 | 17 | 3 |
| I | 373 | 1 | 24 | 6 | 21 | 5 |
| J | 435 | 18 | 1 | 0 | 2 | 2 |
| K | 337 | 4 | 22 | 0 | 6 | 5 |
| L | 290 | 7 | 9 | 11 | 9 | 0 |
| M | 265 | 9 | 1 | 0 | 3 | 7 |
| N | 219 | 8 | 3 | 0 | 4 | 1 |
| Sampled trips | 4 390 | 2 171 | 913 | 203 | 358 | 745 |
| Cluster . | Sampled trips . | S Aegean . | N Aegean . | Evvoikos–Maliakos . | Argosaronikos . | Crete . |
|---|---|---|---|---|---|---|
| A | 276 | 8 | 3 | 1 | 2 | 9 |
| B | 284 | 3 | 5 | 63 | 7 | 4 |
| C | 180 | 5 | 1 | 0 | 0 | 7 |
| D | 375 | 11 | 5 | 0 | 5 | 11 |
| E | 420 | 9 | 4 | 1 | 14 | 19 |
| F | 241 | 5 | 1 | 0 | 2 | 16 |
| G | 326 | 5 | 11 | 3 | 8 | 11 |
| H | 369 | 8 | 10 | 13 | 17 | 3 |
| I | 373 | 1 | 24 | 6 | 21 | 5 |
| J | 435 | 18 | 1 | 0 | 2 | 2 |
| K | 337 | 4 | 22 | 0 | 6 | 5 |
| L | 290 | 7 | 9 | 11 | 9 | 0 |
| M | 265 | 9 | 1 | 0 | 3 | 7 |
| N | 219 | 8 | 3 | 0 | 4 | 1 |
| Sampled trips | 4 390 | 2 171 | 913 | 203 | 358 | 745 |
The number of sampled trips by area and by landings profile is also provided. For each area, the landing profiles with a contribution ≥10% are shown emboldened.
Percentage distribution of the recorded fishing trips of the 14 set-longline-identified landing profiles in each main geographic area of the Aegean Sea.
| Cluster . | Sampled trips . | S Aegean . | N Aegean . | Evvoikos–Maliakos . | Argosaronikos . | Crete . |
|---|---|---|---|---|---|---|
| A | 276 | 8 | 3 | 1 | 2 | 9 |
| B | 284 | 3 | 5 | 63 | 7 | 4 |
| C | 180 | 5 | 1 | 0 | 0 | 7 |
| D | 375 | 11 | 5 | 0 | 5 | 11 |
| E | 420 | 9 | 4 | 1 | 14 | 19 |
| F | 241 | 5 | 1 | 0 | 2 | 16 |
| G | 326 | 5 | 11 | 3 | 8 | 11 |
| H | 369 | 8 | 10 | 13 | 17 | 3 |
| I | 373 | 1 | 24 | 6 | 21 | 5 |
| J | 435 | 18 | 1 | 0 | 2 | 2 |
| K | 337 | 4 | 22 | 0 | 6 | 5 |
| L | 290 | 7 | 9 | 11 | 9 | 0 |
| M | 265 | 9 | 1 | 0 | 3 | 7 |
| N | 219 | 8 | 3 | 0 | 4 | 1 |
| Sampled trips | 4 390 | 2 171 | 913 | 203 | 358 | 745 |
| Cluster . | Sampled trips . | S Aegean . | N Aegean . | Evvoikos–Maliakos . | Argosaronikos . | Crete . |
|---|---|---|---|---|---|---|
| A | 276 | 8 | 3 | 1 | 2 | 9 |
| B | 284 | 3 | 5 | 63 | 7 | 4 |
| C | 180 | 5 | 1 | 0 | 0 | 7 |
| D | 375 | 11 | 5 | 0 | 5 | 11 |
| E | 420 | 9 | 4 | 1 | 14 | 19 |
| F | 241 | 5 | 1 | 0 | 2 | 16 |
| G | 326 | 5 | 11 | 3 | 8 | 11 |
| H | 369 | 8 | 10 | 13 | 17 | 3 |
| I | 373 | 1 | 24 | 6 | 21 | 5 |
| J | 435 | 18 | 1 | 0 | 2 | 2 |
| K | 337 | 4 | 22 | 0 | 6 | 5 |
| L | 290 | 7 | 9 | 11 | 9 | 0 |
| M | 265 | 9 | 1 | 0 | 3 | 7 |
| N | 219 | 8 | 3 | 0 | 4 | 1 |
| Sampled trips | 4 390 | 2 171 | 913 | 203 | 358 | 745 |
The number of sampled trips by area and by landings profile is also provided. For each area, the landing profiles with a contribution ≥10% are shown emboldened.
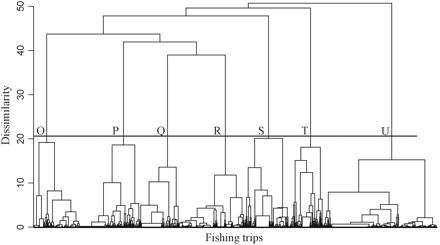
From the Ionian Sea, 47 species were recorded, and 26 were retained in the analysis (after removing the rare species). For those fishing trips, seven principal components were retained based on the scree diagram and on the contribution of each component to the total variance (Figure 2b). These seven components accounted for 66% of the total variation in the original data. The HAC analysis of the fishing trips based on the seven principal components led to the identification of seven clusters (O–U), representing different landings profiles (Figure 5). The clusters identified had different landings profiles (Table 3), different geographical distribution among the main subareas of the Ionian Sea (Table 4), and there were dissimilar temporal patterns in the frequency of the profiles (Figure 6).
Dendrogram of set longline fishing trips in the Ionian Sea, based on log-transformed landings profiles. Seven clusters (O–U) were identified, representing different landings profiles.
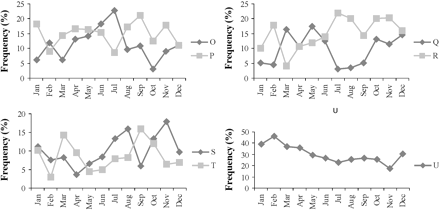
Monthly variation in percentage frequencies of occurrence of identified landings profiles from fishing trips sampled in the Ionian Sea. Landings profiles O–U are defined in Table 3.
Average landings profiles of the seven clusters identified in the Ionian Sea (Figure 5), given as a proportion (%) of the landings of each species to the total landings of each cluster.
| . | Landings profile . | |||||||
|---|---|---|---|---|---|---|---|---|
| Taxon . | O . | P . | Q . | R . | S . | T . | U . | Total . |
| Merluccius merluccius | 0.0 | 0.1 | 2.0 | 0.0 | 0.4 | 1.6 | 61.6 | 17.9 |
| Dilplodus sargus | 44.3 | 68.4 | 0.0 | 0.3 | 7.4 | 9.0 | 0.0 | 16.9 |
| Dentex dentex | 0.1 | 7.7 | 75.5 | 0.1 | 18.2 | 11.3 | 0.0 | 11.4 |
| Epinephelus marginatus | 0.0 | 0.0 | 0.0 | 0.1 | 60.8 | 9.6 | 0.0 | 7.5 |
| Pagrus pagrus | 0.0 | 0.0 | 0.0 | 0.0 | 0.1 | 55.3 | 0.0 | 4.7 |
| Phycis spp. | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 16.3 | 4.6 |
| Oblada melanura | 35.6 | 0.0 | 2.3 | 0.0 | 0.4 | 0.7 | 0.0 | 4.4 |
| Epinephelus costae | 16.5 | 0.2 | 6.7 | 0.7 | 6.4 | 3.4 | 0.0 | 3.7 |
| Epinephelus aeneus | 0.3 | 3.8 | 7.8 | 9.5 | 3.1 | 1.1 | 0.6 | 3.5 |
| Conger conger | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 10.7 | 3.0 |
| Pagellus erythrinus | 0.0 | 4.9 | 0.1 | 11.7 | 0.0 | 1.5 | 0.1 | 2.8 |
| Sharks | 0.0 | 0.0 | 0.1 | 8.9 | 0.0 | 0.2 | 3.8 | 2.6 |
| Seriola dumerili | 0.0 | 0.9 | 1.2 | 11.8 | 0.2 | 0.5 | 0.8 | 2.5 |
| Sparus aurata | 2.2 | 7.0 | 1.2 | 3.5 | 0.5 | 0.6 | 0.2 | 2.1 |
| Rays | 0.0 | 0.0 | 0.9 | 12.0 | 0.1 | 0.0 | 0.2 | 2.1 |
| Pomatomus saltatrix | 0.1 | 0.0 | 0.2 | 7.9 | 1.0 | 1.1 | 0.0 | 1.5 |
| Dentex macrophthalmus | 0.3 | 0.5 | 0.0 | 4.4 | 0.0 | 0.0 | 1.9 | 1.4 |
| Dicentrarchus labrax | 0.2 | 2.6 | 0.7 | 4.5 | 0.0 | 0.0 | 0.0 | 1.2 |
| Boops boops | 0.0 | 0.3 | 0.0 | 5.2 | 0.0 | 0.0 | 0.6 | 1.1 |
| Trachurus spp. | 0.0 | 0.3 | 0.0 | 1.7 | 0.0 | 0.0 | 2.4 | 1.0 |
| Diplodus annularis | 0.4 | 2.0 | 0.0 | 2.1 | 0.0 | 1.5 | 0.0 | 0.8 |
| Polyprion americanus | 0.0 | 0.0 | 0.0 | 2.3 | 0.9 | 0.5 | 0.5 | 0.7 |
| Spondyliosoma cantharus | 0.2 | 0.0 | 0.6 | 1.3 | 0.0 | 1.9 | 0.0 | 0.5 |
| Other species | 0.0 | 1.2 | 0.5 | 11.9 | 0.5 | 0.2 | 0.4 | 2.3 |
| . | Landings profile . | |||||||
|---|---|---|---|---|---|---|---|---|
| Taxon . | O . | P . | Q . | R . | S . | T . | U . | Total . |
| Merluccius merluccius | 0.0 | 0.1 | 2.0 | 0.0 | 0.4 | 1.6 | 61.6 | 17.9 |
| Dilplodus sargus | 44.3 | 68.4 | 0.0 | 0.3 | 7.4 | 9.0 | 0.0 | 16.9 |
| Dentex dentex | 0.1 | 7.7 | 75.5 | 0.1 | 18.2 | 11.3 | 0.0 | 11.4 |
| Epinephelus marginatus | 0.0 | 0.0 | 0.0 | 0.1 | 60.8 | 9.6 | 0.0 | 7.5 |
| Pagrus pagrus | 0.0 | 0.0 | 0.0 | 0.0 | 0.1 | 55.3 | 0.0 | 4.7 |
| Phycis spp. | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 16.3 | 4.6 |
| Oblada melanura | 35.6 | 0.0 | 2.3 | 0.0 | 0.4 | 0.7 | 0.0 | 4.4 |
| Epinephelus costae | 16.5 | 0.2 | 6.7 | 0.7 | 6.4 | 3.4 | 0.0 | 3.7 |
| Epinephelus aeneus | 0.3 | 3.8 | 7.8 | 9.5 | 3.1 | 1.1 | 0.6 | 3.5 |
| Conger conger | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 10.7 | 3.0 |
| Pagellus erythrinus | 0.0 | 4.9 | 0.1 | 11.7 | 0.0 | 1.5 | 0.1 | 2.8 |
| Sharks | 0.0 | 0.0 | 0.1 | 8.9 | 0.0 | 0.2 | 3.8 | 2.6 |
| Seriola dumerili | 0.0 | 0.9 | 1.2 | 11.8 | 0.2 | 0.5 | 0.8 | 2.5 |
| Sparus aurata | 2.2 | 7.0 | 1.2 | 3.5 | 0.5 | 0.6 | 0.2 | 2.1 |
| Rays | 0.0 | 0.0 | 0.9 | 12.0 | 0.1 | 0.0 | 0.2 | 2.1 |
| Pomatomus saltatrix | 0.1 | 0.0 | 0.2 | 7.9 | 1.0 | 1.1 | 0.0 | 1.5 |
| Dentex macrophthalmus | 0.3 | 0.5 | 0.0 | 4.4 | 0.0 | 0.0 | 1.9 | 1.4 |
| Dicentrarchus labrax | 0.2 | 2.6 | 0.7 | 4.5 | 0.0 | 0.0 | 0.0 | 1.2 |
| Boops boops | 0.0 | 0.3 | 0.0 | 5.2 | 0.0 | 0.0 | 0.6 | 1.1 |
| Trachurus spp. | 0.0 | 0.3 | 0.0 | 1.7 | 0.0 | 0.0 | 2.4 | 1.0 |
| Diplodus annularis | 0.4 | 2.0 | 0.0 | 2.1 | 0.0 | 1.5 | 0.0 | 0.8 |
| Polyprion americanus | 0.0 | 0.0 | 0.0 | 2.3 | 0.9 | 0.5 | 0.5 | 0.7 |
| Spondyliosoma cantharus | 0.2 | 0.0 | 0.6 | 1.3 | 0.0 | 1.9 | 0.0 | 0.5 |
| Other species | 0.0 | 1.2 | 0.5 | 11.9 | 0.5 | 0.2 | 0.4 | 2.3 |
The most important species of each profile (with a proportion in landings >10%) are shown emboldened.
Average landings profiles of the seven clusters identified in the Ionian Sea (Figure 5), given as a proportion (%) of the landings of each species to the total landings of each cluster.
| . | Landings profile . | |||||||
|---|---|---|---|---|---|---|---|---|
| Taxon . | O . | P . | Q . | R . | S . | T . | U . | Total . |
| Merluccius merluccius | 0.0 | 0.1 | 2.0 | 0.0 | 0.4 | 1.6 | 61.6 | 17.9 |
| Dilplodus sargus | 44.3 | 68.4 | 0.0 | 0.3 | 7.4 | 9.0 | 0.0 | 16.9 |
| Dentex dentex | 0.1 | 7.7 | 75.5 | 0.1 | 18.2 | 11.3 | 0.0 | 11.4 |
| Epinephelus marginatus | 0.0 | 0.0 | 0.0 | 0.1 | 60.8 | 9.6 | 0.0 | 7.5 |
| Pagrus pagrus | 0.0 | 0.0 | 0.0 | 0.0 | 0.1 | 55.3 | 0.0 | 4.7 |
| Phycis spp. | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 16.3 | 4.6 |
| Oblada melanura | 35.6 | 0.0 | 2.3 | 0.0 | 0.4 | 0.7 | 0.0 | 4.4 |
| Epinephelus costae | 16.5 | 0.2 | 6.7 | 0.7 | 6.4 | 3.4 | 0.0 | 3.7 |
| Epinephelus aeneus | 0.3 | 3.8 | 7.8 | 9.5 | 3.1 | 1.1 | 0.6 | 3.5 |
| Conger conger | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 10.7 | 3.0 |
| Pagellus erythrinus | 0.0 | 4.9 | 0.1 | 11.7 | 0.0 | 1.5 | 0.1 | 2.8 |
| Sharks | 0.0 | 0.0 | 0.1 | 8.9 | 0.0 | 0.2 | 3.8 | 2.6 |
| Seriola dumerili | 0.0 | 0.9 | 1.2 | 11.8 | 0.2 | 0.5 | 0.8 | 2.5 |
| Sparus aurata | 2.2 | 7.0 | 1.2 | 3.5 | 0.5 | 0.6 | 0.2 | 2.1 |
| Rays | 0.0 | 0.0 | 0.9 | 12.0 | 0.1 | 0.0 | 0.2 | 2.1 |
| Pomatomus saltatrix | 0.1 | 0.0 | 0.2 | 7.9 | 1.0 | 1.1 | 0.0 | 1.5 |
| Dentex macrophthalmus | 0.3 | 0.5 | 0.0 | 4.4 | 0.0 | 0.0 | 1.9 | 1.4 |
| Dicentrarchus labrax | 0.2 | 2.6 | 0.7 | 4.5 | 0.0 | 0.0 | 0.0 | 1.2 |
| Boops boops | 0.0 | 0.3 | 0.0 | 5.2 | 0.0 | 0.0 | 0.6 | 1.1 |
| Trachurus spp. | 0.0 | 0.3 | 0.0 | 1.7 | 0.0 | 0.0 | 2.4 | 1.0 |
| Diplodus annularis | 0.4 | 2.0 | 0.0 | 2.1 | 0.0 | 1.5 | 0.0 | 0.8 |
| Polyprion americanus | 0.0 | 0.0 | 0.0 | 2.3 | 0.9 | 0.5 | 0.5 | 0.7 |
| Spondyliosoma cantharus | 0.2 | 0.0 | 0.6 | 1.3 | 0.0 | 1.9 | 0.0 | 0.5 |
| Other species | 0.0 | 1.2 | 0.5 | 11.9 | 0.5 | 0.2 | 0.4 | 2.3 |
| . | Landings profile . | |||||||
|---|---|---|---|---|---|---|---|---|
| Taxon . | O . | P . | Q . | R . | S . | T . | U . | Total . |
| Merluccius merluccius | 0.0 | 0.1 | 2.0 | 0.0 | 0.4 | 1.6 | 61.6 | 17.9 |
| Dilplodus sargus | 44.3 | 68.4 | 0.0 | 0.3 | 7.4 | 9.0 | 0.0 | 16.9 |
| Dentex dentex | 0.1 | 7.7 | 75.5 | 0.1 | 18.2 | 11.3 | 0.0 | 11.4 |
| Epinephelus marginatus | 0.0 | 0.0 | 0.0 | 0.1 | 60.8 | 9.6 | 0.0 | 7.5 |
| Pagrus pagrus | 0.0 | 0.0 | 0.0 | 0.0 | 0.1 | 55.3 | 0.0 | 4.7 |
| Phycis spp. | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 16.3 | 4.6 |
| Oblada melanura | 35.6 | 0.0 | 2.3 | 0.0 | 0.4 | 0.7 | 0.0 | 4.4 |
| Epinephelus costae | 16.5 | 0.2 | 6.7 | 0.7 | 6.4 | 3.4 | 0.0 | 3.7 |
| Epinephelus aeneus | 0.3 | 3.8 | 7.8 | 9.5 | 3.1 | 1.1 | 0.6 | 3.5 |
| Conger conger | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 10.7 | 3.0 |
| Pagellus erythrinus | 0.0 | 4.9 | 0.1 | 11.7 | 0.0 | 1.5 | 0.1 | 2.8 |
| Sharks | 0.0 | 0.0 | 0.1 | 8.9 | 0.0 | 0.2 | 3.8 | 2.6 |
| Seriola dumerili | 0.0 | 0.9 | 1.2 | 11.8 | 0.2 | 0.5 | 0.8 | 2.5 |
| Sparus aurata | 2.2 | 7.0 | 1.2 | 3.5 | 0.5 | 0.6 | 0.2 | 2.1 |
| Rays | 0.0 | 0.0 | 0.9 | 12.0 | 0.1 | 0.0 | 0.2 | 2.1 |
| Pomatomus saltatrix | 0.1 | 0.0 | 0.2 | 7.9 | 1.0 | 1.1 | 0.0 | 1.5 |
| Dentex macrophthalmus | 0.3 | 0.5 | 0.0 | 4.4 | 0.0 | 0.0 | 1.9 | 1.4 |
| Dicentrarchus labrax | 0.2 | 2.6 | 0.7 | 4.5 | 0.0 | 0.0 | 0.0 | 1.2 |
| Boops boops | 0.0 | 0.3 | 0.0 | 5.2 | 0.0 | 0.0 | 0.6 | 1.1 |
| Trachurus spp. | 0.0 | 0.3 | 0.0 | 1.7 | 0.0 | 0.0 | 2.4 | 1.0 |
| Diplodus annularis | 0.4 | 2.0 | 0.0 | 2.1 | 0.0 | 1.5 | 0.0 | 0.8 |
| Polyprion americanus | 0.0 | 0.0 | 0.0 | 2.3 | 0.9 | 0.5 | 0.5 | 0.7 |
| Spondyliosoma cantharus | 0.2 | 0.0 | 0.6 | 1.3 | 0.0 | 1.9 | 0.0 | 0.5 |
| Other species | 0.0 | 1.2 | 0.5 | 11.9 | 0.5 | 0.2 | 0.4 | 2.3 |
The most important species of each profile (with a proportion in landings >10%) are shown emboldened.
Percentage distribution of the recorded fishing trips of the seven identified set longline landings profiles in each main geographic area of the Ionian Sea.
| Cluster . | Sampled trips . | C–S Ionian . | Korinthiakos . | N Ionian . |
|---|---|---|---|---|
| O | 158 | 16 | 0 | 4 |
| P | 207 | 20 | 0 | 13 |
| Q | 134 | 12 | 3 | 8 |
| R | 225 | 18 | 2 | 40 |
| S | 151 | 15 | 0 | 8 |
| T | 118 | 10 | 0 | 15 |
| U | 393 | 8 | 95 | 13 |
| Sampled trips | 1 386 | 926 | 316 | 144 |
| Cluster . | Sampled trips . | C–S Ionian . | Korinthiakos . | N Ionian . |
|---|---|---|---|---|
| O | 158 | 16 | 0 | 4 |
| P | 207 | 20 | 0 | 13 |
| Q | 134 | 12 | 3 | 8 |
| R | 225 | 18 | 2 | 40 |
| S | 151 | 15 | 0 | 8 |
| T | 118 | 10 | 0 | 15 |
| U | 393 | 8 | 95 | 13 |
| Sampled trips | 1 386 | 926 | 316 | 144 |
The number of sampled trips by area and by landing profile is also provided. For each area, the landing profiles with a contribution ≥15% are shown emboldened.
Percentage distribution of the recorded fishing trips of the seven identified set longline landings profiles in each main geographic area of the Ionian Sea.
| Cluster . | Sampled trips . | C–S Ionian . | Korinthiakos . | N Ionian . |
|---|---|---|---|---|
| O | 158 | 16 | 0 | 4 |
| P | 207 | 20 | 0 | 13 |
| Q | 134 | 12 | 3 | 8 |
| R | 225 | 18 | 2 | 40 |
| S | 151 | 15 | 0 | 8 |
| T | 118 | 10 | 0 | 15 |
| U | 393 | 8 | 95 | 13 |
| Sampled trips | 1 386 | 926 | 316 | 144 |
| Cluster . | Sampled trips . | C–S Ionian . | Korinthiakos . | N Ionian . |
|---|---|---|---|---|
| O | 158 | 16 | 0 | 4 |
| P | 207 | 20 | 0 | 13 |
| Q | 134 | 12 | 3 | 8 |
| R | 225 | 18 | 2 | 40 |
| S | 151 | 15 | 0 | 8 |
| T | 118 | 10 | 0 | 15 |
| U | 393 | 8 | 95 | 13 |
| Sampled trips | 1 386 | 926 | 316 | 144 |
The number of sampled trips by area and by landing profile is also provided. For each area, the landing profiles with a contribution ≥15% are shown emboldened.
Common sea bream (Pagrus pagrus), white sea bream (Diplodus sargus), and hake (Merluccius merluccius) contributed most to longliner landings in the Aegean Sea (40.2%), and hake, white sea bream, and common dentex (Dentex dentex) most (46.2%) in the Ionian Sea. Overall, 16 species in the Aegean Sea and 15 in the Ionian Sea accounted for 90% of the landings (Tables 1 and 3).
The main characteristics of the identified set longline landings profiles and potential métiers in the Aegean and Ionian Seas are summarized in Table 5. The two identified mixed landings profiles in the Aegean Sea (J and M) were combined in a single métier (AEG-LLS-10), because most species were common (with different landings ratios), and the main difference was the absence of large-scaled scorpionfish (Scorpaena scrofa) from the landings of profile J, whereas it constituted 30.3% of the landings in métier M. In both profiles, P. pagrus and Pagellus erythrinus were commercially the most important target species (with their higher market value). Both are found over rocky, rubble, or sandy substrata mostly at depths <100 m, with greater abundance in the bathymetric range 20–80 m (Labropoulou et al., 1999; Somarakis and Machias, 2002; Katsanevakis and Maravelias, 2009), i.e. in a rather narrow bathymetric range. Large-scale scorpionfish were common in the catches between March and June, when profile M peaked, but less frequent in winter, when profile J peaked (Figure 4). Both profiles are observed in all landing ports of the same area (South Aegean), so the absence of large-scaled scorpionfish from cluster J is rather by chance or possible seasonality in scorpionfish catches than the result of a different fishing strategy. The main target species of profiles I and N was hake, but in profile N there was also a substantial catch of large-scaled scorpionfish, which was completely absent from profile I. We preferred to characterize the two profiles as separate métiers because they refer to different fishing grounds (profile I was observed mainly in the North Aegean and Argosaronikos, and N in the South Aegean).
Description of the identified set longline landings profiles and potential métiers.
| Cluster (Fishery) . | Cluster size . | Fishery characterization . | Main species . | Typical fishing locations . | Métier . |
|---|---|---|---|---|---|
| Aegean Sea | |||||
| A | 276 | Dusky grouper | Epinephelus marginatus (62.6%), Dentex dentex (17.0%) | S Aegean, Crete | AEG-LLS-1 |
| B | 284 | European sea bass | Dicentrarchus labrax (36.9%) | Evvoikos | AEG-LLS-2 |
| C | 180 | Large-eye dentex | Dentex macrophthalmus (37.1%), Oblada melanura (29.4%) | S Aegean, Crete | AEG-LLS-3 |
| D | 375 | Common sea bream | Pagrus pagrus (93.4%) | S Aegean, Crete | AEG-LLS-4 |
| E | 420 | White sea bream | Diplodus sargus (72.8%), Pagrus pagrus (12.4%) | Crete, Argosaronikos, S Aegean | AEG-LLS-5 |
| F | 241 | Goldblotch grouper | Epinephelus costae (47.7%), Epinephelus marginatus (14.0%), Pagrus pagrus (13.2%) | S Aegean, Crete | AEG-LLS-6 |
| G | 326 | Common dentex | Dentex dentex (68.4%), Pagrus pagrus (13.0%) | S and N Aegean, Crete, Argosaronikos | AEG-LLS-7 |
| H | 369 | Common pandora | Pagellus erythrinus (50.7%), Diplodus sargus (14.0%) | S and N Aegean, Argosaronikos, Evvoikos | AEG-LLS-8 |
| I | 373 | Hake | Merluccius merluccius (84.3%) | N Aegean, Argosaronikos | AEG-LLS-9 |
| J | 435 | Mixed 1 | Pagrus pagrus (24.9%), Serranus cabrilla (22.0%), Conger conger (16.4%), Pagellus erythrinus (12.5%), Diplodus sargus (11.8%) | S Aegean | AEG-LLS-10a |
| K | 337 | Black sea bream | Spondyliosoma cantharus (41.5%), Diplodus sargus (14.7%), Pagrus pagrus (13.3%) | N Aegean | AEG-LLS-11 |
| L | 290 | Gilthead sea bream | Sparus aurata (72.5%) | Argosaronikos | AEG-LLS-12 |
| M | 265 | Mixed 2 | Scorpaena scrofa (30.3%), Serranus cabrilla (17.5%), Pagrus pagrus (16.8%), Pagellus erythrinus (16.5%) | S Aegean, Crete | AEG-LLS-10a |
| N | 219 | Hake–Largescaled scorpionfish | Merluccius merluccius (65.2%), Scorpaena scrofa (28.3%) | S Aegean | AEG-LLS-13 |
| Ionian Sea | |||||
| O | 158 | White sea bream–Saddled sea bream | Diplodus sargus (44.3%), Oblada melanura (35.6%), Epinephelus costae (16.5%) | C–S Ionian | ION-LLS-1 |
| P | 207 | White sea bream | Diplodus sargus (68.4%) | C–S and N Ionian | ION-LLS-2 |
| Q | 134 | Common dentex | Dentex dentex (75.5%) | C–S and N Ionian | ION-LLS-3 |
| R | 225 | Mixed 3 | Rays (12.0%), Seriola dumerili (11.8%), Pagellus erythrinus (11.7%), Epinephelus aeneus (9.5%) | N and C–S Ionian | ION-LLS-4 |
| S | 151 | Dusky grouper | Epinephelus marginatus (60.8%), Dentex dentex (18.2%) | C–S Ionian | ION-LLS-5 |
| T | 118 | Common sea bream | Pagrus pagrus (55.3%), Dentex dentex (11.3%) | N and C–S Ionian | ION-LLS-6 |
| U | 393 | Hake | Merluccius merluccius (61.6%), Phycis spp. (16.3%), Conger conger (10.7%) | Korinthiakos, N Ionian | ION-LLS-7 |
| Cluster (Fishery) . | Cluster size . | Fishery characterization . | Main species . | Typical fishing locations . | Métier . |
|---|---|---|---|---|---|
| Aegean Sea | |||||
| A | 276 | Dusky grouper | Epinephelus marginatus (62.6%), Dentex dentex (17.0%) | S Aegean, Crete | AEG-LLS-1 |
| B | 284 | European sea bass | Dicentrarchus labrax (36.9%) | Evvoikos | AEG-LLS-2 |
| C | 180 | Large-eye dentex | Dentex macrophthalmus (37.1%), Oblada melanura (29.4%) | S Aegean, Crete | AEG-LLS-3 |
| D | 375 | Common sea bream | Pagrus pagrus (93.4%) | S Aegean, Crete | AEG-LLS-4 |
| E | 420 | White sea bream | Diplodus sargus (72.8%), Pagrus pagrus (12.4%) | Crete, Argosaronikos, S Aegean | AEG-LLS-5 |
| F | 241 | Goldblotch grouper | Epinephelus costae (47.7%), Epinephelus marginatus (14.0%), Pagrus pagrus (13.2%) | S Aegean, Crete | AEG-LLS-6 |
| G | 326 | Common dentex | Dentex dentex (68.4%), Pagrus pagrus (13.0%) | S and N Aegean, Crete, Argosaronikos | AEG-LLS-7 |
| H | 369 | Common pandora | Pagellus erythrinus (50.7%), Diplodus sargus (14.0%) | S and N Aegean, Argosaronikos, Evvoikos | AEG-LLS-8 |
| I | 373 | Hake | Merluccius merluccius (84.3%) | N Aegean, Argosaronikos | AEG-LLS-9 |
| J | 435 | Mixed 1 | Pagrus pagrus (24.9%), Serranus cabrilla (22.0%), Conger conger (16.4%), Pagellus erythrinus (12.5%), Diplodus sargus (11.8%) | S Aegean | AEG-LLS-10a |
| K | 337 | Black sea bream | Spondyliosoma cantharus (41.5%), Diplodus sargus (14.7%), Pagrus pagrus (13.3%) | N Aegean | AEG-LLS-11 |
| L | 290 | Gilthead sea bream | Sparus aurata (72.5%) | Argosaronikos | AEG-LLS-12 |
| M | 265 | Mixed 2 | Scorpaena scrofa (30.3%), Serranus cabrilla (17.5%), Pagrus pagrus (16.8%), Pagellus erythrinus (16.5%) | S Aegean, Crete | AEG-LLS-10a |
| N | 219 | Hake–Largescaled scorpionfish | Merluccius merluccius (65.2%), Scorpaena scrofa (28.3%) | S Aegean | AEG-LLS-13 |
| Ionian Sea | |||||
| O | 158 | White sea bream–Saddled sea bream | Diplodus sargus (44.3%), Oblada melanura (35.6%), Epinephelus costae (16.5%) | C–S Ionian | ION-LLS-1 |
| P | 207 | White sea bream | Diplodus sargus (68.4%) | C–S and N Ionian | ION-LLS-2 |
| Q | 134 | Common dentex | Dentex dentex (75.5%) | C–S and N Ionian | ION-LLS-3 |
| R | 225 | Mixed 3 | Rays (12.0%), Seriola dumerili (11.8%), Pagellus erythrinus (11.7%), Epinephelus aeneus (9.5%) | N and C–S Ionian | ION-LLS-4 |
| S | 151 | Dusky grouper | Epinephelus marginatus (60.8%), Dentex dentex (18.2%) | C–S Ionian | ION-LLS-5 |
| T | 118 | Common sea bream | Pagrus pagrus (55.3%), Dentex dentex (11.3%) | N and C–S Ionian | ION-LLS-6 |
| U | 393 | Hake | Merluccius merluccius (61.6%), Phycis spp. (16.3%), Conger conger (10.7%) | Korinthiakos, N Ionian | ION-LLS-7 |
aProfiles J and M were combined as métier AEG-LLS-10.
Description of the identified set longline landings profiles and potential métiers.
| Cluster (Fishery) . | Cluster size . | Fishery characterization . | Main species . | Typical fishing locations . | Métier . |
|---|---|---|---|---|---|
| Aegean Sea | |||||
| A | 276 | Dusky grouper | Epinephelus marginatus (62.6%), Dentex dentex (17.0%) | S Aegean, Crete | AEG-LLS-1 |
| B | 284 | European sea bass | Dicentrarchus labrax (36.9%) | Evvoikos | AEG-LLS-2 |
| C | 180 | Large-eye dentex | Dentex macrophthalmus (37.1%), Oblada melanura (29.4%) | S Aegean, Crete | AEG-LLS-3 |
| D | 375 | Common sea bream | Pagrus pagrus (93.4%) | S Aegean, Crete | AEG-LLS-4 |
| E | 420 | White sea bream | Diplodus sargus (72.8%), Pagrus pagrus (12.4%) | Crete, Argosaronikos, S Aegean | AEG-LLS-5 |
| F | 241 | Goldblotch grouper | Epinephelus costae (47.7%), Epinephelus marginatus (14.0%), Pagrus pagrus (13.2%) | S Aegean, Crete | AEG-LLS-6 |
| G | 326 | Common dentex | Dentex dentex (68.4%), Pagrus pagrus (13.0%) | S and N Aegean, Crete, Argosaronikos | AEG-LLS-7 |
| H | 369 | Common pandora | Pagellus erythrinus (50.7%), Diplodus sargus (14.0%) | S and N Aegean, Argosaronikos, Evvoikos | AEG-LLS-8 |
| I | 373 | Hake | Merluccius merluccius (84.3%) | N Aegean, Argosaronikos | AEG-LLS-9 |
| J | 435 | Mixed 1 | Pagrus pagrus (24.9%), Serranus cabrilla (22.0%), Conger conger (16.4%), Pagellus erythrinus (12.5%), Diplodus sargus (11.8%) | S Aegean | AEG-LLS-10a |
| K | 337 | Black sea bream | Spondyliosoma cantharus (41.5%), Diplodus sargus (14.7%), Pagrus pagrus (13.3%) | N Aegean | AEG-LLS-11 |
| L | 290 | Gilthead sea bream | Sparus aurata (72.5%) | Argosaronikos | AEG-LLS-12 |
| M | 265 | Mixed 2 | Scorpaena scrofa (30.3%), Serranus cabrilla (17.5%), Pagrus pagrus (16.8%), Pagellus erythrinus (16.5%) | S Aegean, Crete | AEG-LLS-10a |
| N | 219 | Hake–Largescaled scorpionfish | Merluccius merluccius (65.2%), Scorpaena scrofa (28.3%) | S Aegean | AEG-LLS-13 |
| Ionian Sea | |||||
| O | 158 | White sea bream–Saddled sea bream | Diplodus sargus (44.3%), Oblada melanura (35.6%), Epinephelus costae (16.5%) | C–S Ionian | ION-LLS-1 |
| P | 207 | White sea bream | Diplodus sargus (68.4%) | C–S and N Ionian | ION-LLS-2 |
| Q | 134 | Common dentex | Dentex dentex (75.5%) | C–S and N Ionian | ION-LLS-3 |
| R | 225 | Mixed 3 | Rays (12.0%), Seriola dumerili (11.8%), Pagellus erythrinus (11.7%), Epinephelus aeneus (9.5%) | N and C–S Ionian | ION-LLS-4 |
| S | 151 | Dusky grouper | Epinephelus marginatus (60.8%), Dentex dentex (18.2%) | C–S Ionian | ION-LLS-5 |
| T | 118 | Common sea bream | Pagrus pagrus (55.3%), Dentex dentex (11.3%) | N and C–S Ionian | ION-LLS-6 |
| U | 393 | Hake | Merluccius merluccius (61.6%), Phycis spp. (16.3%), Conger conger (10.7%) | Korinthiakos, N Ionian | ION-LLS-7 |
| Cluster (Fishery) . | Cluster size . | Fishery characterization . | Main species . | Typical fishing locations . | Métier . |
|---|---|---|---|---|---|
| Aegean Sea | |||||
| A | 276 | Dusky grouper | Epinephelus marginatus (62.6%), Dentex dentex (17.0%) | S Aegean, Crete | AEG-LLS-1 |
| B | 284 | European sea bass | Dicentrarchus labrax (36.9%) | Evvoikos | AEG-LLS-2 |
| C | 180 | Large-eye dentex | Dentex macrophthalmus (37.1%), Oblada melanura (29.4%) | S Aegean, Crete | AEG-LLS-3 |
| D | 375 | Common sea bream | Pagrus pagrus (93.4%) | S Aegean, Crete | AEG-LLS-4 |
| E | 420 | White sea bream | Diplodus sargus (72.8%), Pagrus pagrus (12.4%) | Crete, Argosaronikos, S Aegean | AEG-LLS-5 |
| F | 241 | Goldblotch grouper | Epinephelus costae (47.7%), Epinephelus marginatus (14.0%), Pagrus pagrus (13.2%) | S Aegean, Crete | AEG-LLS-6 |
| G | 326 | Common dentex | Dentex dentex (68.4%), Pagrus pagrus (13.0%) | S and N Aegean, Crete, Argosaronikos | AEG-LLS-7 |
| H | 369 | Common pandora | Pagellus erythrinus (50.7%), Diplodus sargus (14.0%) | S and N Aegean, Argosaronikos, Evvoikos | AEG-LLS-8 |
| I | 373 | Hake | Merluccius merluccius (84.3%) | N Aegean, Argosaronikos | AEG-LLS-9 |
| J | 435 | Mixed 1 | Pagrus pagrus (24.9%), Serranus cabrilla (22.0%), Conger conger (16.4%), Pagellus erythrinus (12.5%), Diplodus sargus (11.8%) | S Aegean | AEG-LLS-10a |
| K | 337 | Black sea bream | Spondyliosoma cantharus (41.5%), Diplodus sargus (14.7%), Pagrus pagrus (13.3%) | N Aegean | AEG-LLS-11 |
| L | 290 | Gilthead sea bream | Sparus aurata (72.5%) | Argosaronikos | AEG-LLS-12 |
| M | 265 | Mixed 2 | Scorpaena scrofa (30.3%), Serranus cabrilla (17.5%), Pagrus pagrus (16.8%), Pagellus erythrinus (16.5%) | S Aegean, Crete | AEG-LLS-10a |
| N | 219 | Hake–Largescaled scorpionfish | Merluccius merluccius (65.2%), Scorpaena scrofa (28.3%) | S Aegean | AEG-LLS-13 |
| Ionian Sea | |||||
| O | 158 | White sea bream–Saddled sea bream | Diplodus sargus (44.3%), Oblada melanura (35.6%), Epinephelus costae (16.5%) | C–S Ionian | ION-LLS-1 |
| P | 207 | White sea bream | Diplodus sargus (68.4%) | C–S and N Ionian | ION-LLS-2 |
| Q | 134 | Common dentex | Dentex dentex (75.5%) | C–S and N Ionian | ION-LLS-3 |
| R | 225 | Mixed 3 | Rays (12.0%), Seriola dumerili (11.8%), Pagellus erythrinus (11.7%), Epinephelus aeneus (9.5%) | N and C–S Ionian | ION-LLS-4 |
| S | 151 | Dusky grouper | Epinephelus marginatus (60.8%), Dentex dentex (18.2%) | C–S Ionian | ION-LLS-5 |
| T | 118 | Common sea bream | Pagrus pagrus (55.3%), Dentex dentex (11.3%) | N and C–S Ionian | ION-LLS-6 |
| U | 393 | Hake | Merluccius merluccius (61.6%), Phycis spp. (16.3%), Conger conger (10.7%) | Korinthiakos, N Ionian | ION-LLS-7 |
aProfiles J and M were combined as métier AEG-LLS-10.
The mixed métier (AEG-LLS-10, profiles J and M) was the one most frequently observed in the Aegean Sea samples, followed by métiers targeting white sea bream in Crete, Argosaronikos, and the South Aegean (AEG-LLS-5, profile E), hake in the North Aegean and Argosarinokos (AEG-LLS-9, profile I), common sea bream in the South Aegean and Crete (AEG-LLS-4, profile D), and common pandora (P. erythrinus) in most subareas (AEG-LLS-8, profile H). The European sea bass (Dicentrarchus labrax) métier (AEG-LLS-2, profile B) was operated almost exclusively in the Evvoikos Gulf and represented 63% of the fishing trips recorded there. It was highly seasonal and was prosecuted primarily during summer (Figure 4). The métier targeting gilthead sea bream (Sparus aurata; AEG-LLS-12, profile L) was highly seasonal, mostly between November and January (Figure 4).
In the Ionian Sea, the most frequently observed métier was that targeting hake in the Korinthiakos Gulf and the North Ionian (ION-LLS-7, profile U). That was by far the most important métier in the Korinthiakos Gulf, representing 95% of the recorded fishing trips. It exhibited a seasonal pattern, and operated mainly during the cold season (Figure 4). The mixed métier (ION-LLS-4, profile R) was the one most frequently observed in the North Ionian Sea, yielding 40% of the fishing trips recorded.
Discussion
The set longline fisheries in the Mediterranean primarily involve coastal fishing on board small boats <10 m long, on short trips rarely lasting >12 h. This essentially small-scale activity is also reflected in social terms. Most of the coastal vessels are operated by one or two professional fishers, and their operation is mainly a family affair. Fishers generally own their vessel and equipment and support their family with their activity, sometimes as a supplement to another job. They also develop their fishing skills within the family, from their elders. With its centuries-old traditions, Mediterranean fishing has tremendous socio-cultural importance for certain coastal communities, particularly in the islands (Tzanatos et al., 2005). Small-scale fishing is an important constituent of the local economies and contributes substantially to the sustainability of local communities. Yet its impact on resources should not be underestimated; it can be very efficient when large gears are used.
Catches by longline are influenced by factors that affect selectivity and efficiency, including choice of fishing site, timing of fishing, and duration of soak (Erzini et al., 1996, and references therein), as well as gear characteristics such as hook size and design, gangion features (length, diameter, colour, type), gangion accessories such as floats and swivels, bait characteristics (type, size, shape, smell, colour), and diameter of the main line. Fishers make choices based on all these factors, depending on their target species. Métier definition summarizes all these choices.
Métier choice is a trade-off among a combination of factors such as species abundance, catchability, market value, and accessibility of the fishing grounds. Owing to the geographic variability in fish assemblages and the differences in market values among locations and seasons, fisher motivation to engage in a specific métier varies both spatially and temporarily. Here we found a marked seasonality in some métiers. For example, AEG-LLS-2 and ION-LLS-1 were mostly operated during summer, AEG-LLS-12 and ION-LLS-7 typically during winter, and AEG-LLS-11 peaked between August and October. However, the exact pattern of métier shift in each location through a year cannot be deduced from the present analysis, and other gears (e.g. nets and traps) that are typically operated by coastal fishing vessels are also involved.
Studies conducted on a local scale are likely to identify just a subgroup of the total number of métiers operated at a national scale. For example, Tzanatos et al. (2005) attempted to identify the main métiers of the small-scale fisheries in Greece, based on 227 questionnaires from 9 of the 40 coastal prefectures of Greece. They identified seven longline métiers, with the main target species of common sea bream, common dentex, white sea bream, common pandora, and striped sea bream (Lithognathus mormyrus). Except the métier targeting striped sea bream, we also identified the same ones, i.e. AEG-LLS-5, AEG-LLS-7, AEG-LLS-8, AEG-LLS-10, and ION-LLS-6. In a classification of small-scale fisheries in the Patraikos Gulf (Ionian Sea) by Tzanatos et al. (2006), three métiers were identified targeting common dentex, dusky grouper (Epinephelus marginatus), hake, and greater amberjack (Seriola dumerili), corresponding here to ION-LLS-4, ION-LLS-5, and ION-LLS-7. The total annual effort of Greek set longliners amounts to ∼250 000 d at sea (HCMR, 2008), so our sample represents ∼0.5% of all fishing trips. Although the proportion of sampled fishing trips is low, the absolute number of sampled fishing trips is high, with a good spatial coverage, so we expect to have identified all major longline métiers. However, it is likely that some local métiers of minor importance were not represented in our sample.
Métiers targeting small and medium sea breams (Sparidae), such as AEG-LLS-5, AEG-LLS-8, AEG-LLS-12, ION-LLS-1, and ION-LLS-2, usually operate in shallow water down to 100 m, and utilize hooks with nominal numbers usually between 11 and 16. The longline consists of a monofilament main line 0.5–1.0 mm thick, and monofilament gangions of variable length and 0.2–0.6 mm thick (Erzini et al., 1996). Métiers targeting larger and/or deeper dwelling sea breams (common sea bream or black sea bream, Spondyliosoma cantharus), common dentex, large-eye dentex (Dentex macrophthalmus), or groupers (dusky grouper, and goldblotch grouper, Epinephelus costae), such as AEG-LLS-1, AEG-LLS-3, AEG-LLS-4, AEG-LLS-6, AEG-LLS-7, AEG-LLS-11, ION-LLS-3, ION-LLS-5, and ION-LLS-6, usually operate in deeper water down to 180 m, and utilize hooks with nominal numbers between 8 and 12. The diameter of the main line then is usually between 0.5 and 1.2 mm and of the gangions between 0.4 and 0.8 mm (Adamidou, 2007). Métiers targeting hake (AEG-LLS-9, AEG-LLS-13, ION-LLS-7) operate in deep water, usually between 100 and 600 m. The usual nominal number of hooks in that fishery is 3–7, the main line is 1–2.5 mm thick, and the gangions have a diameter of 0.5–1.0 mm, and are sometimes made of wire (Adamidou, 2007).
Profiles of catches or landings have been used extensively in the literature to define métiers (Lewy and Vinther, 1994; He et al., 1997; Pelletier and Ferraris, 2000; Ulrich and Andersen, 2004; Marchal, 2008). However, there are limitations to such analyses. A landings profile may not reflect a fisher's intentions strictly, but rather approximate what was initially targeted and also the relative abundance and distribution of the species. The real catch may sometimes differ noticeably from the targeted species, and métiers might not persist over time. There is always uncertainty regarding the outcome of a fishing trip, and there is no guarantee that the fisher's choices of gear, fishing ground, or timing will have the anticipated result. In a comparative analysis of métiers and resulting landings profiles in seven French demersal and pelagic fleets, Marchal (2008) found that, in pelagic trawlers and gillnetters, the uncertainty in forecasting métiers from landings profiles was low. In contrast, he reported great uncertainty when forecasting bottom-trawl métiers from landings profiles.
Additionally, in the absence of detailed data on the gear characteristics of each fishing trip (as in this study), an analysis based on landings profile does not account explicitly for gear specification. Hence, métiers were defined solely based on landings profiles (which were assumed to be representative of target species) and fishing grounds, and it was assumed that each métier defined corresponded to a specific setting of the gear.
The multivariate approaches used to define landings profiles are inherently descriptive, and the clusters of fishing trips cannot be determined exclusively by statistical criteria, but analysis has to be supplemented by a number of arbitrary choices (Ulrich and Andersen, 2004, and references therein). A clear distinction between two profiles of landings, i.e. two potential groups of target species, is not always easy, because there is often a smooth transition between clusters. Some fishing trips may be attributed equally to two groups or may constitute an intermediate group. Defining the threshold in a cluster analysis of fishing trips to group the trips to homogenous clusters is not straightforward and may be variable in both time and space, because species assemblages vary according to stock distribution and dynamics.
Although landings profiles have been used extensively to define métiers, scientists have not agreed upon a unique multivariate method for métier definition. There is a great variety of differing methods for this in the literature (Lewy and Vinther, 1994; He et al., 1997; Pelletier and Ferraris, 2000; Ulrich and Andersen, 2004; Campos et al., 2007; Marchal, 2008). Depending on the multivariate approach, data transformation, dissimilarity measure, and linkage type in cluster analysis, decision criteria for the choice of dissimilarity threshold in the resulting dendrogram, and other choices when analysing fishing trip data, slightly different conclusions may be reached.
For all the reasons listed above, the métiers identified in this study have to be characterized as potential in the sense that further verification based on carefully designed interviews with fishers on a national scale, covering all local fisheries, would be desirable to finalize their identification. However, in the absence of such information, landings profiles are an inexpensive and readily available (through EC Data Collection Regulation national programmes) source of data for a preliminary definition of métiers. Identifying métiers from landings profiles requires care, and expert knowledge is often necessary to decide upon their final level of aggregation. The use of values of landings instead of landings profiles might prove to be an improvement of the methodology for métier identification, because the target species and hence the métier choice depend largely on market value. Species with a relatively small proportion of the total catch but a high market value might be the actual target species instead of the most abundant species in the catches. However, more data (that are not currently collected under the framework of the DCR) are needed to apply such an approach.
Nevertheless, the identification of métiers from landings profiles could promote classification and definition of fishing activities, a key issue in fisheries management. In recent years, there has been growing interest within the fisheries science community in the use of fleet-/area- and fisheries-based models to represent and analyse the short-term dynamics of fishing effort in response to regulation. In particular, attention has been devoted to representing explicitly the spatial and temporal evolution of harvesting, and its determinants, where fleets can change fishing techniques/target species and move across different areas. This interest has arisen largely from the debate on the potential impacts of technical measures on the economic and ecological status of fisheries.
Our study represents a modest first attempt to identify longline métiers over a wide area of the eastern Mediterranean. As such, its findings may have practical implications. In the absence of TACs or quotas in the Mediterranean, the management system depends heavily on technical measures and effort-control programmes, including temporal/spatial closures. To this end, the management of specific métiers might help to protect certain life cycles/species. For instance, by managing either spatially or temporally the métiers targeting small sea breams, such as AEG-LLS-5 and AEG-LLS-8, it might help to rebuild a depleted stock.
Management measures are regularly subject to change and modification, but it is often difficult to anticipate how fleets and fishers will respond to them. Historically, management measures have often had unexpected consequences, e.g. closures leading to undesirable effort redistribution. Therefore, improved knowledge of the set longline métiers acquired here could, we believe, prove useful in understanding how fishers will adapt their behaviour and survival strategies under various management systems and incentives.
In conclusion, this study was based on a large (compared with previous analyses) and well-distributed sample among all fishing areas off Greece, and identified the main métiers on a national level. It is hoped that the findings will contribute towards a better understanding of the set longline operation in Greece and will be a useful step in providing scientific advice needed for the future management of Mediterranean fisheries.
Acknowledgements
The study was carried out with financial support from the Commission of the European Communities, specific FP6 RTD programme “Specific Support to Policies”, SSP-2006-044168-AFRAME (A Framework for Fleet- and Area-based Fisheries Management). The results do not necessarily reflect the views and in no way anticipates the future policy of the European Commission. Two anonymous reviewers and the editor made useful comments that helped us improve the quality of the manuscript.