-
PDF
- Split View
-
Views
-
Cite
Cite
Quentin Bodard, Frederic Roca, Baptiste Dilly, Druesne Laurent, Philippe Chassagne, Acute cardiac failure secondary to senile systemic amyloidosis, Age and Ageing, Volume 45, Issue 6, 2 November 2016, Pages 908–909, https://doi.org/10.1093/ageing/afw073
Close - Share Icon Share
Abstract
Hereditary or senile transthyretin (TTR) cardiac amyloidosis is a rare and underestimated cause of heart failure in old patients. New diagnostic methods, particularly cardiac MRI and proteomic analysis, have been recently developed that enable both earlier identification and development of specific treatments.
We report a case of cardiac amyloidosis revealed through late-onset heart failure.
Case report
A 93-year-old woman was hospitalised for a first acute cardiac decompensation. Her past history revealed hypertension, type II diabetes and age-related macular degeneration with blindness.
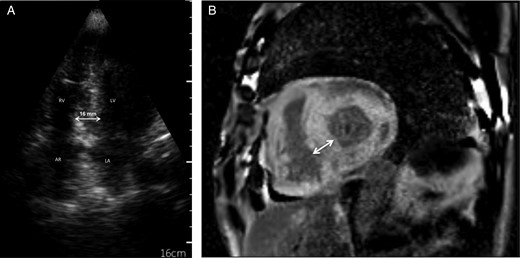
(A) Cardiac image Section 4 cavities of an ultraportable screening echocardiograph: thickening of the interventricular septum measured at 16 mm with a grained aspect in the ‘brightness’ of the myocardium. LV, left ventricle; RV, right ventricle; LA, left atrium; AR, right atrium; (B) Cardiac MRI section short-axis T1 sequence with injection of gadolinium: late diffuse circumferential enhancement of the myocardium with an interventricular septum measured at 22 mm.
A cardiac MRI showed late circumferential sub-endocardial enhancement with interventricular septal thickness (22 mm) (Figure 1).
The routine biopsy (without xerostomia) of minor salivary glands showed birefringent deposits in polarised light with Congo red staining. These deposits were resistant to permanganate that elinimated a diagnosis of AA amyloidosis. Immunohistochemistry did not provide complementary results, because no proteomic analysis has been carried out. Immunoelectrophoresis of the proteins and research for light chains were negative in blood and urine. These features suggested senile or hereditary TTR cardiac amyloidosis. In addition to the symptomatic treatment of her heart failure, no specific treatment (such as TTR stabiliser) for this cardiac amyloidosis was initiated, according to her age and functional status.
The three main types of amyloidosis responsible for heart disease are AL amyloidosis (deposits of light chains of monoclonal immunoglobulin), hereditary TTR amyloidosis (associated with an autosomal dominant genetic mutation) and senile TTR amyloidosis (non-mutated). Senile TTR amyloidosis is usually diagnosed in late life, predominantly in male patients, and would have a better prognosis [1]. AA amyloidosis (secondary to chronic inflammatory diseases where deposits of serum amyloid A (SAA) proteins are present) represents only 2% of cardiac amyloidosis cases.
The clinical signs that reveal the condition are not specific and suggest a hypertrophic heart disease with a preserved ejection fraction. The diagnosis of cardiac amyloidosis should be suspected when microvoltage or conduction disorders on the electrocardiogram are present, associated with a myocardial ‘brightness’ aspect and thickening of the interventricular septum (>12 mm) on the echocardiogram [1, 2].
Because myocardial biopsies are rarely carried out routinely in very old patients, the diagnosis is based on the data obtained from the cardiac MRI, combined with a more accessible organ biopsy such as of the minor salivary glands.
The amyloid deposits are visible with polarised light after staining with Congo red, and immunohistochemistry defines the type of amyloidosis (anti-light chain, anti-SAA or anti-TTR antibodies). Recent proteomic analysis makes now possible amyloidosis characterisation in >95% of cases [3].
In addition, it is essential to search for a monoclonal component in the blood and urine to avoid AL amyloidosis whose prognosis is worst and requires specific treatments.
Cardiac imaging techniques including 99mTc-DPD scintigraphy and especially MRI make possible an indirect diagnosis of cardiac amyloidosis with high sensitivity and specificity scores [1, 4]. In addition, cardiac MRI could differentiate AL from TTR forms [5].
The symptomatic treatment of heart failure is based on diuretics [1]. The use of β-blockers and digoxin should be cautious. Indeed these drugs may worsen dysautonomic disorders and be stored in amyloid deposits including an overdose risk. The stabilisers of the TTR tetramer decrease its degradation into a monomer and thus their intra-tissue deposits [1]. They are proposed in the treatment of the peripheral neuropathies of hereditary amyloidosis and are being evaluated in hereditary and senile cardiac disorders [6].
Senile TTR cardiac amyloidosis is an underestimated cause of heart failure in old patients.
Diagnosis of cardiac amyloidosis should be considered in older subject with late-onset heart disease without cardiac history.
Cardiac MRI and proteomic analysis improve characterisation of amyloidosis.
Stabilisers of the transthyretin tetramer are promising treatment in senile cardiac amyloidosis.
Authors’ contributions
All authors contributed to this paper.
Conflicts of interest
None declared.




Comments