-
PDF
- Split View
-
Views
-
Cite
Cite
Bruno Fogliani, Gildas Gâteblé, Matthieu Villegente, Isabelle Fabre, Nicolas Klein, Nicolas Anger, Carol C Baskin, Charlie P Scutt, The morphophysiological dormancy in Amborella trichopoda seeds is a pleisiomorphic trait in angiosperms, Annals of Botany, Volume 119, Issue 4, March 2017, Pages 581–590, https://doi.org/10.1093/aob/mcw244
Close - Share Icon Share
Abstract
Recent parsimony-based reconstructions suggest that seeds of early angiosperms had either morphophysiological or physiological dormancy, with the former considered as more probable. The aim of this study was to determine the class of seed dormancy present in Amborella trichopoda, the sole living representative of the most basal angiosperm lineage Amborellales, with a view to resolving fully the class of dormancy present at the base of the angiosperm clade.
Drupes of A. trichopoda without fleshy parts were germinated and dissected to observe their structure and embryo growth. Pre-treatments including acid scarification, gibberellin treatment and seed excision were tested to determine their influence on dormancy breakage and germination. Character-state mapping by maximum parsimony, incorporating data from the present work and published sources, was then used to determine the likely class of dormancy present in early angiosperms.
Germination in A. trichopoda requires a warm stratification period of at least approx. 90 d, which is followed by endosperm swelling, causing the water-permeable pericarp–mesocarp envelope to split open. The embryo then grows rapidly within the seed, to radicle emergence some 17 d later and cotyledon emergence after an additional 24 d. Gibberellin treatment, acid scarification and excision of seeds from the surrounding drupe tissues all promoted germination by shortening the initial phase of dormancy, prior to embryo growth.
Seeds of A. trichopoda have non-deep simple morphophysiological dormancy, in which mechanical resistance of the pericarp–mesocarp envelope plays a key role in the initial physiological phase. Maximum parsimony analyses, including data obtained in the present work, indicate that morphophysiological dormancy is likely to be a pleisiomorphic trait in flowering plants. The significance of this conclusion for studies of early angiosperm evolution is discussed.
INTRODUCTION
Seed dormancy can be defined as the retardation of radicle emergence (germination) under otherwise favourable conditions for germination (Bewley et al., 2015). There are five major classes of seed dormancy: morphological, physiological, morphophysiological, physical and combinational, within each of which further sub-classes may be recognized (Baskin and Baskin, 2004). Among the five major classes of dormancy, physiological dormancy (PD) is characterized by the presence in the mature seed of a fully developed embryo, which takes >30 d following imbibition before the radicle emerges. In contrast, morphological dormancy (MD) is characterized by the presence in the mature seed of an incompletely developed embryo that, under appropriate conditions, begins to grow immediately following imbibition, but takes <30 d to complete development before the radicle emerges. In morphophysiological dormancy (MPD), a combination of PD and MD is present, delaying germination for >30 d.
As dormancy is a key trait for survival and fitness (Linkies et al., 2010), the reconstruction of the evolution of dormancy mechanisms is of particular interest. In the angiosperms, such reconstruction work is now possible as studies performed over the last approx. 15 years (summarized at http://www.mobot.org/MOBOT/research/APweb/; Byng et al., 2016) have provided a solid phylogenetic framework. According to the majority of phylogenetic studies, the orders Amborellales, Nymphaeales and Austrobaileyales, collectively termed the ANA grade, diverged sequentially from a remaining lineage from which all other living angiosperms, the euangiosperms, are descended. A slightly different evolutionary hypothesis, which has emerged from a few analyses (Qiu et al., 2001, 2010; Xi et al., 2014), places Amborellales and Nymphaeales together in a first-diverging angiosperm clade, with Austrobaileyales as a second-diverging lineage, sister to euangiosperms. The euangiosperms are composed of the major groups of eudicots and monocots, in addition to the moderately sized magnoliid clade and the two smaller taxa of Chloranthaceae and Ceratophyllum.
As a result of the largely successful resolution of angiosperm phylogeny, analyses of the basally diverging ANA-grade lineages have been particularly informative for the reconstruction of character states at the base of the flowering plants. The likely earliest diverging ANA-grade order, Amborellales, contains Amborella trichopoda Baill. as its only living representative. This species is a dioecious shrub, endemic to the understorey of humid tropical forests on the southern Pacific island of New Caledonia. The mature A. trichopoda embryo is short and broad (Bailey and Swamy, 1948; Tobe et al., 2000), suggesting the possible presence of MD. Fourcade et al. (2015) refer to unpublished studies of germination and dormancy in this species, though no detailed work has previously been undertaken.
The likely second-earliest diverging ANA-grade order, Nymphaeales, contains three aquatic families, Nymphaeceae, Cabombaceae and Hydatellaceae, of which the latter is indicated by molecular phylogeny to be basally diverging (Saarela et al., 2007). Physiological dormancy has been reported in the Nymphaeaceae genera Nymphaea (six species studied) and Nuphar (one species studied) and in the Cabombaceae genus Cabomba (one species studies), as summarized by Willis et al. (2014). In contrast, seeds of Trithuria (three species studied), the only genus of Hydatellaceae, have MPD (Tuckett et al., 2010). Interestingly, MPD in Trithuria contains an extreme morphological component as embryos in mature Trithuria seeds show very limited differentiation and consist of only about 18–23 cells, depending on the species (Tuckett et al., 2010; Friedman et al., 2012).
The third-diverging ANA-grade order, Austrobaileyales, contains three families of woody species, Austrobaileyaceae, Trimeniaceae and Schisandraceae. The Schisandraceae genera Schisandra (three species studied) and Illicium (five species studied) both have MPD (summarized by Willis et al., 2014), while dormancy in the remaining genera of Austrobaileyales has yet to be assessed.
A recent large-scale study of seed dormancy types (Willis et al., 2014) has collated data on 13 939 angiosperm and 297 gymnosperm species, and concluded the likely ancestral state of dormancy in the seed plants to be MPD. According to this study, the most recent common ancestor (MRCA) of the angiosperms analysed (which included some Nymphaeales and Austrobaileyales, but not Amborellales) had either MPD or PD. Similarly, the MRCA of euangiosperms (i.e. all living angiosperms, excluding the ANA grade) was not completely resolved between MPD and PD, though MPD was considered more likely.
In the present work, we focus on seed dormancy in the key ANA-grade species A. trichopoda, likely sister to all other living flowering plants. We perform detailed quantitative and microscopic observations on germinating seeds, coupled with studies of the water permeability of seed-covering layers and the effects on germination of the exogenous application of the plant hormone GA3. We conclude that the precise type of dormancy in A. trichopoda seeds corresponds to the non-deep simple level of MPD. We incorporate these data, together with recently published data on Hydatellaceae, into character-state reconstructions in the angiosperms and conclude from this analysis that the likely pleisiomorphic state of the living flowering plants was MPD.
MATERIALS AND METHODS
Fruit collection
Mature fruits of Amborella trichopoda were harvested during the season of natural dispersal from plants growing at an altitude of around 600 m on the Plateau de Dogny in the central part of Grande Terre (S21°37′21·6″, E165°52′06·3″), New Caledonia. One batch, collected on 8 July 2008 and sown 7 d later, was used for dissection and observations on embryo development, while a second batch was harvested on 23 December 2008 and used for germination experiments, starting on 15 January 2009. Empty drupes were removed under vacuum, and fleshy parts of the remaining drupes were removed by gently rubbing fruits on a sieve under running water prior to further studies. Drupes without fleshy parts (DWFP) were stored prior to use in plastic zip-lock bags at room temperature (25 ± 2 °C).
Physical characteristics of mature DWFP and seeds
To assess A. trichopoda for the possible presence of dormancy, we first studied the physical characteristics of its mature DWFP and seed sensu stricto. Immediately after harvesting four replicates of 100 DWFP and of 100 seeds sensu stricto extracted from the DWFP were weighed. Also, three replicates of 20 DWFP and 20 seeds sensu stricto were measured, and average dimensions were determined. The length and width of entire DWFP, as well as those that had been cut transversally or longitudinally, were measured using a dissecting microscope equipped with a micrometer. DWFP were mounted on scanning electron microscope specimen holders using double-sided carbon adhesive tape and carbon-metalized (10–15 nm), as described by Rabier et al. (2008). Observations and size measurements were made using a Philips XL 30 environmental scanning electron microscope. Moisture content of four replicates of 25 DWFP was determined gravimetrically (103 ± 2 °C for 17 ± 1 h), by the protocol of MacKay et al. (2002), observing standard procedures (ISTA, 1999).
Embryo growth and germination
As the physical characteristics of A. trichopoda seeds suggested the presence of dormancy, we conducted preliminary germination assays to evaluate directly the presence of dormancy and gain insights into the overall class of dormancy present. The embryo length:seed length (E:S) ratio was determined for 50 freshly matured DWFP collected in July and cut longitudinally. Fresh DWFP were placed in four sowing trays (25 DWFP per tray) on a 1:1 perlite:peat substrate, covered with a 4 mm layer of vermiculite and incubated at 25 ± 4 °C (corresponding to the average diurnal temperature in A. trichopoda’s natural habitat) on a greenhouse sowing table. Intact and longitudinally cut DWFP were photographed after 93 d, and the E:S ratio was determined from 20 DWFP that were beginning to break open.
DWFP permeability
To elucidate the basis of dormancy in A. trichopoda seeds, we tested the water permeability of the mature DWFP. The seed sensu stricto and pit (pericarp/sclerified mesocarp) from three replicates of 20 DWFP were weighed before and after soaking for 72 h in distilled water and means were compared using the Student t-test. Seeds were imbibed in 1 % (w/v) aqueous methylene blue dye and dissected to determine the path of water entry, as described by Orozco-Segovia et al. (2007).
Scarification treatment and removal of endocarp and hard layer of mesocarp
To investigate the mechanical role of the DWFP in A. trichopoda seed dormancy, we compared the time course of germination in intact DWFP, DWFP scarified by acid treatment, and seed sensu stricto that had been excised from all surrounding tissues. A total of 100 DWFP were scarified for 90 min in 10 mL of concentrated sulphuric acid, rinsed with abundant tap water and soaked in three consecutive distilled water baths until all soft tissue remnants had been removed. Scarified and non-treated (control) DWFP were sown and incubated as described above, but under conditions of 95 % relative humidity generated using a fog system. DWFP were monitored for hypocotyl emergence twice weekly for 1 year.
After removal of fleshy tissues, DWFP were surfaced sterilized with 2 % chlorine bleach for 5 min, then rinsed three times over a 5 min period in sterile distilled water. Extraction of the seed sensu stricto from the endocarp and the hard inner part of the mesocarp (pit) was performed using a thin scalpel blade and tweezers, being careful not to damage the seed coat or seed. After isolation, seeds sensu stricto were surface-sterilized under a laminar hood using 2 % (w/v) bleach for 5 min and rinsed three times in sterile distilled water. Sterile seeds were sown in Petri dishes on two layers of germination paper moistened with sterile distilled water. A total of 100 seeds were sown (25 per dish, four replicates) and incubated at 25 °C under continuous light. The criterion used for germination was radicle protrusion to a length of 1 mm.
Effects of gibberellic acid on germination
To investigate physiological dormancy in A. trichopoda seed, we used a discriminatory test which relies on the effect of gibberellin treatment; moderate concentrations of exogenous gibberellic acid (GA3) are known to promote germination in seeds showing non-deep PD, but not deep PD (Baskin and Baskin, 2004). Non-scarified DWFP were placed in Petri dishes on germination paper moistened with 0, 50, 100, 500 or 1000 ppm GA3. A total of 100 DWFP were sown for each treatment (25 DWFP per dish, four replicates) and incubated at 25 °C under continuous light, and examined weekly for radicle emergence. Time of germination was recorded at radicle protrusion, and means were compared using the Student t-test.
Test of epicotyl MPD
Four types of non-deep simple epicotyl MPD and four of deep simple epicotyl MPD have been recognized (Baskin and Baskin, 2014). To determine if seeds of A. trichopoda have epicotyl MPD, we measured the time necessary for cotyledon emergence, following radicle emergence. Ninety-eight DWFP were incubated for 24 h in water and 100 DWFP in 50 ppm GA3 (Sigma-Aldrich), before being sown on trays at high humidity, as described above, and observations were made on radicle and cotyledon emergence every 2 or 3 d.
To determine whether GA3 would promote cotyledon emergence (and thus whether a type of non-deep simple epicotyl MPD was present), GA3 (50 ppm, Sigma-Aldrich) dissolved in lanolin (anhydrous wool fat; Probiotec, Australia) was applied with a paintbrush to 5 mm long rootlets, sparing the tip, of the 38 seeds which had germinated from the group of 98 seeds incubated in water (above). This stage was reached 1–2 d before secondary rootlet formation, and approx. 11 d after radicle emergence. The normality of the data distribution within each treatment group was assessed using the Shapiro–Wilk normality test (α = 0·05). Statistical comparisons of the time between radicle and cotyledon emergence was made using the Welch test (α = 0·05) for normal distributions and the Mann–Whitney Wilcoxon test (α = 0·05) for non-normal distributions.
Character reconstruction by parsimony
Cladograms representing two alternative topologies for the base of angiosperm phylogeny were manually created in Newick format, incorporating information from the Angiosperm Phylogeny Group III Website (Byng et al., 2016) and from Xi et al. (2014), respectively, with additional data from Saarela et al. (2007) and Löhne et al. (2007). The internal phylogeny of Nymphaeaceae was simplified by representing Nymphaea, Victoria, Euryale and Ondinea as a polytomy as, according to the phylogeny of Löhne et al. (2007), Nymphaea is polyphyletic, one section being sister to Victoria + Euryale, while another includes Ondinea. Character states given in Tuckett et al. (2010), Willis et al. (2014) and obtained in the present work were mapped onto both cladeograms in Mesquite (Maddison and Maddison, 2015) using parsimony reconstruction.
RESULTS
Physical characteristics of mature DWFP and seeds
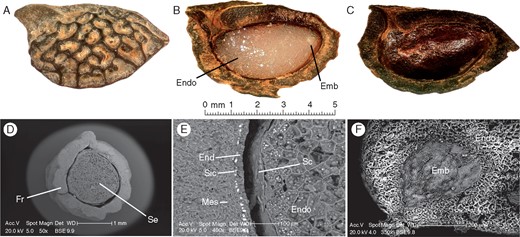
Physical characteristics of Amborella trichopoda seeds suggest the presence of dormancy. The mass of the A. trichopoda seed sensu stricto represented approx. 27 % of the mass of the mature DWFP, while seed length and width were 70 and 45 %, respectively, those of the mature DWFP (Table 1). The seed possessed a thin seed coat (Fig. 1B, E), surrounded by a hard, thick pit (Fig. 1A–D) composed of an endocarp and a sclerified layer from the mesocarp (Fig. 1D), previously described by Bobrov et al. (2005). Interestingly, X-ray microscopic analysis revealed an amorphous silicate-crystal layer located between these two parts of the pericarp (Fig. 1E). In freshly matured DWFP of A. trichopoda, the embryo, which was surrounded by a copious endosperm (Fig. 1B), was found to be arrested at the heart-shaped stage (Fig. 1F). At this stage, the E:S ratio was 0·08 ± 0·01.
Amborella trichopoda drupe without fleshly part (DWFP) structure. (A–C) Binocular microscopy, (D–F) scanning electron microscopy. (A) Whole DWFP. (B) Longitudinally cut DWFP. (C) An opened drupe showing the pit and seed sensu stricto. (D) Transverse section. (E) Magnified transverse section showing the meso-endocarp of the fruit separated by a silicate-crystal layer. (F) Longitudinal section of a seed sensu stricto showing the heart-shaped underdeveloped embryo (Emb, embryo; End, endocarp; Endo, endosperm; Fr, fruit parts; Mes, mesocarp; Sc, seed coat; Sic, silicate-crystal layer).
Characteristics of Amborella trichopoda drupes without fleshly parts (DWFP) and of seeds sensu stricto
| . | Average weight (g ± s.d.) . | Average length (mm ± s.d.) . | Average width (mm ± s.d.) . | Water content (% ± s.d.) . | E:S ratio . |
|---|---|---|---|---|---|
| For 100 DWFP | 2·15 ± 0·08 | 5·98 ± 0·24 | 3·49 ± 0·11 | 8·70 ± 0·06 | 0·08 ± 0·01 |
| For 100 seed sensu stricto | 0·58 ± 0·06 | 4,19 ± 0·99 | 1·57 ± 0·37 | 8·99 ± 0·65 |
| . | Average weight (g ± s.d.) . | Average length (mm ± s.d.) . | Average width (mm ± s.d.) . | Water content (% ± s.d.) . | E:S ratio . |
|---|---|---|---|---|---|
| For 100 DWFP | 2·15 ± 0·08 | 5·98 ± 0·24 | 3·49 ± 0·11 | 8·70 ± 0·06 | 0·08 ± 0·01 |
| For 100 seed sensu stricto | 0·58 ± 0·06 | 4,19 ± 0·99 | 1·57 ± 0·37 | 8·99 ± 0·65 |
E.S ratio, embryo length:seed length ratio.
Characteristics of Amborella trichopoda drupes without fleshly parts (DWFP) and of seeds sensu stricto
| . | Average weight (g ± s.d.) . | Average length (mm ± s.d.) . | Average width (mm ± s.d.) . | Water content (% ± s.d.) . | E:S ratio . |
|---|---|---|---|---|---|
| For 100 DWFP | 2·15 ± 0·08 | 5·98 ± 0·24 | 3·49 ± 0·11 | 8·70 ± 0·06 | 0·08 ± 0·01 |
| For 100 seed sensu stricto | 0·58 ± 0·06 | 4,19 ± 0·99 | 1·57 ± 0·37 | 8·99 ± 0·65 |
| . | Average weight (g ± s.d.) . | Average length (mm ± s.d.) . | Average width (mm ± s.d.) . | Water content (% ± s.d.) . | E:S ratio . |
|---|---|---|---|---|---|
| For 100 DWFP | 2·15 ± 0·08 | 5·98 ± 0·24 | 3·49 ± 0·11 | 8·70 ± 0·06 | 0·08 ± 0·01 |
| For 100 seed sensu stricto | 0·58 ± 0·06 | 4,19 ± 0·99 | 1·57 ± 0·37 | 8·99 ± 0·65 |
E.S ratio, embryo length:seed length ratio.
The potential resistance to embryo growth of the pericarp–mesocarp envelope, including the newly discovered amorphous silicate-crystal layer, is compatible with the possible presence of PD mechanisms in A. trichopoda. In addition, the early arrest of embryo development and the low E:S ratio in the freshly matured seed are both compatible with the possible presence of MD. We thus conducted preliminary germination assays to evaluate directly the presence of dormancy and gain insights into the overall type of any such mechanisms present.
Embryo growth and germination
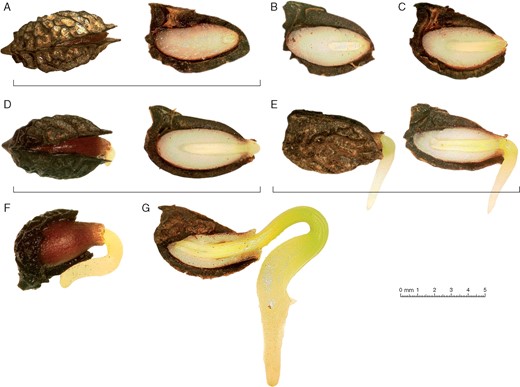
A preliminary time course of embryo growth and germination indicated that A. trichopoda seeds have a level of simple MPD. The pit (endocarp and hard part of the mesocarp) was observed to split open after a minimum of approx. 90 d incubation at 25 °C. At this stage, the embryo was small and heart shaped (rudimentary), as in the freshly matured seed (Fig. 2A). The enlargement of the seed, which resulted in the opening of both sides of the pericarp envelope, must therefore have been due to endosperm swelling, rather than to embryo growth. After the envelope had split open, the embryo began to grow (Fig. 2B) and developed from the heart to the torpedo stage (Fig. 2C). Following this, the embryo formed two well-developed cotyledons, and radicle protrusion occurred after a further 17 d (Fig. 2D), on average, by which time the E:S ratio had become 0·48 ± 0·07. After radicle emergence, the cotyledons continued to grow inside the seed, digesting the endosperm reserves (Fig. 2E, F), and emerged on average 23·5 d after the emergence of the radicle (Fig. 2G).
(A) Reconstructed chronological sequence of Amborella trichopoda seed germination. Underlining indicates drupes without fleshly parts (DWFP) viewed whole or longitudinally cut. (A) Slightly open DWFP, whole (top view) and longitudinally cut (side view). (B and C) Embryos at later developmental stages, showing wider openings. (D) Whole and longitudinally cut DWFP at the stage of radicle emergence. (E–G) Subsequent developmental stages.
The minimum incubation period of > 100 d to radicle emergence clearly indicated the presence of seed dormancy in A. trichopoda. The absence of embryo development for at least the first 90 d of incubation indicated the presence of a physiological component to that dormancy, while the incompletely developed (heart-stage) embryo, following the breakage of PD, revealed the additional presence of MD. We therefore concluded that A. trichopoda must have a form of MPD. MPD is divided into the two principal sub-classes of ‘simple MPD’, in which the embryo requires warm (≥15 °C) temperatures to grow and ‘complex MPD’ in which embryo requires cold (0–10 °C) temperatures to grow. Our preliminary observations clearly indicated the former scenario, and we therefore concluded the presence of a form of simple MPD in A. trichopoda.
Physiological components of dormancy
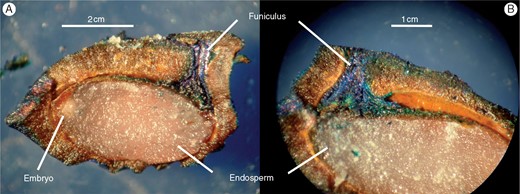
To elucidate the mechanistic basis of the PD component in A. trichopoda dormancy, we began by testing the water permeability of the mature drupe. When soaked in water for 72 h, the mass of both the DWFP and seed sensu stricto increased (Table 2), indicating that the pit was water permeable. Methylene blue solution entered the DWFP through the funiculus, opposite the hilum and embryo (Fig. 3), but stopped near the hilum, possibly because the dye molecules were unable to enter the cells of the seed coat.
Longitudinal section of a drupe without its fleshly part (DWFP) soaked for 72 h in 1 % methylene blue solution. (A) Entire DWFP. (B) Zoom near the funiculum.
Permeability of Amborella trichopoda drupes without fleshly part (DWFP) estimated by mass increase after 72 h, compared with controls, i.e. before imbibition
| Part (mean of three replicates of 20 samples) . | Mass of control (mg ± s.d.) . | Mass after 72h (mg ± s.d.) . | Mass increase (mg ± s.d.) . | % increase . |
|---|---|---|---|---|
| DWFP | 428 ± 20 | 474 ± 20 | 47* ± 10 | 10·9 %* |
| Seed sensu stricto | 114 ± 10 | 133 ± 10 | 18* ± 10 | 16·0 %* |
| Pit | 313 ± 20 | 342 ± 20 | 28* ± 10 | 9·1 %* |
| Part (mean of three replicates of 20 samples) . | Mass of control (mg ± s.d.) . | Mass after 72h (mg ± s.d.) . | Mass increase (mg ± s.d.) . | % increase . |
|---|---|---|---|---|
| DWFP | 428 ± 20 | 474 ± 20 | 47* ± 10 | 10·9 %* |
| Seed sensu stricto | 114 ± 10 | 133 ± 10 | 18* ± 10 | 16·0 %* |
| Pit | 313 ± 20 | 342 ± 20 | 28* ± 10 | 9·1 %* |
Asterisks indicate statistically significant (P < 0·05) increases in mass.
Permeability of Amborella trichopoda drupes without fleshly part (DWFP) estimated by mass increase after 72 h, compared with controls, i.e. before imbibition
| Part (mean of three replicates of 20 samples) . | Mass of control (mg ± s.d.) . | Mass after 72h (mg ± s.d.) . | Mass increase (mg ± s.d.) . | % increase . |
|---|---|---|---|---|
| DWFP | 428 ± 20 | 474 ± 20 | 47* ± 10 | 10·9 %* |
| Seed sensu stricto | 114 ± 10 | 133 ± 10 | 18* ± 10 | 16·0 %* |
| Pit | 313 ± 20 | 342 ± 20 | 28* ± 10 | 9·1 %* |
| Part (mean of three replicates of 20 samples) . | Mass of control (mg ± s.d.) . | Mass after 72h (mg ± s.d.) . | Mass increase (mg ± s.d.) . | % increase . |
|---|---|---|---|---|
| DWFP | 428 ± 20 | 474 ± 20 | 47* ± 10 | 10·9 %* |
| Seed sensu stricto | 114 ± 10 | 133 ± 10 | 18* ± 10 | 16·0 %* |
| Pit | 313 ± 20 | 342 ± 20 | 28* ± 10 | 9·1 %* |
Asterisks indicate statistically significant (P < 0·05) increases in mass.
We therefore attempted to evaluate whether the PD component in A. trichopoda was due to mechanical resistance of the pericarp–mesocarp envelope, surrounding the seed. To do this, we compared the time course of germination in intact drupes, drupes scarified by acid treatment and seed sensu stricto that had been excised from all surrounding tissues (Fig. 4).
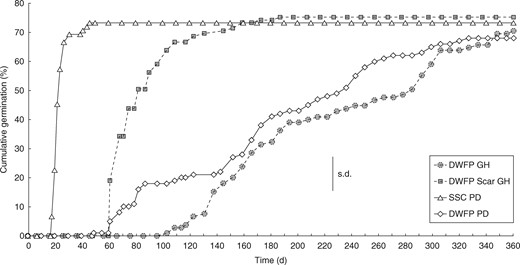
Time course of germination for Amborella trichopoda seeds. Seeds sensu stricto (SSC PD) and drupes without fleshly part (DWPF PD) in Petri dishes at 25 ± 1 °C with continuous light, and for DWFP with (DWPF GH) or without (DWPF Scar GH) acid scarification on trays at 25 ± 4 °C in a greenhouse at high humidity. Means were derived from four replicates of 25 seeds sensu stricto or DWFP. The certical bar denotes the largest s.d.
Scarification resulted in a reduction in the minimum germination time under greenhouse conditions from approx. 100 d to 60 d (Fig. 4). In agreement with this finding, a maximum of 70 % germination was reached after 138 d in scarified DWFP, compared with 362 d in intact DWFP. Mechanical excision of the seed sensu stricto had an even greater effect on dormancy; excised seeds germinated in Petri dishes in a minimum of 18 d, compared with 62 d for non-scarified control DWFP. Maximum germination of 73 % was reached after 48 d for excised seed in Petri dishes, compared with 68 % after 342 d for non-scarified DWFP. Germination followed a logarithmic curve both for scarified DWFP in the greenhouse and excised seeds in Petri dishes. Taken together, these data clearly indicate that mechanical resistance of the pericarp–mesocarp envelope, external to the seed sensu stricto, makes a major contribution to the physiological component of simple MPD in A. trichopoda.
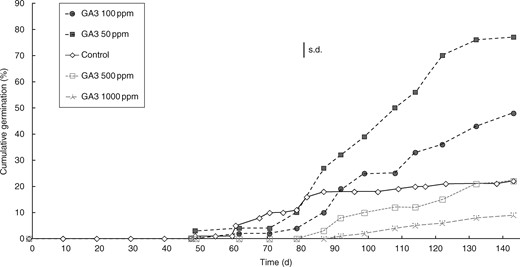
The period of time (approx. 90 d) required to break the physiological component of MPD in A. trichopoda is closely comparable with that in numerous species displaying non-deep simple MPD, rather than the alternative deep simple MPD (Baskin and Baskin, 2014), suggesting the presence of the former dormancy type. To confirm this hypothesis, we used a discriminatory test using exogenous GA3. The percentages of germination after 143 d and the rapidity of germination of non-scarified DWFP were significantly increased by incubation in GA3 solutions of 50 or 100 ppm, though higher concentrations of 500 or 1000 ppm GA3 were inhibitory to germination (Fig. 5). These data, taken together with our observations that seeds only require incubation at temperatures of ≥ 15 °C for dormancy break and germination, confirmed the presence of non-deep PD in seeds of A. trichopoda and thus the presence of non-deep simple MPD.
Time course of germination for non-scarified Amborella trichopoda drupes without fleshly parts (DWFP) in Petri dishes. DWFP were incubated at 25 ± 1 °C with continuous light on germination paper imbibed with solutions of 0, 50, 100, 500 and 1000 ppm of GA3. Means were derived from four replicates of 25 DWFP. The vertical bar denotes the largest s.d.
Test for epicotyl MPD
Six distinct sub-classes of simple MPD have been recognized (Baskin and Baskin, 2004), four of which include specific forms of epicotyl dormancy. To evaluate the presence of epicotyl dormancy in A. trichopoda, we measured the time necessary for cotyledon emergence, following radicle emergence. Cotyledon emergence from non-scarified DWFP occurred rapidly after radicle emergence, from 15 d for the earliest appearance of cotyledons to 30 d for the latest, giving an overall mean of 23·5 d (Table 3). The application of 50 ppm GA3 in lanolin paste had no significant effect on the speed of this process (P < 0·05). There are two sub-classes of simple MPD which do not involve epicotyl dormancy: deep simple MPD and non-deep simple MPD. As our findings indicate the absence of epicotyl dormancy in A. trichopoda, these are consistent with the identification in this species (above) of the non-deep simple level of MPD.
Time required for radicle and cotyledon emergence from Amborella trichopoda seeds and the effects on this process of GA3
| Treatment (24 h incubation) . | Mean no. of days between pit opening and radicle emergence . | Sub-treatment on 5 mm long rootlets . | Mean no. of days between radicle protrusion and cotyledon emergence . |
|---|---|---|---|
| Water (n = 98) | 17·6* (n = 95) | Untreated control (n = 41) | 23·5* (n = 39) |
| GA3 in lanolin (n = 38) | 24·6* (n = 37) | ||
| 50 ppm GA3 (n = 100) | 15·5* (n = 98) | – | – |
| Treatment (24 h incubation) . | Mean no. of days between pit opening and radicle emergence . | Sub-treatment on 5 mm long rootlets . | Mean no. of days between radicle protrusion and cotyledon emergence . |
|---|---|---|---|
| Water (n = 98) | 17·6* (n = 95) | Untreated control (n = 41) | 23·5* (n = 39) |
| GA3 in lanolin (n = 38) | 24·6* (n = 37) | ||
| 50 ppm GA3 (n = 100) | 15·5* (n = 98) | – | – |
Asterisks indicate statistically significant (P < 0·05) differences between the treatments shown in each column (50 ppm GA3 vs. water and GA3 in lanolin vs. untreated controls, respectively).
Numbers of replicates (n) in each treatment group are indicated in parentheses.
GA3 in lanolin treaments and their respective controls were performed on 79 surviving seedlings (n = 41 and 38, respectively) from the group of 98 seeds previously incubated in water only.
Time required for radicle and cotyledon emergence from Amborella trichopoda seeds and the effects on this process of GA3
| Treatment (24 h incubation) . | Mean no. of days between pit opening and radicle emergence . | Sub-treatment on 5 mm long rootlets . | Mean no. of days between radicle protrusion and cotyledon emergence . |
|---|---|---|---|
| Water (n = 98) | 17·6* (n = 95) | Untreated control (n = 41) | 23·5* (n = 39) |
| GA3 in lanolin (n = 38) | 24·6* (n = 37) | ||
| 50 ppm GA3 (n = 100) | 15·5* (n = 98) | – | – |
| Treatment (24 h incubation) . | Mean no. of days between pit opening and radicle emergence . | Sub-treatment on 5 mm long rootlets . | Mean no. of days between radicle protrusion and cotyledon emergence . |
|---|---|---|---|
| Water (n = 98) | 17·6* (n = 95) | Untreated control (n = 41) | 23·5* (n = 39) |
| GA3 in lanolin (n = 38) | 24·6* (n = 37) | ||
| 50 ppm GA3 (n = 100) | 15·5* (n = 98) | – | – |
Asterisks indicate statistically significant (P < 0·05) differences between the treatments shown in each column (50 ppm GA3 vs. water and GA3 in lanolin vs. untreated controls, respectively).
Numbers of replicates (n) in each treatment group are indicated in parentheses.
GA3 in lanolin treaments and their respective controls were performed on 79 surviving seedlings (n = 41 and 38, respectively) from the group of 98 seeds previously incubated in water only.
Character reconstruction by parsimony
Character-state mapping indicates that the MRCA of living angiosperms possessed MPD and reveals a transition to PD within Nymphaeales. We combined the identification of the level of MPD in A. trichopoda with published data on the class of seed dormancy in the three most basal lineages of flowering plants, Amborellales, Nymphaeales and Austrobaileyales. Notably, this analysis included data on three species of Trithuria from the basally diverging family Hydatellaceae (Tuckett et al., 2010), not included in previous reconstructions on the evolution of dormancy (Willis et al., 2014). We also included in our analysis the likely dormancy type in the MRCA of the remaining angiosperm lineage, the euangiosperms, which was inferred by Willis et al. (2014) as ambiguous between MPD and PD.
We investigated the evolution of dormancy mechanisms in the angiosperms using the consensus view of angiosperm phylogeny (Byng et al., 2016) in which Amborellales alone is sister to all other living angiosperms, (Fig. 6A, B), and the alternative view (Qiu et al., 2001, 2010; Xi et al., 2014) in which a clade consisting of Amborellales + Nymphaeales is sister to all other living angiosperms (Fig. 6C, D). These analyses included the dormancy type in the MRCA of euangiosperms, as reconstructed by Willis et al. (2014). Therefore, all living ANA-grade genera, and a very wide sampling of euangiosperms (previously performed by Willis et al., 2014), are represented in these analyses. Parsimony reconstruction, omitting data from A. trichopoda and Trithuria spp., failed to resolve the dormancy type at the base of the flowering plants in both of the angiosperm topologies considered (Fig. 6A, C). However, the addition of data from these taxa unambiguously resolved the dormancy type at the base of the angiosperm tree in both topologies considered (Fig. 6B, D). Using either of these alternative evolutionary scenarios, the base of the living angiosperms was resolved unambiguously as possessing MPD. We therefore conclude MPD as the likely pleisiomorphic condition of the living angiosperms. From our reconstruction, it appears that the PD present in Nymphaeaceae and Cabombaceae arose through the loss of the morphological component of dormancy from MPD that was present in the MRCA of all living Nymphaeales (Fig. 6B, D).
Mapping of germination classes by parsimony onto two alternative topologies for the base of angiosperm phylogeny. Alternative phylogenies are considered in which either Amborellales alone (A and B), or Amborellales + Nymphaeales (C and D) are sister to all other living angiosperms. When dormancy data from Amborella and Trithuria are excluded (A and C), the analysis fails to resolve the dormancy class at the base of living angiosperms (red arrows) between PD and MPD. The inclusion of dormancy data from Amborella and Trithuria (B and D) results in the resolution of the basal character state in the living angiosperms to MPD for both phylogenetic topologies considered. A transition from MPD to PD (blue arrows) can be inferred within Nymphaeales, in a common ancestor of Cabombaceae and Nymphaeaceae, in either phylogenetic topology (B and D). Classification of genera into families and orders is shown in (A).
DISCUSSION
Multiple significance of seed dormancy studies in Amborella trichopoda
In this study, we have determined the precise type of seed dormancy present in A. trichopoda, the only known representative of Amborellales and likely sister to all other flowering plants (Figs 1–5). We conclude that seeds of this species have non-deep simple MPD, in which the PD component is broken before the MD component (and is, therefore, ‘simple’; Fig. 2), and in which embryo growth occurred at high temperatures and GA3 was effective in promoting germination (Fig. 4). The PD component in A. trichopoda depends heavily on mechanical resistance to embryo growth from the pericarp–mesocarp envelope of the drupe (Fig. 4). Interestingly, we reveal the presence of an amorphous silicate-crystal layer, situated between the endocarp and mesocarp (Fig. 1E), which may make a significant contribution to the mechanical properties of the drupe, and therefore to the physiological component of dormancy in A. trichopoda.
The significance of our results is 3-fold. First, no character-state mapping at the base of the living angiosperms can be complete without the inclusion of data from A. trichopoda. Our study has thus enabled, for the first time, the precise reconstruction of the evolution of dormancy classes in the MRCA of living angiosperms, which we conclude to have possessed MPD (Fig. 6). This conclusion is useful for the interpretation of the subsequent evolution of dormancy classes within the angiosperms, such as the transition from MPD to PD that we infer to have occurred in a common ancestor of Nymphaeaceae and Cabombaceae (Fig. 6). Secondly, our findings contribute to a growing picture of the ecophysiology of A. trichopoda within its endemic habitat of the New Caledonian rain forest. We thus add data on seed dormancy and germination to studies of photosynthesis (Feild et al., 2001), stomatal function (Rudall and Knowles, 2013), vascular function (Feild et al., 2000; Turgeon and Medville, 2011; Feild and Wilson, 2012), flowering time (Fourcade et al., 2015), pollination biology (Thien et al., 2009), seed physiology (Poncet et al., 2015), population structure (Thien et al., 2003; Poncet et al., 2013) and genetic diversity (Poncet et al., 2012). These diverse studies of A. trichopoda make a major contribution to the concerted reconstruction of the ecophysiological features of the first flowering plants (see Feild et al., 2004; Feild and Arens, 2007). Thirdly, the investigation of dormancy in A. trichopoda is of practical use both for species conservation (e.g. for use in seed banks) and for the exploitation of A. trichopoda as an experimental model representing ANA-grade angiosperms. Such experimental uses may require the manipulation of dormancy and germination to facilitate numerous procedures including genetic crosses, tissue culture and plant transformation (see Scutt and Vandenbussche, 2014).
The conclusion that morphophysiological dormancy was present at the base of the living angiosperms is robust and should withstand any reasonably foreseeable change to angiosperm or gymnosperm phylogeny
The recently published study by Willis et al. (2014) concluded the presence of MPD in the MRCA of all living seed plants (angiosperms + gymnosperms) and indicated that MPD was the probable dormancy class near the base of living angiosperms. However, the principal conclusions of Willis et al. (2014) may have depended heavily on the version of gymnosperm phylogeny included in their analysis. The phylogenetic topology used by Willis et al. (2014) included Cycadales, followed by Gingoales, as the two most basal clades of living gymnosperms, and placed Gnetales in sister position to all conifers. This placement of Gnetales is consistent with several published phylogenies (e.g. Chaw et al., 1997; Wickett et al., 2014), though others place Gnetales within conifers, as sister to Pinaceae (e.g. Bowe et al., 2000; Zhong et al., 2010), or as sister to remaining conifers, excluding Pinaceae (e.g. Nickrent et al., 2000; Doyle, 2006). Importantly, two gymnosperm phylogenies have recently been proposed in which Gnetales are placed as sister to all other gymnosperms (de la Torre-Barcena et al., 2009; Lee et al., 2011). As all three monotypic families within Gnetales possess PD, rather than MPD (summarized by Willis et al., 2014), the phylogenetic placement of Gnetales is likely to have a critical impact on the reconstruction of dormancy mechanisms at the base of gymnosperms and, by extension, at the base of the living seed plants.
In contrast to the situation in gymnosperms, the basal nodes of angiosperm phylogeny, as explained above, have been broadly established, with only minor differences between a consensus view supported by the majority of studies (Byng et al., 2016), and an alternative view supported by a smaller number of studies (Qiu et al., 2001, 2010; Xi et al., 2014). We therefore preferred, in the present work, to reconstruct the type of dormancy of the MRCA of all living angiosperms using data exclusively from angiosperms. We performed four analyses (Fig. 6), including both of the alternative evolutionary scenarios that emerge from the current literature, in each case with and without data from A. trichopoda (from this work) and Trithuria spp. (Tuckett et al., 2010), which were not available to, or not included in, previous evolutionary reconstructions. We show that the addition of data from these key basal taxa leads to the resolution of the MPD in the MCRA of living angiosperms, regardless of which of the two phylogenetic topologies that we analysed is considered. We do not, therefore, expect this evolutionary conclusion to be brought into question by any reasonably foreseeable change to the consensus view of angiosperm phylogeny. In addition, as our principal evolutionary conclusion was reached using well-established data exclusively from angiosperms, it should not be affected by the current flux and uncertainty in gymnosperm phylogeny.
Importantly, our conclusion that MPD was the likely dormancy class in the first angiosperms supports the model proposed by Willis et al. (2014) for the evolution within angiosperms of PD and MD, via alternative routes, from MPD. The establishment of MPD at the base of the angiosperms also supports a second hypothesis advanced by Willis et al. (2014) that key ancestral species within the euangiosperms, in which PD had already evolved from MPD, then acted as foci for the evolutionary diversification of seed dormancy mechanisms.
ACKNOWLEDGEMENTS
We acknowledge grant funding from to the Fondation Ars-Cuttoli-Paul Appell under the aegis of the Fondation de France. We are also grateful to the Province Sud de Nouvelle-Calédonie for permission for seed collection and for additional financial support. We thank Jacques Rabier, Roger Notonier and Alain Tonetto at the SCME, University of Provence, France for help with SEM, and finally special thanks to C. Zongo (deceased) for field collection of seeds.