-
PDF
- Split View
-
Views
-
Cite
Cite
Victoria Cairns, Jon Godwin, Post-Lyme borreliosis syndrome: a meta-analysis of reported symptoms, International Journal of Epidemiology, Volume 34, Issue 6, December 2005, Pages 1340–1345, https://doi.org/10.1093/ije/dyi129
Close - Share Icon Share
Abstract
Background This meta-analysis compares the prevalence of fatigue, musculoskeletal pain, and neurocognitive difficulties in patients who have had Lyme borreliosis (LB) and control subjects without LB.
Methods Titles and abstracts in PubMed were reviewed for studies with data on the symptoms listed above that compared patients who had had LB with controls from the general population. Five studies with 504 patients and 530 controls were included in the meta-analysis.
Results The prevalence of symptoms was significantly higher in the LB patients, with P-values between <0.00001 and 0.007 for 8 of the 10 symptoms in the three categories listed above. The higher prevalence of certain neurocognitive symptoms but not others, in the same pattern as reported in the literature, is further confirmation of this syndrome. The pattern of symptoms appears to be different from that seen in fibromyalgia, depression, and chronic fatigue syndrome.
Conclusions This meta-analysis provides strong evidence that some patients with LB have fatigue, musculoskeletal pain, and neurocognitive difficulties that may last for years despite antibiotic treatment.
There is increasing evidence that some patients with the bacterial infection Lyme borreliosis (LB) have symptoms that persist for years afterwards, despite having received the recommended antibiotic treatment. The main symptoms of fatigue, musculoskeletal pain, and neurocognitive difficulties1 are attributed to a post-LB syndrome, also known as post-treatment chronic Lyme disease. Nevertheless, some still question whether such symptoms can be a direct consequence of LB, or believe the symptoms are simply from depression following an acute infection. They are sceptical owing to the long-term, indistinct nature of the symptoms and because some studies did not show a statistically significantly greater prevalence of symptoms in patients who have had LB. All studies so far have been relatively small, so this meta-analysis was performed to clarify the issue.
Methods
A search through PubMed was made up to October 18, 2004. Six studies2–7 were found that (1) compared patients after LB with controls without LB from the general population and (2) recorded data on the simple frequency of symptoms of fatigue, musculoskeletal pain, and neurocognitive difficulties. Four other studies8–11 were found comparing patients after LB with controls from the general population, but no data on the above symptoms were collected in these studies.
The mean or median time since onset or diagnosis of LB was 3–6 years in five of the studies and ∼15 years in the sixth study. A meta-analysis was performed on the five studies (Table 1). The sixth study was not included because it was carried out much later after the onset of LB. The frequencies of symptoms reported by the patients and controls were taken directly from the publications. All data were included for fatigue (one symptom), musculoskeletal pain (three symptoms: joint or muscle pain, muscle aches, and swollen joints), and neurocognitive difficulties (six symptoms: memory problems, poor concentration, difficulties in formulating ideas, difficulties in word finding, difficulties in judgement, and difficulties in naming objects). Analyses were done separately for each symptom.
Studies included in the meta-analysis
| Study . | No. of patients/No. of controls . | Patients . | Controls . | Age . | Time since onset or diagnosis of LB . |
|---|---|---|---|---|---|
| Seltzer et al.2 | 212/212 | LB cases selected from public health department records | Matched for age and area of residence (1:1) | 71% adults, median 45 years; 29% children, median 8 years | Median ∼4.3 years |
| Shadick et al.3 | 38/43 | LB cases, met CDCa case definition13 | Same geographic area | Adults: mean 49 years in patients, 49 years in controls | Mean 6.2 years |
| Shadick et al.4 | 186/167 | LB cases, met CDC case definition | Same geographic area, balanced by age tertile and sex | Adults: mean 48 years in patients, 50 years in controls | Mean 6.0 years |
| Vazquez et al.5 | 43/86 | Facial nerve palsy attributed to LB; met CDC case definition | Matched for age and geographic area (1:2) | Children: median 14 years | Mean 4.1 years |
| Wang et al.6 | 25/26 | LB cases, met CDC case definition | Same geographic area, balanced by age group and sex | Children: age ≤16 years | Mean 3.2 years |
| Study . | No. of patients/No. of controls . | Patients . | Controls . | Age . | Time since onset or diagnosis of LB . |
|---|---|---|---|---|---|
| Seltzer et al.2 | 212/212 | LB cases selected from public health department records | Matched for age and area of residence (1:1) | 71% adults, median 45 years; 29% children, median 8 years | Median ∼4.3 years |
| Shadick et al.3 | 38/43 | LB cases, met CDCa case definition13 | Same geographic area | Adults: mean 49 years in patients, 49 years in controls | Mean 6.2 years |
| Shadick et al.4 | 186/167 | LB cases, met CDC case definition | Same geographic area, balanced by age tertile and sex | Adults: mean 48 years in patients, 50 years in controls | Mean 6.0 years |
| Vazquez et al.5 | 43/86 | Facial nerve palsy attributed to LB; met CDC case definition | Matched for age and geographic area (1:2) | Children: median 14 years | Mean 4.1 years |
| Wang et al.6 | 25/26 | LB cases, met CDC case definition | Same geographic area, balanced by age group and sex | Children: age ≤16 years | Mean 3.2 years |
Centers for Disease Control and Prevention.
Studies included in the meta-analysis
| Study . | No. of patients/No. of controls . | Patients . | Controls . | Age . | Time since onset or diagnosis of LB . |
|---|---|---|---|---|---|
| Seltzer et al.2 | 212/212 | LB cases selected from public health department records | Matched for age and area of residence (1:1) | 71% adults, median 45 years; 29% children, median 8 years | Median ∼4.3 years |
| Shadick et al.3 | 38/43 | LB cases, met CDCa case definition13 | Same geographic area | Adults: mean 49 years in patients, 49 years in controls | Mean 6.2 years |
| Shadick et al.4 | 186/167 | LB cases, met CDC case definition | Same geographic area, balanced by age tertile and sex | Adults: mean 48 years in patients, 50 years in controls | Mean 6.0 years |
| Vazquez et al.5 | 43/86 | Facial nerve palsy attributed to LB; met CDC case definition | Matched for age and geographic area (1:2) | Children: median 14 years | Mean 4.1 years |
| Wang et al.6 | 25/26 | LB cases, met CDC case definition | Same geographic area, balanced by age group and sex | Children: age ≤16 years | Mean 3.2 years |
| Study . | No. of patients/No. of controls . | Patients . | Controls . | Age . | Time since onset or diagnosis of LB . |
|---|---|---|---|---|---|
| Seltzer et al.2 | 212/212 | LB cases selected from public health department records | Matched for age and area of residence (1:1) | 71% adults, median 45 years; 29% children, median 8 years | Median ∼4.3 years |
| Shadick et al.3 | 38/43 | LB cases, met CDCa case definition13 | Same geographic area | Adults: mean 49 years in patients, 49 years in controls | Mean 6.2 years |
| Shadick et al.4 | 186/167 | LB cases, met CDC case definition | Same geographic area, balanced by age tertile and sex | Adults: mean 48 years in patients, 50 years in controls | Mean 6.0 years |
| Vazquez et al.5 | 43/86 | Facial nerve palsy attributed to LB; met CDC case definition | Matched for age and geographic area (1:2) | Children: median 14 years | Mean 4.1 years |
| Wang et al.6 | 25/26 | LB cases, met CDC case definition | Same geographic area, balanced by age group and sex | Children: age ≤16 years | Mean 3.2 years |
Centers for Disease Control and Prevention.
The observed minus the expected number of patients with symptoms in each study was calculated and weighted by the inverse of the sampling variance using the Peto method in a modification of the Mantel–Haenszel method.12 All P-values were two-tailed (2P). Heterogeneity between the studies was assessed using a χ2-test.
The prevalence of the symptoms was higher in LB patients in 28 of the 33 comparisons within the studies but, with the relatively small sample sizes, not all P-values fell below 0.05. To account for the multiple testing and preserve the overall a level of 0.05 in the meta-analysis, the symptom with the smallest P-value was tested at a level 0.05/10 (the total number of tests), the symptom with the next smallest P-value at 0.05/9, etc, proceeding only if the P-value fell below the limit.
The subjects in the five studies in the meta-analysis had been randomly selected from patient registers or from telephone or address lists with subsequent identification of LB status (in one study5, all patients found had been included). The subjects in two of the five studies4,6 had been first approached by mail, while the subjects in the other three studies had been contacted directly by telephone. In all five studies, once contact had been made, some subjects had declined to participate. All five studies were performed in the USA. Given the different geographic locations and different age groups, it is likely that the only overlap of subjects between the studies was that reported by Vazquez et al.5 with the study by Seltzer et al.2 (∼2–3 LB patients). The meta-analysis was repeated excluding the study by Vazquez et al.5
Of the five studies two were on adults,3,4 two on children,5,6 and one on adults and children.2 The patients in one study5 were selected because they had had facial nerve palsy attributed to LB. The patients in the other four studies had had a diagnosis of LB without further specification. The diagnoses of LB had been substantiated in four of the studies3–6 to see whether they met the CDC case definition,13 where appropriate by review of the patients' medical records. In three of the studies,3,5,6 all the LB patients had received antibiotic treatment. In the study by Seltzer et al.,2 6.7% of the patients had not received treatment, and for 7.7% of the patients the treatment was not known. In the study by Shadick et al.,4 4.8% had not received treatment, and for 12.9% the treatment was not known. All except one of the studies5 were performed before treatment guidelines were published by the Infectious Diseases Society of America.14
Results
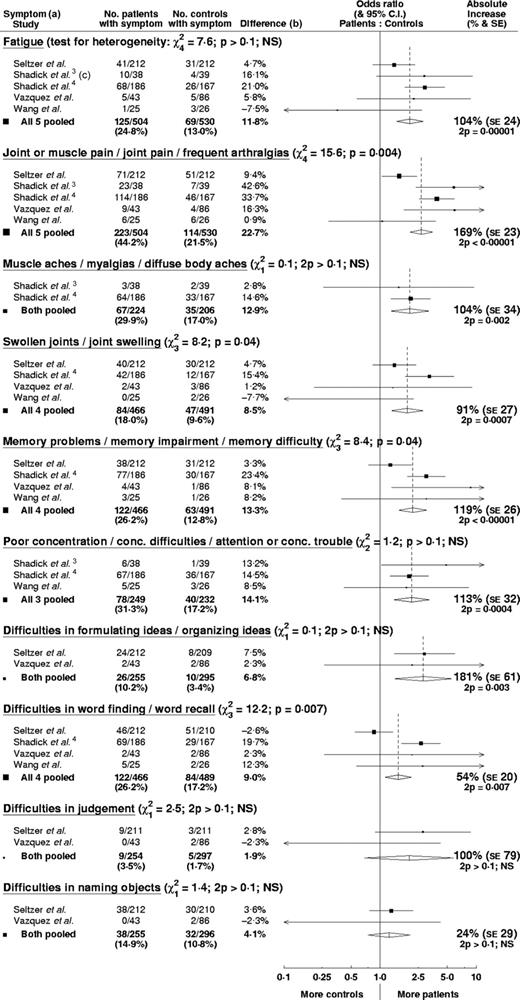
After pooling the data in the meta-analysis (Figure 1), fatigue and all three musculoskeletal symptoms (joint or muscle pain, muscle aches, and swollen joints) were significantly more frequent in patients who had had LB than controls, with low P-values between 2P < 0.00001 and 0.002. Of the six neurocognitive symptoms, four were significantly more frequent in the LB patients (memory problems, poor concentration, difficulties in formulating ideas, and difficulties in word finding), with low P-values between 2P < 0.00001 and 0.007. A significantly higher prevalence was not seen in the other two neurocognitive symptoms (difficulties in judgement and difficulties in naming objects).
Meta-analysis comparing patients after Lyme borreliosis with controls. Open diamonds represent pooled results with 95% confidence interval. Areas of closed squares are proportional to the amount of information contributed. (a) Symptoms as described in original publications. (b) Prevalence in patients minus prevalence in controls. (c) Data for ‘unusual fatigue’
All five studies showed a higher prevalence of symptoms in LB patients, but there was some heterogeneity between the studies. This is seen in the underlying prevalence in the control groups (probably owing to the different age structures) and in the difference in prevalence between patients and controls. Higher differences in prevalence are seen in studies that (i) had a higher proportion of patients with neurological symptoms in the acute phase,3–5 (ii) were run in earlier years when diagnosis and treatment were more likely to have been delayed3,4 (both reported to be associated with persistent symptoms), or (iii) did not include children.3,4 There are some claims that children are less likely to have persistent symptoms. One of the five studies5 comprised a subset of LB patients—children with facial nerve palsy attributed to LB. Excluding this study did not alter the results.
Significant heterogeneity was seen for four of the symptoms (joint or muscle pain, swollen joints, memory problems, and difficulties in word finding), but a possible explanation for the observed differences is given above. The heterogeneity tended to be quantitative rather than qualitative, i.e. the studies varied more in the extent of the increased prevalence in patients than in whether there was an increased prevalence. The meta-analysis P-value still provides a meaningful test of the null hypothesis that there was no increased prevalence in the studies. Despite the heterogeneity between the studies, overall the increase in prevalence of symptoms of fatigue, musculoskeletal pain, or neurocognitive difficulties in patients who have had LB appears to be (breve)5%.
Discussion
This meta-analysis provides strong evidence that some patients have fatigue, musculoskeletal pain, and neurocognitive difficulties that persist for years after LB. The pattern seen in the meta-analysis of an increased prevalence of certain neurocognitive difficulties but not others is the same as that seen in other studies on post-LB syndrome. Memory problems, particularly verbal memory, have been observed in many other studies.8,15–20 Poor concentration, particularly auditory attention, has been noted in some studies,16,19,20 but not others.8,15 Diminished mental flexibility8 has also been seen, which is similar to difficulties in formulating ideas. In contrast, no increased difficulties in intellectual and problem solving skills,8 global intellectual functioning,15 or conceptual thinking15 were seen, which is consistent with the lack of increased difficulties in judgement seen in the meta-analysis. Also, no report in other studies of difficulties in naming objects was found. In confirmation of this pattern, Fallon et al.21 observed significantly reduced blood flow in certain white matter areas of the brain in patients with post-LB syndrome compared with healthy subjects, and the flow reductions were significantly associated with deficits in memory and visuospatial organization.
The possibility of bias in these studies must be considered. For example, LB patients with symptoms may be more likely than LB patients without symptoms to agree to participate in a study of chronic illness. However, even if the denominators for the patient groups are increased to include the non-participants and it is assumed that the non-participants had none of the symptoms, the results are scarcely altered. Furthermore, Shadick et al.4 noted in a substudy that the controls who declined to participate did not differ from the patients who declined to participate in that they were also less likely to have symptoms. Therefore, selection bias does not appear to be the explanation for the consistent results.
There is also no evidence of a reporting bias, since the available data from physical examinations and neurocognitive tests confirm the subjective reports. Physical examinations were carried out in three of the studies. Shadick et al.3 detected more abnormal joint test results on the American College of Rheumatology Glossary joint index in patients (2P = 0.02). Shadick et al.4 observed that knee, hip, and proximal interphalangeal pain on motion and knee swelling were seen more often in patients than controls (all 2P < 0.10). Physical examinations in the study by Wang et al.6 identified the same number of controls with joint pain and the same number of patients and controls with swollen joints as had been reported, but revealed that two more patients had joint pain than had been reported.
Two of the studies compared patients and controls using objective neurocognitive tests. Shadick et al.3 reported that significantly more patients had abnormal test results than controls (12/38 vs 5/43, 2P = 0.01) and that the patients had more deficits on average in all the neurocognitive tests (2 out of 13 P-values were below 0.05). However, Shadick et al.4 reported that the proportion of patients and controls with an abnormal performance did not differ significantly. No details were provided. They used a less restrictive definition of abnormal, and with their new definition ∼25% of the controls may have been labelled ‘abnormal’. If 5% (or 10%) of patients have neurocognitive difficulties following LB, then a study this size will have only 10% (or 30%) power to detect this using their definition. Therefore, without further details, little can be concluded from this study with regard to the neurocognitive testing.
Finally, non-significant results may be less likely to be published, resulting in a publication bias. However, many of these authors concluded that the differences between patients and controls were not significant, so a bias owing to missing non-significant studies seems unlikely.
There have been claims that children are less likely to have persistent symptoms after LB, and that the pattern of persistent symptoms differs in children, but there are few comparative studies. In the two relatively small studies5,6 exclusively on children in this meta-analysis, a higher proportion of LB patients had symptoms than controls in most of the 12 comparisons on the first eight symptoms in Figure 1, and in six comparisons the prevalence was >5% higher in LB patients. The meta-analysis could not be run separately for adults and children since the results were not available separately for adults and children in the one study including subjects of all ages.2 More investigation of this issue is needed.
The pattern of symptoms reported following LB appears to be different from that seen in fibromyalgia, depression, and chronic fatigue syndrome. The fatigue following LB has been variously described in the literature as profound, notable, unusual, debilitating, and extreme, not as a vague symptom of tiredness. The musculoskeletal pain in this syndrome is often roving, asymmetrical pain in the limbs, whereas fibromyalgia is characterized by widespread, symmetrical pain at many sites over the whole body. Kaplan et al.18 observed that patients with LB encephalopathy were more impaired on standardized memory tests than patients with either fibromyalgia or depression, while patients with fibromyalgia scored significantly higher than both other groups on the scale for somatic concerns, and the depressed patients scored significantly higher on the scales for depression and anxiety. They reported that the subjective memory difficulties in the patients with fibromyalgia and depression were primarily due to psychological distress. Gaudino et al.16 compared patients with post-LB syndrome and persistent severe fatigue with patients with chronic fatigue syndrome. They found that, despite symptom overlap, the patients with post-LB syndrome showed significantly more global cognitive impairment in neurocognitive tests. Regarding the emotions, Elkins et al.22 observed lower positive affect (emotions such as joy, enthusiasm, and pleasure) but normal negative affect (emotions such as sadness, hopelessness, and guilt) in patients with post-LB syndrome. Positive affect is likely to be dampened by profound fatigue. Raised negative affect, a key component of depression, was not reported.
This meta-analysis shows very clearly that there is a connection between LB and a distinct set of persistent symptoms known as post-LB syndrome. Ongoing infection has not been excluded as a possible explanation, but, in the screening of over 1800 patients for one study on post-LB syndrome, no patient was found to have a positive PCR assay or culture for borrelia.23 Steere et al.24 wrote ‘it is hypothesized that Borrelia burgdorferi may trigger immunologic or neurohormonal processes in the brain that cause persistent pain, neurocognitive, or fatigue symptoms, despite spirochaetal killing with antibiotic therapy’. Klempner et al.25 and Kaplan et al.26 found that three more months of antibiotic treatment after the recommended antibiotic treatment did not improve symptoms or cognitive function more than placebo. In a similar study, on patients with profound fatigue persisting after treatment for LB, Krupp et al.27 also found that additional antibiotic treatment did not improve cognitive function more than placebo. They observed a significantly greater improvement in fatigue in treated patients, to a larger extent in those who had previously only received oral antibiotics and were receiving intravenous antibiotics for the first time in their study.
There are roughly 120 000 new cases of LB every year in Europe28,29 and 150 000 every year in the USA30,31 (after taking into account the estimated under-reporting in the USA32,33). If 5% of LB patients have symptoms persisting for years as a direct consequence of the LB in both Europe and the USA, then the combined incidence of post-LB syndrome is over 13 000 per year, and the prevalence of patients will be several times higher. Few published data are available on the course of this disorder, but anecdotal reports indicate that many patients recover very slowly over years, without further treatment. Further research is needed focusing on the aetiology of the disorder and a search for effective therapies.
Disclaimer
I (VC) was employed for many years in clinical research at Aventis (formerly Hoechst). Aventis (now Sanofi Aventis) develops and produces antibiotics that are used in the treatment of LB. I am now working as a consultant for Boehringer Ingelheim in the clinical development of medication for the treatment of depression, pain associated with depression, fibromyalgia, diabetic peripheral neuropathic pain, and generalized anxiety disorder. However, there does not appear to be any conflict of interest as no medication has been shown to be effective in resolving the problems of post-LB syndrome.
I have had LB with persistent symptoms and this led to my interest in the topic. It is still debated whether there is such a thing as post-LB syndrome, or chronic LB. A meta-analysis of the available data, therefore, seems to be exactly what is needed.
This meta-analysis provides strong evidence that some patients with Lyme borreliosis have fatigue, musculoskeletal pain, and neurocognitive difficulties that may last for years.
The higher prevalence seen in these patients of certain neurocognitive symptoms but not others, in the same pattern as reported in the literature, is further confirmation of the syndrome.
The pattern of persistent symptoms appears to be different from that seen in fibromyalgia, depression, and chronic fatigue syndrome.
References
Seltzer EG, Gerber MA, Cartter ML, Freudigman K, Shapiro ED. Long-term outcomes of persons with Lyme disease.
Shadick NA, Phillips CB, Logigian EL et al. The long-term clinical outcomes of Lyme disease.
Shadick NA, Phillips CB, Sangha O et al. Musculoskeletal and neurologic outcomes in patients with previously treated Lyme disease.
Vazquez M, Sparrow SS, Shapiro ED. Long-term neuropsychologic and health outcomes of children with facial nerve palsy attributable to Lyme disease.
Wang TJ, Sangha O, Phillips CB et al. Outcomes of children treated for Lyme disease.
Kalish RA, Kaplan RF, Taylor E, Jones-Woodward L, Workman K, Steere AC. Evaluation of study patients with Lyme disease. 10–20-year follow-up.
Benke T, Gasse T, Hittmair-Delazer M, Schmutzhard E. Lyme encephalopathy: long-term neuropsychological deficits years after acute neuroborreliosis.
Adams WV, Rose CD, Eppes SC, Klein JD. Cognitive effects of Lyme disease in children.
Sangha O, Phillips CB, Fleischmann KE et al. Lack of cardiac manifestations among patients with previously treated Lyme disease.
Shotland LI, Mastrioanni MA, Choo DL et al. Audiologic manifestations of patients with post-treatment Lyme disease syndrome.
Wharton M, Chorba TL, Vogt RL, Morse DL, Buehler JW. Centers for disease control and prevention. Case definitions for public health surveillance.
Yusuf S, Peto R, Lewis J, Collins R, Sleight P. Beta-blockade during and after myocardial infarction: an overview of the randomized trials.
Wormser GP, Nadelman RB, Dattwyler RJ et al. Practice guidelines for the treatment of Lyme disease. Guidelines from the Infectious Diseases Society of America.
Krupp LB, Masur D, Schwartz J et al. Cognitive functioning in late Lyme borreliosis.
Gaudino EA, Coyle PK, Krupp LB. Post-Lyme syndrome and chronic fatigue syndrome. Neuropsychiatric similarities and differences.
Kaplan RF, Jones-Woodward L, Workman K, Steere AC, Logigian EL, Meadows ME. Neuropsychological deficits in Lyme disease patients with and without other evidence of central nervous system pathology.
Kaplan RF, Meadows ME, Vincent LC, Logigian EL, Steere AC. Memory impairment and depression in patients with Lyme encephalopathy: comparison with fibromyalgia and nonpsychotically depressed patients.
Bujak DI, Weinstein A, Dornbush RL. Clinical and neurocognitive features of the post Lyme syndrome.
Tager FA, Fallon BA, Keilp J, Rissenberg M, Jones CR, Liebowitz MR. A controlled study of cognitive deficits in children with chronic Lyme disease.
Fallon BA, Keilp J, Prohovnik I, Heertum RV, Mann JJ. Regional blood flow and cognitive deficits in chronic Lyme disease.
Elkins LE, Pollina DA, Scheffer SR, Krupp LB. Psychological states and neuropsychological performances in chronic Lyme disease.
Weinstein A, Klempner MS. Treatment of patients with persistent symptoms and a history of Lyme disease.
Klempner MS, Linden TH, Evans J et al. Two controlled trials of antibiotic treatment in patients with persistent symptoms and a history of Lyme disease.
Kaplan RF, Trevino RP, Johnson GM et al. Cognitive function in post-treatment Lyme disease. Do additional antibiotics help?
Krupp LB, Hyman LG, Grimson R et al. Study and treatment of post Lyme disease (STOP-LD): a randomized double masked clinical trial.
German National Reference Centre for Borreliosis. Lyme Borreliosis [in German]. Available at: http://pollux.mpk.med.uni-muenchen.de/alpha1/nrz-borrelia/lb (Accessed November 25, 2004).
O'Connell S, Granstroem M, Gray JS, Stanek G. Epidemiology of European Lyme borreliosis.
Orloski KA, Hayes EB, Campbell GL, Dennis DT. Surveillance for Lyme disease—United States 1992–1998. Centers for Disease Control and Prevention Surveillance Summaries.
Centers for Disease Control and Prevention. Lyme disease—United States, 2001–2002.
Meek JI, Roberts CL, Smith EV Jr, Cartter ML. Underreporting of Lyme disease by Connecticut physicians, 1992.