-
PDF
- Split View
-
Views
-
Cite
Cite
Jacqueline A. Gilbert, Susan L. Kalled, Jane Moorhead, Donna M. Hess, Paul Rennert, Zhifang Li, M. Zareen Khan, J. Paul Banga, Treatment of Autoimmune Hyperthyroidism in a Murine Model of Graves’ Disease with Tumor Necrosis Factor-Family Ligand Inhibitors Suggests a Key Role for B Cell Activating Factor in Disease Pathology, Endocrinology, Volume 147, Issue 10, 1 October 2006, Pages 4561–4568, https://doi.org/10.1210/en.2006-0507
Close - Share Icon Share
Hyperthyroid Graves’ disease is a common autoimmune disorder mediated by agonistic antibodies to the TSH receptor, termed thyroid stimulating antibodies (TSAbs). Recently members of the TNF superfamily, B cell activating factor (BAFF) and a proliferation-inducing ligand (APRIL), have been identified along with their receptors, B cell maturation antigen and transmembrane activator and calcium-modulator and cyclophilin ligand interactor, and the BAFF-specific receptor. BAFF is a fundamental B cell survival/maturation factor, and both BAFF and APRIL have been implicated in antibody production. We investigated the effect of interfering with BAFF- and APRIL-mediated signals in an induced model of Graves’ disease by blockade of these factors using soluble decoy receptors. In a therapeutic setting in mice with established hyperthyroidism, we show that blockade of BAFF or BAFF+APRIL with BAFF-specific receptor-Fc and B cell maturation antigen-Fc, respectively, leads to significant reductions in the induced hyperthyroidism. This was supported by a parallel pattern of declining TSAbs in the responding animals. Histopathological analysis of splenic sections from treated animals revealed marked reductions in the B cell follicle regions, but staining with anti-CD138 revealed the persistence of plasma cells. Thus, the reductions in TSAbs in the treated animals were not related to overall plasma cell numbers in the secondary lymphoid organs. Our results are the first to demonstrate attenuation of established hyperthyroidism by therapeutic intervention aimed at autoreactive B cells and indicate that both BAFF and APRIL appear to play important roles in the development and survival of the autoantibody producing cells in this model.
HYPERTHYROID GRAVES’ DISEASE is a common autoimmune disorder with a prevalence of up to 2.7% in some Caucasian populations, with predominance in females of child-bearing age (1). The hyperthyroidism is characterized by the production of a unique subset of autoantibodies to the TSH receptor (TSHR) with powerful agonistic properties (2). These autoantibodies, known as thyroid stimulating antibodies (TSAbs), lead to hyperstimulation of the gland resulting in copious secretion of thyroid hormones. Due to the pathogenic role of autoantibodies in hyperthyroid Graves’ disease, this condition is considered an archetypal Th2-type mediated disorder (3). Treatment for the disease has changed little over the past 50 yr and typically consists of medical (Europe) or radioiodine (United States) intervention, the latter often leading to hypothyroidism requiring life-long hormone replacement (4). This may be associated with long-term increased morbidity and mortality (5). Extrathyroidal complications such as ophthalmopathy develop in a proportion of patients with Graves’ disease. Interestingly, cross-reactivity of anti-TSHR antibodies with orbital tissues as the primary event leading to orbital inflammation has recently been demonstrated (6, 7). Unfortunately, severe ophthalmopathy is often difficult to manage clinically and is usually treated by blanket immunosuppression with steroids or orbital radiotherapy (8). The pathogenesis of the various extrathyroidal complications remains to be fully elucidated. An interesting aspect of the pathogenic TSAbs is the frequent restriction of their H and L chains, leading to predominantly IgG1/λ subclass antibodies (9, 10). The restricted repertoire of the TSAbs has led to suggestions of a defect in either B cell development or negative selection in germinal centers for peripheral tolerance contributing to the disorder. No other animal species have been recorded to spontaneously develop hyperthyroid Graves’ disease; thus, the lack of a suitable animal model has hindered studies on assessing therapeutic options for the disorder.
The TNF superfamily consists of related ligands and receptors, some of which play essential roles in immunity and B cell biology (11). Two closely related ligands belonging to the TNF family, termed B cell-activating factor (BAFF) (also known as BlyS) and a proliferation-inducing ligand (APRIL), have been identified as crucial players in B cell development and maintenance of antibody production, respectively (12). In transgenic studies in mice, overexpression of BAFF leads to elevated peripheral B cell numbers and serum Ig, resulting in autoimmune manifestations akin to systemic lupus erythematosus and Sjogren’s syndrome (13, 14). BAFF and APRIL can bind multiple receptors belonging to the TNF receptor superfamily. BAFF receptor (BAFFR) interacts solely with BAFF, and this BAFF/BAFFR axis is essential for B cell growth and survival (12, 15). Two other receptors, termed B cell maturation antigen (BCMA) and transmembrane activator and calcium-modulator and cyclophilin ligand interactor, interact with both BAFF and APRIL (12, 15). Data indicate that transmembrane activator and calcium-modulator and cyclophilin ligand interactor functions as a negative regulator of B cell growth, and BCMA may be important for promotion of antigen-presenting function of B cells (16) and survival of long-lived plasma cells (17). However, the precise role played by APRIL in B cell development remains to be defined because APRIL-deficient mice in different laboratories have demonstrated disparate characteristics and thus failed to provide consistent clues to its function (18, 19). Although both BAFF and APRIL bind to BCMA, it has recently been recognized that APRIL binding to monomeric soluble BCMA is a higher-affinity interaction (×100- to 1000-fold), suggesting that the APRIL/BCMA interaction may be the relevant physiological axis for APRIL (20, 21). Moreover, blocking the action of BAFF and BAFF+APRIL with soluble decoy receptor-Fc fusion proteins has led to amelioration of spontaneous and induced autoimmune diseases such as systemic lupus erythematosus, experimental autoimmune encephalomyelitis, and rheumatoid arthritis (22–25). For this reason, there is a great deal of interest in using these compounds to treat autoimmune diseases (26, 27).
The development of induced experimental models of hyperthyroid Graves’ disease has led to an increased understanding of the disease mechanisms. Almost all studies that have attempted to modify the course of disease have focused on using cytokine-deficient mice or agents to perturb the Th1 and Th2 balance of the immune response to the TSHR (28–32). Recent data suggest that inducing polarization of the immune response to a Th1 type in a preventive setting before the onset of disease may be beneficial. However, application of this strategy as a treatment protocol in established disease fails to modify the disorder. In this study, we examined the effect of blocking the action of the ligands BAFF and BAFF+APRIL with BAFFR-Fc and BCMA-Fc fusion proteins, respectively, in modulating an experimental model of hyperthyroid Graves’ disease. We used the recently developed adenovirus model using the A-subunit of TSHR in female BALB/c animals (33) and focused on a treatment protocol using animals with established bona fide hyperthyroidism. Our data show that immunological intervention to eliminate autoreactive B cells in animals with established hyperthyroid disease is efficacious in treating this disease and gives new insight into the role of both BAFF and APRIL in this B cell-mediated disorder.
Materials and Methods
Mice and adenovirus constructs of TSHR
Recombinant adenovirus expressing TSHR-A subunit (amino acids 1–289) (referred to as A-subunit-Ad) was a generous gift from Drs. Sandra McLachlan and Basil Rapoport (33). As control, adenovirus containing β-galactosidase (β-Gal) was used. All virus constructs were propagated in HEK293 cells and purified twice over CsCl gradient centrifugation (33), dialyzed against PBS, and viral concentration determined by optical absorbance at 280 nm. Purified adenovirus were aliquoted and stored at −80 C.
Female BALB/c mice (100 mice, aged 7–8 wk; Harlan UK Ltd., Bicester, UK) were injected im with a low-dose immunization protocol of A-subunit-Ad (50 μl containing 109 particles) in PBS two times at 3-wk intervals (34). The animals were bled 1 wk after the second injection and tested individually for hyperthyroid status by measuring total serum T4 and TSAb activity. Control animals (10 mice) were injected as described above with β-Gal-Ad (50 μl containing 109 particles in PBS). All animals were housed under nonbarrier-free conditions and conducted in accord with accepted standards of humane animal care, as outlined in the ethical guidelines of the journal. Moreover, all experimental procedures were approved by strict regulations of the Home Office in the United Kingdom.
Induction and assessment of experimental autoimmune hyperthyroidism
Hyperthyroid status in the immunized mice was ascertained by measuring total T4 and TSAbs in serum samples collected by saphenous vein bleeding. T4 was determined in 25 μl serum sample using a commercial kit (Diagnostic Systems Laboratories, Oxford, UK). TSAbs were assayed in duplicate samples by bioassay using a CHO cell line stably transfected with human TSHR (JP09 cells) under salt-free isotonic buffer conditions as described (35). The cAMP released into the supernatant from stimulated JP09 cell cultures was determined by a RIA kit (R&D Systems, Oxford, UK) and the results expressed in picomoles per milliliter. Hyperthyroid status in the animals was assessed 1 wk after the second immunization.
Animals with significantly elevated levels of T4 and/or TSAbs were selected for the treatment study and randomized into four groups. In addition, serum samples from the diseased animals at the start of the treatment protocol were retained and stored frozen at −20 C to run in parallel in assays with serum samples at the end of the immunomodulatory protocol.
TSH binding inhibitory immunoglobulins (TBIIs)
Antibodies to TSHR are routinely detected by commercial radioreceptor assays, in which 125I-labeled bovine TSH and antibodies compete for binding sites on immobilized TSHR (2). The antibody bound to the receptor is expressed as the percentage of inhibition of the labeled TSH binding, with the antibody activity commonly referred to as TBIIs. The assay was performed with the human TRAK II kits (DYNOtest; BRAHMS AG, Berlin, Germany) using 100 μl undiluted serum in single determination according to manufacturer’s instructions. Inhibition greater than 10–15% is generally considered positive (35).
Treatment with BAFFR-Fc and BCMA-Fc
Human BAFFR-Fc and BCMA-Fc were expressed as recombinant proteins in CHO cells and purified as described (36). Therapy with BAFFR-Fc and BCMA-Fc was commenced within 7 d after the testing of the serum samples for hyperthyroid status. Animals were treated for 12 wk with 200 μg BAFFR-Fc or BCMA-Fc in 500 μl PBS given ip every 3–4 d. The control groups included PBS (500 μl) and an untreated group. Ten days after the last injection of the treatment protocol, mice were killed over a 2-d period, blood collected by cardiac puncture for enumeration of B cell numbers by fluorescence-activated cell sorter (FACS) analysis, sera harvested, and thyroid glands and spleens excised for immunohistochemistry.
Enumeration of B cells in peripheral blood by FACS analysis
B220+ B cells were measured in the peripheral blood by FACS analysis using fluorescein isothiocyanate (FITC)-labeled antimouse B220 (BD Biosciences, Oxford, UK). Whole blood (100 μl) was mixed with 1 ml lysis buffer [0.155 m NH4Cl, 0.01 m KHCO3, 0.1 mm EDTA (pH 7.2)] to lyse red blood cells by incubation for 5 min on ice. The cells were washed twice by low-speed centrifugation for 5 min at 1000 × g, resuspended in PBS/1% BSA, and incubated with 1 μl (0.01 μg) rat antimouse CD16/CD32 monoclonal antibody (mAb) combination for 30 min on ice to block the FcγIII and II receptors, respectively (Fc-Block; BD Biosciences). This was followed by the addition of 1 μl FITC-labeled rat antimouse CD45R/B220 mAb combination (BD Biosciences); for control, a parallel sample was also incubated with an FITC IgG2a isotype control mAb (BD Biosciences) and incubated for 30 min on ice. After washing as described, the cells were resuspended in 450 μl PBS/1% BSA and analyzed using FACSCalibur (BD Biosciences) and data analyzed using CELLQuest program (BD Biosciences).
Immunohistochemistry
Spleens were divided in half. One spleen segment and the thyroid glands were fixed in formalin, embedded in methacrylate, and 4-μm sections stained with hematoxylin-eosin. Plasma B cells were initially enumerated by an experienced histopathologist by histomorphological techniques. The results were then confirmed by immunohistochemistry by freezing the remainder of the spleen segment in liquid N2 and plasma cells stained with antimouse CD138 followed by detection with the ImmuoCruz antigoat kit (Santa Cruz Biotechnology, Santa Cruz, CA).
Statistical analysis
Wilcoxon signed rank test and Mann-Whitney U tests were used to examine differences within and between treatment groups respectively. P < 0.05 denoted significance.
Results
Induction of hyperthyroid Graves’ disease
Because this report deals with the outcome of a treatment study on the in vivo effect of BAFFR-Fc and BCMA-Fc in established disease, we first induced hyperthyroid Graves’ disease in female BALB/c mice. We used the TSHR A-subunit-Ad model, which to date is the most effective protocol for inducing hyperthyroid Graves’ disease, with the highest reported disease incidence (33). Furthermore, to preferentially induce TSAbs in the model, the lower viral dose immunization protocol in conjunction with the two injection protocol of Chen et al. (34) was used. Animals (n = 100) were challenged twice at 3-wk intervals with A-subunit-Ad. The induction of hyperthyroid Graves’ disease was monitored by assessment of serum T4 and TSAbs in serum collected 1 week after the second immunization (T4 in control β-Gal-Ad-injected mice serum, range 4.88 ± 1.25 μg/dl, greater than mean + 2 sd = 7.0 μg/dl). A total of 38 diseased animals with experimental Graves’ disease, demonstrating elevated T4 and/or TSAb levels, was identified at the end of the injection schedule and selected for the immunomodulatory study. The disease incidence was in agreement with the A-subunit model described by Chen et al. (33, 34). From these animals (n = 38), 52% of the animals demonstrated elevated levels of both T4 and TSAbs, 27% elevated T4 alone, and 21% elevated TSAbs alone. In addition, because a wide range of elevated TSAb values were observed, mice with elevated TSAbs were also randomized according to the following TSAb categories (stimulatory cAMP levels in picomoles per milliliter): 25–100, 6.25–24.9, 2–6.24, and 1–1.9. The animals were randomized to the treatment groups of BAFFR-Fc and BCMA-Fc; as controls, one group of animals was treated with PBS (10 diseased animals/group). Another group of diseased animals was used as a control group that remained unmanipulated and referred to as the natural history group. Due to the paucity of animals with elevated levels of both T4 and TSAbs, the majority of animals in the natural history group demonstrated either elevated T4 alone or TSAbs alone (eight diseased animals/group).
BAFFR-Fc and BCMA-Fc treatment leads to amelioration of hyperthyroid Graves’ disease in mice
To examine the effects of blockade of BAFF or BAFF+APRIL with decoy receptor-Fc fusion proteins on hyperthyroid Graves’ disease, diseased animals were treated with 200 μg BAFFR-Fc or BCMA-Fc given ip every 3–4 d for 12 wk. Control groups were composed of vehicle injections (PBS) or remained unmanipulated as the natural history group. This protocol with the immunomodulators is known to have a significant effect on the number of peripheral blood B cells as well as in the B cell areas of secondary lymphoid organs such as the spleen (36, 37). We continued the treatment protocol for 12 wk to ensure clearance of all serum IgG present at the beginning of treatment. To minimize interassay variations for the T4 and TSAb measurements, serum samples from the start of the treatment protocol (stored at −20 C) were assayed simultaneously with serum from the corresponding animal at the end of the treatment protocol. Animals were killed 10–12 d after the last injection of fusion protein.
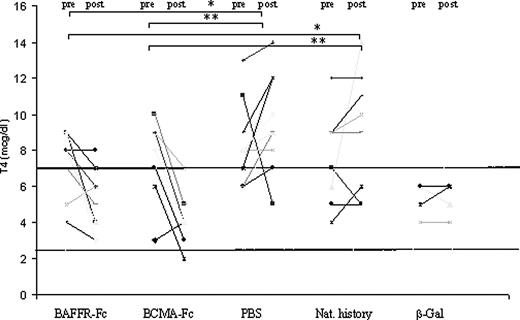
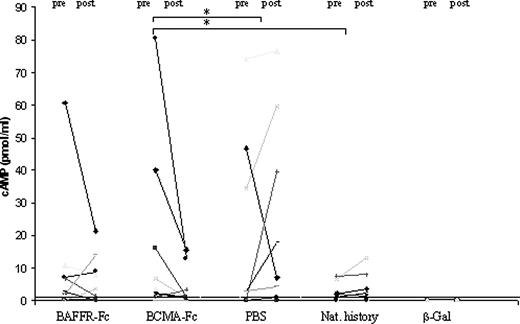
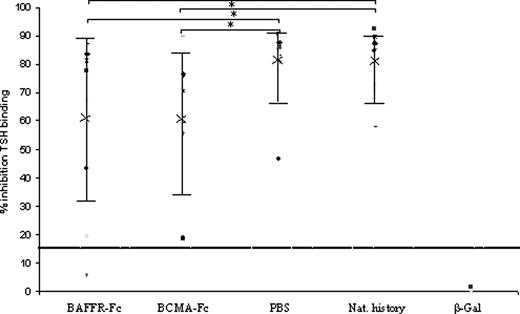
Like the human disease, a hallmark of this induced model of Graves’ disease is an elevation in serum T4. We found that treatment with BAFFR-Fc, which blocks BAFF activity, led to a significant reduction in serum T4 in 78% of the animals, compared with pretreatment levels (P < 0.01) (Fig. 1). In comparison with the control PBS treatment and natural history groups, the reductions in T4 were significant (P < 0.05). No significant differences were observed between the control PBS and unmanipulated natural history groups. Likewise, treatment with BCMA-Fc, which blocks both BAFF and APRIL, demonstrated similar effects on thyroid function in the diseased animals with significant reductions in serum T4 levels, compared with pretreatment levels (P < 0.01) and the control PBS treatment and natural history groups (P < 0.01). In addition, these mice also develop TSAbs, measured in bioassay by their ability to stimulate the TSHR in a stably transfected CHO cell line (33). Importantly, treatment with either BAFFR-Fc or BCMA-Fc also led to declining serum TSAb responses (BCMA-Fc; P < 0.05) (Fig. 2). Together the results indicate that BAFF blockade with BAFFR-Fc or blockade of both BAFF+APRIL with BCMA-Fc leads to a marked improvement of hyperthyroidism in the animals, revealed by significant reductions in serum T4 and TSAb levels. In parallel, the total anti-TSHR antibody responses were determined in a radioreceptor assay by examining the ability of serum to inhibit 125I-TSH binding to the TSHR (TBII activity). TBII activity was significantly reduced in the BAFFR-Fc- and BCMA-Fc-treated groups, compared with the PBS and natural history control groups (P < 0.05) (Fig. 3). No significant differences were observed between the control PBS and unmanipulated natural history groups.
Serum T4 levels before the commencement of the treatment (labeled pre) and after completion of the treatment protocol (labeled post) in all study cohorts. All the samples were measured at the same time in a single assay to minimize interassay variability. Treatment with BAFFR-Fc led to a reduction in T4 in 78% of diseased animals, whereas BCMA-Fc led to a reduction in T4 in 100% of the diseased animals, compared with pretreatment levels (P < 0.01). In comparison with the PBS and natural history control groups, the reduction in T4 was also significant. No significant differences were observed between the control PBS and unmanipulated natural history groups. Nine data points are shown for BAFFR-Fc, BCMA-Fc, and PBS groups, eight data points for natural history group. Normal range as shown 2.38–7 μg/dl (dotted line). *, P < 0.05; **, P < 0.01.
TSAb levels in terms of cAMP stimulation (picomoles per milliliter) before the commencement of the treatment (labeled pre) and after completion of the treatment protocol (labeled post) in all the study cohorts. All the samples were measured at the same time in a single assay to minimize interassay variability. Treatment with BCMA-Fc led to a reduction in TSAbs in seven of nine animals, compared with pretreatment levels (P < 0.05). In comparison with the PBS and natural history control groups, the reduction in TSAbs was also significant. No significant differences were observed between the control PBS and unmanipulated natural history groups. Some points within the groups are superimposed. Nine data points are shown for BAFFR-Fc, BCMA-Fc, and PBS groups, eight data points for natural history group. Normal range is less than 1 pmol/ml (dotted line). *, P < 0.05.
Assessment of overall anti-TSHR antibodies at posttreatment measured as TSH binding inhibitory activity in the TRAK assay. Significant reductions were observed in the BAFFR-Fc- and BCMA-Fc-treated groups, compared with the PBS and natural history control groups. Nine data points are shown for BAFFR-Fc, BCMA-Fc, and PBS groups, eight data points for natural history group. Means and sds are shown. Normal value is less than 15% (dotted line). *, P < 0.05).
BAFFR-Fc and BCMA-Fc treatment leads to reduction in B cell numbers and anti-TSHR antibodies
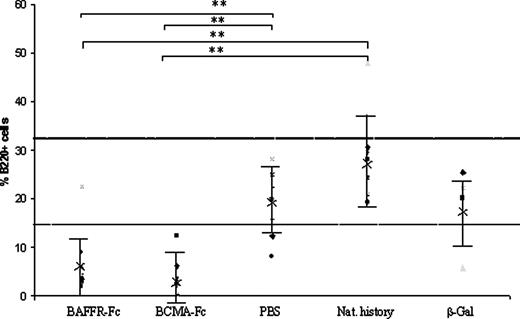
We examined the effect on overall peripheral blood B cell numbers in mice challenged with BAFFR-Fc and BCMA-Fc by FACS analysis. As expected, these were significantly reduced in comparison with the control PBS and natural history groups (P < 0.01) (Fig. 4).
B220+ve cells in the periphery at posttreatment enumerated by FACS analysis. Compared with the PBS and natural history control groups, significant reductions in the percent of peripheral blood B cells after treatment with both BAFFR-Fc and BCMA-Fc were observed. Nine data points are shown for BAFFR-Fc, BCMA-Fc, and PBS groups, eight data points for natural history group. Means and sds are shown. Normal range is between 14 and 32% (dotted line). **, P < 0.01.
Histological assessment
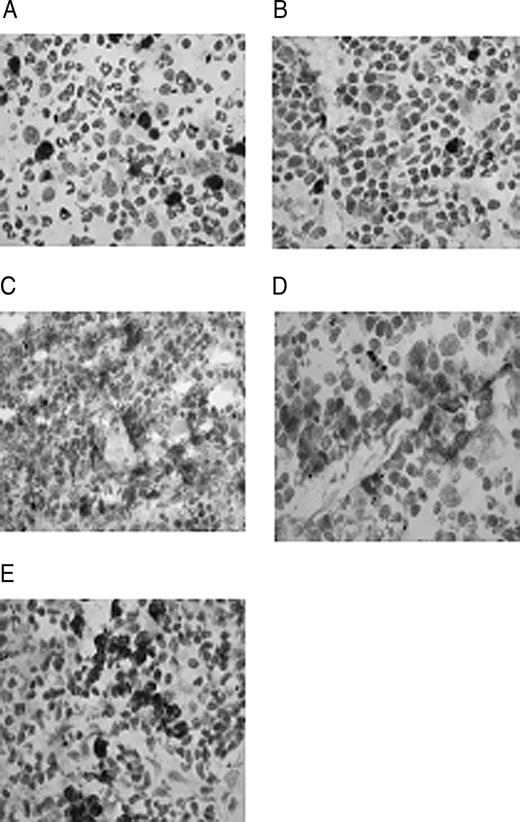
Histopathological examination of spleen sections in mice at the end of the treatment period revealed marked reductions in both the number and size of B cell follicles from both the BAFFR-Fc- and BCMA-Fc-treated groups, compared with the control groups (not shown). Immunohistochemical staining for plasma cells with antimouse syndecan-1 (anti-CD138) clearly show comparable levels of background staining (light brown stain) in all groups. Areas of darker staining plasma cells (dark brown stain) were also present in all groups, although this appeared to be more heterogeneous in the BAFFR-Fc- and BCMA-Fc-treated groups (Fig. 5, A and B), compared with control groups of PBS, unmanipulated and the β-Gal-treated animals (Fig. 5, C–E). Thus, despite the significant reduction in the peripheral blood B cell numbers and antibody response, there did not appear to be marked differences in the number of plasma cells in the secondary lymphoid organs such as the spleen after treatment with BAFFR-Fc and BCMA-Fc.
Immunohistochemical staining with syndecan-1 (CD138) for plasma cells in spleen sections of treated animals. BAFFR-Fc (A), BCMA-Fc (B), PBS (C), natural history (D), and β-Gal (E). Dark shade indicates positive staining; magnification, ×60.
Thyroid gland histology was not examined formally in this study, but in a separate cohort of BALB/c mice immunized with A-subunit-Ad, thyroid follicular cell morphology demonstrated tall, columnar cells, indicating an activated state but was not associated with any inflammation (our unpublished data). These results are similar to the findings reported in other laboratories (29, 33, 34).
Discussion
BAFF/APRIL inhibitors have demonstrated efficacy in a variety of animal models that are believed to be autoantibody mediated. Indeed, these studies have led to the development of BAFF and BAFF+APRIL inhibitors that are currently being tested in clinical trials (26, 27, 38). Hyperthyroid Graves’ disease is one of the most common antibody mediated autoimmune disorders, and in the study described herein we provide clear evidence for the ability of BAFFR-Fc and BCMA-Fc fusion proteins to ameliorate established Graves’ disease in an animal model. The effect of the BAFF and BAFF+APRIL inhibitors, BAFFR-Fc and BCMA-Fc, respectively, led to reductions of both T4 and anti-TSHR antibodies in almost all of the animals with resultant improvements in the hyperthyroid status of the diseased mice; there were also exceptions in which some animals in the groups failed to show lowering of T4 and TSAbs with the treatment, which may be due to heterogeneity of the disease, despite the use of inbred animals (34, 35).
Our results are also in agreement with previous studies showing that treatment with BAFFR-Fc and BCMA-Fc leads to a reduction in the number of peripheral blood B cells as well as B cell follicle regions of secondary lymphoid tissue such as the spleen (15, 26, 36). The resultant decline in peripheral B cell numbers likely contributed to the overall reduction in anti-TSHR antibody levels seen in the BAFFR-Fc- and BCMA-Fc-treated groups due to anticipated loss of TSHR-specific immature and mature B cells. It is also possible, however, that BAFF and BAFF+APRIL blockade adversely influenced the antigen presenting function of B cells with resultant effect on T cell function (39) or inhibited plasma cell differentiation and/or survival. Recent data suggest that signaling by BCMA is important for antigen presentation by B cells (16) and plasma cell survival (17), although it is unclear whether BAFF, APRIL, or both ligands can mediate this signal. Splenic plasma cells, enumerated by anti-CD138 staining, continued to persist after BAFFR-Fc and BCMA-Fc treatment, suggesting that the reduction in pathogenic TSAbs observed in the serum of treated animals does not strictly correlate with elimination of plasma cells in the spleen. Further studies are required to identify the source of the autoantibodies in this system to elucidate the mechanism of action of BAFF/APRIL blockade.
Hyperthyroxinemia is determined by measurement of serum thyroid hormones (4) and occurs as a result of chronic stimulation of the TSHR in thyroid follicular cells by TSAbs. Treatment with BAFFR-Fc and BCMA-Fc led to reductions in serum thyroid hormone levels in the majority of the animals. There was also a trend in the reduction in serum TSAbs in animals treated with BAFFR-Fc and BCMA-Fc, which reached significance in the latter treated group. Whereas the BAFF/BAFFR axis appears to be important for the growth and maintenance of the pathogenic, anti-TSHR antibody secreting autoreactive B cells in this model as demonstrated by the effect of BAFFR-Fc, the role of the APRIL/BCMA axis remains to be clarified. It is known that BCMA-Fc blocks the effect of both BAFF and APRIL, but because the affinity of BCMA for APRIL is considerably higher than that of BAFF, the effects of BCMA-Fc in this model may also be mediated in part by blockade of APRIL (20, 21). Hence, the role of the individual axes in the Graves’ disease model warrants further study. The fact that APRIL-deficient mice have failed to define the specific role of APRIL in B cell biology (18, 19) means that the use of gene targeted mice may not prove useful for this model. The properties of an APRIL-specific BCMA molecule, derived by protein engineering, have been described (21), and studying the effect of APRIL-specific blockade in this model may give new insights into the role of BAFF and APRIL in autoimmune hyperthyroid Graves’ disease. Recently Ingold et al. (40) reported APRIL-specific receptors (proteoglycans) and unique functions attributed to the effects of APRIL or APRIL+BAFF (but not BAFF alone). An interesting aspect of this study was the description of the role of APRIL or APRIL+BAFF (but not BAFF alone) in modulating the survival of bone marrow B cells, indicating demarcation of function between the two ligands (40). Similar distinct functions may also be important in the roles played by BAFF and APRIL in maintaining the autoreactive B cells secreting anti-TSHR antibodies in the Graves’ disease model.
Different populations of mature, immunocompetent B cell subsets have been defined, which comprise B1, marginal zone, and germinal center cells (41). It is not known which B cell population is responsible for producing the pathogenic anti-TSHR antibodies, although recent studies have indicated that the intrathyroidal B cells in Graves’ disease patients exhibit a phenotype related to marginal zone B cells (42). Attempts have also been made to study the relationship between TSHR antibody titers and the degree of intrathyroidal B cell infiltrate in the thyroid glands of patients with Graves’ disease (43) as well as to quantify the TSHR antigen-specific B cell populations within the germinal centers of the glands. However, no relationship between these parameters was observed (44). Serum levels of both BAFF (25) and APRIL (45) have been found to be elevated in certain patient populations when compared with healthy controls and thus their measurement in new onset Graves’ patients as well as in this model may be interesting in understanding the dysregulated role of BAFF and/or APRIL in autoimmune hyperthyroidism. Dissecting the mechanisms of how the blockade of both APRIL and BAFF ameliorates hyperthyroid Graves’ disease may lead to better targeted therapies for Graves’ disease and other B cell-mediated autoimmune disorders.
Acknowledgments
We thank Drs. Sandra McLachlan and Basil Rapoport for their generosity in providing the A-subunit-Ad virus. We also acknowledge Drs. Teresa Cachero, Fang Qian, and Yen-Ming Hsu for the production and purification of BAFFR-Fc and BCMA-Fc. The TRAK kits were generously provided by Brahms AG. Finally, one of us (J.P.B.) thanks Professor Claude C. A. Bernard (La Trobe University, Victoria, Australia) for his insightful discussions.
Disclosure statement: J.A.G., J.M., M.Z.K., and J.P.B. have nothing to declare. S.L.K., D.M.H., P.R., and Z.L. are employees of and stockholders of Biogen Idec.
Abbreviations:
- APRIL
A proliferation-inducing ligand
- BAFF
B cell-activating factor
- BAFFR
BAFF-specific receptor
- BCMA
B cell maturation antigen
- FACS
fluorescence-activated cell sorter
- FITC
fluorescein isothiocyanate
- β-Gal
β-galactosidase
- mAb
monoclonal antibody
- TBII
TSH binding inhibiting immunoglobulin
- TSAb
thyroid-stimulating antibody
- TSHR
TSH receptor