Abstract
Cryptococcus gattii is a primary pathogenic basidiomycetous yeast comprising four genotypic groups. Here we present data on two mitochondrial loci (MtLrRNA and ATP6). Two of the genotypic groups, namely amplified fragment length polymorphism (AFLP)5/VGIII and AFLP6/VGII, formed monophyletic lineages. The AFLP4/VGI genotypic group, however, possessed five different mitochondrial genotypes that did not form a monophyletic lineage. The majority of these isolates contained mitochondrial genomes that are partially identical to those found in isolates belonging to AFLP6/VGII, which is causing the ongoing and expanding Vancouver Island outbreak. Two out of four AFLP7/VGIV isolates contained an AFLP4/VGI allele of MtLrRNA. These observations are best explained by assuming a process of mitochondrial recombination. If this is true, mitochondrial recombination seems possible between cells belonging to different genotypic groups of C. gattii, especially between AFLP6/VGII or AFLP7/VGIV and AFLP4/VGI. We also have to assume that mitochondria, most likely, were transferred from cells belonging to AFLP6/VGII to AFLP4/VGI. As such a process of mitochondrial recombination is only possible after cell–cell conjugation, this may also allow the further exchange of genetic material, for example nuclear or plasmid in nature, between different genotypes of C. gattii. This may be relevant as it may provide a possible mechanism contributing to the modulation of virulence attributes of isolates, such as has been observed in the ongoing Vancouver Island outbreak of C. gattii.
Introduction
Cryptococcus gattii is a primary pathogenic basidiomycetous yeast that has previously been considered a variety of Cryptococcus neoformans (Kwon-Chung, 1982). Recently, C. gattii has been described as a distinct species because of differences in ecology, epidemiology, biochemical and molecular characteristics (Kwon-Chung, 2002; Kwon-Chung & Varma, 2006). Both C. gattii and C. neoformans may cause meningoencephalitis, but the primary pathogen C. gattii can cause disease in otherwise healthy individuals, whereas the opportunist C. neoformans primarily infects immunocompromised patients (Speed & Dunt, 1995). Cryptococcus gattii has been found on several tree species since its first isolation from Eucalyptus camaldulensis (Ellis & Pfeiffer, 1990; Callejas, 1998; Lazéra, 1998, 2000; Fortes, 2001; Krockenberger, 2002; Fraser, 2003; Randhawa, 2003; Granados & Castañeda, 2005; Escandón, 2006; Kidd, 2007), suggesting that trees might be the primary ecological niche of the species. In contrast, Kidd (2007) suggested that soil may be the principal reservoir for C. gattii. Cryptococcus gattii occurs predominantly in subtropical areas (Kwon-Chung & Bennett, 1984), but has also been isolated in Europe (Viviani, 2006) and in a temperate climate zone in Colombia (Escandón, 2006). Furthermore, C. gattii is responsible for the ongoing outbreak of cryptococcosis on Vancouver Island, Canada (Stephen, 2002; Hoang, 2004; Kidd, 2004) and the further spread of this outbreak to the Pacific Northwest of the United States (MacDougall, 2007; Upton, 2007). Taken together, these reports indicate that C. gattii also occur in more temperate climate zones.
Several molecular methods have identified four major genotypic groups within C. gattii (Ruma, 1996; Ellis, 2000; Chaturvedi, 2002; Biswas, 2003; Latouche, 2003; Meyer, 2003; Butler & Poulter, 2005; Diaz, 2005; Fraser, 2005; Kidd, 2005; Bovers, 2008b, c). An overview of these genotypic groups is given in Table 1. All these studies used nuclear regions to study the genotypic structure of C. gattii. Mitochondrial regions are useful genetic markers as well, because mitochondria evolve independently from the nuclear genome and thus provide an additional, independent dataset. In C. neoformans, mitochondria are usually uniparentally inherited from the MATa parent, although mitochondrial inheritance from the MATα parent may also occur at low frequency (Xu, 2000;; Yan & Xu, 2003; Toffaletti, 2004; Yan, 2004, 2007a, b). In our study, the partial nucleotide sequence of two mitochondrial regions, namely large ribosomal subunit RNA (MtLrRNA) and ATP synthase subunit 6 (ATP6), was investigated for 51 isolates representing all C. gattii genotypic groups. The sequences obtained were used for phylogenetic analyses and the resulting genealogies were compared with the genealogy obtained by analyses of six nuclear regions (Bovers, 2008b). Surprisingly, the analysis of the mitochondrial regions showed that the majority of amplified fragment length polymorphism AFLP4/VGI isolates studied possessed a mitochondrial genome that contained sequences that were partially identical to those found in AFLP6/VGII isolates. These findings are discussed and explained by assuming a process of mitochondrial recombination between the different genotypic groups of C. gattii.
1Overview of Cryptococcus gattii genotypes as identified by several molecular methods
| Serotype | AFLP genotype | Molecular genotype | IGS genotype | ITS genotype | Intein genotype |
| B/(C) | 4A/4B | VGI | 4a/4b/4c | 3/7 | ig-I |
| B/C | 5A/5B/5C | VGIII | 5 | 5 | ig-III |
| B/(C) | 6 | VGII | 3 | 4 | ig-II |
| B/C | 7 | VGIV | 6 | 6 | ig-IV |
| Serotype | AFLP genotype | Molecular genotype | IGS genotype | ITS genotype | Intein genotype |
| B/(C) | 4A/4B | VGI | 4a/4b/4c | 3/7 | ig-I |
| B/C | 5A/5B/5C | VGIII | 5 | 5 | ig-III |
| B/(C) | 6 | VGII | 3 | 4 | ig-II |
| B/C | 7 | VGIV | 6 | 6 | ig-IV |
1Overview of Cryptococcus gattii genotypes as identified by several molecular methods
| Serotype | AFLP genotype | Molecular genotype | IGS genotype | ITS genotype | Intein genotype |
| B/(C) | 4A/4B | VGI | 4a/4b/4c | 3/7 | ig-I |
| B/C | 5A/5B/5C | VGIII | 5 | 5 | ig-III |
| B/(C) | 6 | VGII | 3 | 4 | ig-II |
| B/C | 7 | VGIV | 6 | 6 | ig-IV |
| Serotype | AFLP genotype | Molecular genotype | IGS genotype | ITS genotype | Intein genotype |
| B/(C) | 4A/4B | VGI | 4a/4b/4c | 3/7 | ig-I |
| B/C | 5A/5B/5C | VGIII | 5 | 5 | ig-III |
| B/(C) | 6 | VGII | 3 | 4 | ig-II |
| B/C | 7 | VGIV | 6 | 6 | ig-IV |
Materials and methods
Isolates
Fifty-one haploid isolates belonging to all C. gattii genotypic groups from clinical (59%), veterinary (8%), environmental (24%), laboratory (6%) and unknown (3%) origin were analyzed (Table 2). Cryptococcus neoformans isolates (125.91, H99, WM714, CBS6886 and JEC20) were used as outgroup in phylogenetic analyses, as C. neoformans is the species that is most closely related to C. gattii (Fell, 2000; Kwon-Chung, 2002).
2AFLP genotype, molecular type, serotype and origin of Cryptococcus gattii and Cryptococcus neoformans isolates
| Isolate | Origin | Sero type | Mating type | Nuclear genotype* | AFLP geno type‡§ | Mole cular type¶ | MtLrRNA haplotype | ATP6 haplotype | References /sources | MtLrRNA | ATP6 | ITS | IGS | RPB1 | RPB2 | CNLAC1 | TEF1α |
| C. gattii AFLP4=VGI | | | |
| 48A (=CBS11230) | Lung of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544832 | EF544778 | EF211195 | AJ300934∥ | EF211403 | EF211523 | EF211643 | EF211758 |
| 503 2738 (=WM1251) | Human, Papua, New Guinea | B | α | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544833 | EF544779 | EF211196 | EF211302 | EF211404 | EF211524 | EF211644 | EF211759 |
| 56A (=CBS11231) | Gut of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544834 | EF544780 | EF211197 | AJ300932∥ | EF211405 | EF211525 | EF211645 | EF211760 |
| CBS883 (T) | Infected skin, syntype Cryptococcus hondurianus, Honduras | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544835 | EF544781 | EF211198 | EF211303 | EF211406 | EF211526 | EF211646 | EF211761 |
| CBS919 (T) | Meningoencephalic lesion, type strain of Torulopsis neoformans var. sheppei | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544836 | EF544782 | EF211199 | AJ300928∥ | EF211407 | EF211527 | EF211647 | EF211762 |
| CBS1622 | Man, Tumour, Lille, France | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102067** | EF102038** | EF102028** | EF102032** | EF102061** | EF102051** | EF102071** | EF102047** |
| CBS6289 (=CBS8273, =RV20186, =NIHB-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544837 | EF544783 | EF211200 | AJ300937∥ | EF211408 | EF211528 | EF211648 | EF211763 |
| CBS6290 | Man, Congo (Zaire) | B | α | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544838 | EF544784 | EF211201 | AJ300930∥ | EF211409 | EF211529 | EF211649 | EF211764 |
| CBS6992 (=NIH 17) | Man | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102068** | EF102039** | EF102029** | AJ300923∥ | EF102033** | EF102062** | EF102052** | EF102072** | EF102048** |
| CBS6998 (=NIH 365) | Human, Thailand | B | a | 4 | 4 | ND | 2 | 4 | Boekhout (1997) | EF544839 | EF544785 | EF211202 | AJ300925∥ | EF211410 | EF211530 | EF211650 | EF211765 |
| CBS7229 (T) | Meningitis, type strain Cryptococcus neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (1997) | EF544840 | EF544786 | EF211203 | AJ300926∥ | EF211411 | EF211531 | EF211651 | EF211766 |
| CBS7748 (=IFM50902) | Air in Eucalyptus camaldulensis hollow, Balranald, SA, Australia | B | α | 4 | 4B | VGI | 4 | 2 | Boekhout (1997) | EF544841 | EF544787 | EF211204 | AJ300927∥ | EF211412 | EF211532 | EF211652 | EF211767 |
| CBS8273 (=CBS6289, =RV20186, =NIH B-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4 | ND | 2 | 4 | Boekhout (1997) | EF544842 | EF544788 | EF211205 | EF211304 | EF211413 | EF211533 | EF211653 | EF211768 |
| E566 (=CBS11233) | E. camaldulensis tree #19 hollow 4, Renmark Australia | B | a | 4 | 4 | VGI | 1 | 2 | Halliday (2000) | EF544843 | EF544789 | EF211206 | EF211305 | EF211414 | EF211534 | EF211654 | EF211769 |
| RV20186 (=CBS6289, =CBS8273, =NIH B-3939) (T) | Cerebrospinal fluid, type strain of Cryptococcus gattii, Congo (Zaire) | B | a | 4A | 4A | ND | 2 | 4 | Gatti & Eeckels (1970) | EF544844 | EF544790 | EF211207 | EF211306 | EF211415 | EF211535 | EF211655 | EF211770 |
| RV54130 | Second isolate of C. neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544845 | EF544791 | EF211208 | EF211307 | EF211416 | EF211536 | EF211656 | EF211771 |
| WM176 | Eucalyptus citriodora,USA | B | α | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544846 | EF544792 | EF211209 | EF211308 | EF211417 | EF211537 | EF211657 | EF211772 |
| WM179 (R) | Immunocompetent human, reference strain of molecular type VGI, Sydney, Australia | B | α | 4 | 4 | VGI | 1 | 2 | Meyer (2003) | EF544847 | EF544793 | EF211210 | EF211309 | EF211418 | EF211538 | EF211658 | EF211773 |
| WM276 (=CBS10510) | Eucalyptus tereticornis, Mt. Annan, NSW, Australia | B | α | 4 | 4 | VGI | 2 | 1 | Kidd (2005) | EF544848 | EF544794 | EF211211 | EF211310 | EF211419 | EF211539 | EF211659 | EF211774 |
| WM830 | Immunocompetent, Human, Papua. New Guinea | B | a | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544849 | EF544795 | EF211212 | EF211311 | EF211420 | EF211540 | EF211660 | EF211775 |
| C. gattii AFLP5=VGIII |
| 380C | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (2001) | EF544850 | EF544796 | EF211213 | EF211312 | EF211421 | EF211541 | EF211661 | EF211776 |
| 384C | Patient | C | α | 5 | 5C | ND | 5 | 8 | Boekhout (2001) | EF544851 | EF544797 | EF211214 | EF211313 | EF211422 | EF211542 | EF211662 | EF211777 |
| CBS5758 | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544852 | EF544798 | EF211215 | AJ300929∥ | EF211423 | EF211543 | EF211663 | EF211778 |
| CBS6955 (T) (=NIH 191) | Human, type strain of Cryptococcus bacillisporus, CA, USA | C | a | 5 | 5C | ND | 5 | 7 | Boekhout (1997) | EF544853 | EF544799 | EF211216 | AJ300940∥ | EF211424 | EF211544 | EF211664 | EF211779 |
| CBS6993 (=NIH 18) | Human, CA, USA | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544854 | EF544800 | EF211217 | EF211314 | EF211425 | EF211545 | EF211665 | EF211780 |
| CBS6996 | Man | B | α | 5 | 5C | ND | 5 | 8 | Boekhout (1997) | EF544855 | EF544801 | EF211218 | AJ300939∥ | EF211426 | EF211546 | EF211666 | EF211781 |
| CBS8755 (=HOO58-I-682) | Detritus of almond tree, Colombia | C | α | 5 | 5A | ND | 5 | 5 | Boekhout (2001) | EF544856 | EF544802 | EF211219 | EF211315 | EF211427 | EF211547 | EF211667 | EF211782 |
| CN043 (=CBS11247) | Human, Auckland, New Zealand | B | α | 5 | 5 | VGIII | 5 | 6 | Katsu (2004) | EF544857 | EF544803 | EF211220 | EF211316 | EF211428 | EF211548 | EF211668 | EF211783 |
| WM161 (R) | Eucalyptus camaldulensis wood from hollow, reference strain of molecular type VGIII, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Meyer (2003) | EF544858 | EF544804 | EF211221 | EF211317 | EF211429 | EF211549 | EF211669 | EF211784 |
| WM726 | Eucalyptus citriodora, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544859 | EF544805 | EF211222 | EF211318 | EF211430 | EF211550 | EF211670 | EF211785 |
| WM728 | Eucalyptus sp. debris from car park of zoo, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544860 | EF544806 | EF211223 | EF211319 | EF211431 | EF211551 | EF211671 | EF211786 |
| C. gattii AFLP6=VGII |
| A1M F2866 (=CBS11240) | Dead wild Dall's porpoise lymph node, Shores of Gulf Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544861 | EF544807 | EF211224 | EF211320 | EF211432 | EF211552 | EF211672 | EF211787 |
| A1M F2932 (=CBS11235) | Immunocompetent male, lung, Kelowna, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544862 | EF544808 | EF211225 | EF211321 | EF211433 | EF211553 | EF211673 | EF211788 |
| A1M R265 (=CBS10514) | Immunocompetent male, Duncan, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544863 | EF544809 | EF211226 | EF211322 | EF211434 | EF211554 | EF211674 | EF211789 |
| A1M R269 (=CBS11236) | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544864 | EF544810 | EF211227 | EF211323 | EF211435 | EF211555 | EF211675 | EF211790 |
| A1M R271 (=CBS11237) | Immunocompetent male, Nanoose Bay, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544865 | EF544811 | EF211228 | EF211324 | EF211436 | EF211556 | EF211676 | EF211791 |
| A1M R368 (=CBS11238) | Immunocompetent male, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544866 | EF544812 | EF211229 | EF211325 | EF211437 | EF211557 | EF211677 | EF211792 |
| A1M R406 (=CBS11239) | Immunocompetent female, Nanaimo, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544867 | EF544813 | EF211230 | EF211326 | EF211438 | EF211558 | EF211678 | EF211793 |
| A1M R409 | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544868 | EF544814 | EF211231 | EF211327 | EF211439 | EF211559 | EF211679 | EF211794 |
| CBS1930 | Sick goat, Aruba | B | a | 6 | 6B | ND | 1 | 2 | Boekhout (1997) | EF544869 | EF544815 | EF211232 | AJ300919∥ | EF211440 | EF211560 | EF211680 | EF211795 |
| CBS6956 (=NIH 444, =ATCC32609) | Immunocompetent human, Seattle, WA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544870 | EF544816 | EF211233 | AJ300920∥ | EF211441 | EF211561 | EF211681 | EF211796 |
| CBS7750 | Eucalyptus camaldulensis bark debris, San Francisco, CA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544871 | EF544817 | EF211234 | AJ300922∥ | EF211442 | EF211562 | EF211682 | EF211797 |
| CBS8684 | Nest of wasp, Uruguay | B | α | 6 | 6B | ND | 1 | 2 | Boekhout (2001) | EF544872 | EF544818 | EF211235 | EF211328 | EF211443 | EF211563 | EF211683 | EF211798 |
| HEC11102 | Human, Rio de Janeiro, Brazil | B | a | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544873 | EF544819 | EF211236 | EF211329 | EF211444 | EF211564 | EF211684 | EF211799 |
| ICB184 (=MTPI2, =CBS11251) | Hollow trees, Piaui, Brazil | B | α | 6 | 6B | ND | 1 | 2 | Barreto de Oliveira (2004) | EF544874 | EF544820 | EF211237 | EF211330 | EF211445 | EF211565 | EF211685 | EF211800 |
| RAM2 | Eucalyptus camaldulensis, Arnhemland, NT, Australia | B | α | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544875 | EF544821 | EF211238 | EF211331 | EF211446 | EF211566 | EF211686 | EF211801 |
| WM178 (=IFM 50894) (R) | Immunocompetent human, lung, reference strain of molecular type VGII, Sydney, Australia | B | α | 6 | 6A | VGII | 1 | 2 | Meyer (2003) | EF544876 | EF544822 | EF211239 | EF211332 | EF211447 | EF211567 | EF211687 | EF211802 |
| C. gattii AFLP7=VGIV |
| B-5742 | Human, Punjab, India | C | α | 7 | 7 | VGIV | 3 | 9 | Katsu (2004) | EF544877 | EF544823 | EF211240 | EF211333 | EF211448 | EF211568 | EF211688 | EF211803 |
| B-5748 | HIV positive patient, India | C | α | 7 | 7 | ND | 3 | 9 | Diaz & Fell (2005) | EF544878 | EF544824 | EF211241 | EF211334 | EF211449 | EF211569 | EF211689 | EF211804 |
| M27055 | Clinical, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Latouche (2002) | EF544879 | EF544825 | EF211242 | EF211335 | EF211450 | EF211570 | EF211690 | EF211805 |
| WM779 (=IFM50896) (R) | Cheetah, reference strain of molecular type VGIV, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Meyer (2003) | EF544880 | EF544826 | EF211243 | EF211336 | EF211451 | EF211571 | EF211691 | EF211806 |
| C. neoformans var. grubii outgroup isolates |
| 125.91 (=CBS10512) | Cryptococcal meningitis patient, Tanzania | A | a | 1 | 1 | ND | NA | NA | Lengeler (2002) | EF544827 | EF544773 | EF211129 | EF211249 | EF211337 | EF211457 | EF211577 | EF211692 |
| H99 (=CBS8710, =CBS10515) (T) | Patient with Hodgkin's disease, type strain of Cryptococcus neoformans var. grubii, NY, USA | A | α | 1 | 1 | VNI | NA | NA | Franzot (1999) | EF544828 | EF544774 | EF211146 | EF211264 | EF211354 | EF211474 | EF211594 | EF211709 |
| WM714 | Cat paranasal, Australia | A | α | 1B | 1B | ND | NA | NA | Boekhout (2001) | EF544829 | EF544775 | EF211174 | EF211289 | EF211382 | EF211502 | EF211622 | EF211737 |
| C. neoformans var. neoformans outgroup isolates |
| CBS6886 (=NIH 430) | Dropping of pigeon | D | α | 2 | 2 | ND | NA | NA | Boekhout (1997) | EF544830 | EF544776 | EF211182 | AJ300886∥ | EF211390 | EF211510 | EF211630 | EF211745 |
| JEC20 (=CBS10511, =NIH-B4476) | Congenic pair with JEC21 that differs only in matingtype | D | a | 2 | 2 | ND | NA | NA | Kwon-Chung (1992) | EF544831 | EF544777 | EF211189 | EF211296 | EF211397 | EF211517 | EF211637 | EF211752 |
| Isolate | Origin | Sero type | Mating type | Nuclear genotype* | AFLP geno type‡§ | Mole cular type¶ | MtLrRNA haplotype | ATP6 haplotype | References /sources | MtLrRNA | ATP6 | ITS | IGS | RPB1 | RPB2 | CNLAC1 | TEF1α |
| C. gattii AFLP4=VGI | | | |
| 48A (=CBS11230) | Lung of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544832 | EF544778 | EF211195 | AJ300934∥ | EF211403 | EF211523 | EF211643 | EF211758 |
| 503 2738 (=WM1251) | Human, Papua, New Guinea | B | α | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544833 | EF544779 | EF211196 | EF211302 | EF211404 | EF211524 | EF211644 | EF211759 |
| 56A (=CBS11231) | Gut of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544834 | EF544780 | EF211197 | AJ300932∥ | EF211405 | EF211525 | EF211645 | EF211760 |
| CBS883 (T) | Infected skin, syntype Cryptococcus hondurianus, Honduras | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544835 | EF544781 | EF211198 | EF211303 | EF211406 | EF211526 | EF211646 | EF211761 |
| CBS919 (T) | Meningoencephalic lesion, type strain of Torulopsis neoformans var. sheppei | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544836 | EF544782 | EF211199 | AJ300928∥ | EF211407 | EF211527 | EF211647 | EF211762 |
| CBS1622 | Man, Tumour, Lille, France | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102067** | EF102038** | EF102028** | EF102032** | EF102061** | EF102051** | EF102071** | EF102047** |
| CBS6289 (=CBS8273, =RV20186, =NIHB-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544837 | EF544783 | EF211200 | AJ300937∥ | EF211408 | EF211528 | EF211648 | EF211763 |
| CBS6290 | Man, Congo (Zaire) | B | α | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544838 | EF544784 | EF211201 | AJ300930∥ | EF211409 | EF211529 | EF211649 | EF211764 |
| CBS6992 (=NIH 17) | Man | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102068** | EF102039** | EF102029** | AJ300923∥ | EF102033** | EF102062** | EF102052** | EF102072** | EF102048** |
| CBS6998 (=NIH 365) | Human, Thailand | B | a | 4 | 4 | ND | 2 | 4 | Boekhout (1997) | EF544839 | EF544785 | EF211202 | AJ300925∥ | EF211410 | EF211530 | EF211650 | EF211765 |
| CBS7229 (T) | Meningitis, type strain Cryptococcus neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (1997) | EF544840 | EF544786 | EF211203 | AJ300926∥ | EF211411 | EF211531 | EF211651 | EF211766 |
| CBS7748 (=IFM50902) | Air in Eucalyptus camaldulensis hollow, Balranald, SA, Australia | B | α | 4 | 4B | VGI | 4 | 2 | Boekhout (1997) | EF544841 | EF544787 | EF211204 | AJ300927∥ | EF211412 | EF211532 | EF211652 | EF211767 |
| CBS8273 (=CBS6289, =RV20186, =NIH B-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4 | ND | 2 | 4 | Boekhout (1997) | EF544842 | EF544788 | EF211205 | EF211304 | EF211413 | EF211533 | EF211653 | EF211768 |
| E566 (=CBS11233) | E. camaldulensis tree #19 hollow 4, Renmark Australia | B | a | 4 | 4 | VGI | 1 | 2 | Halliday (2000) | EF544843 | EF544789 | EF211206 | EF211305 | EF211414 | EF211534 | EF211654 | EF211769 |
| RV20186 (=CBS6289, =CBS8273, =NIH B-3939) (T) | Cerebrospinal fluid, type strain of Cryptococcus gattii, Congo (Zaire) | B | a | 4A | 4A | ND | 2 | 4 | Gatti & Eeckels (1970) | EF544844 | EF544790 | EF211207 | EF211306 | EF211415 | EF211535 | EF211655 | EF211770 |
| RV54130 | Second isolate of C. neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544845 | EF544791 | EF211208 | EF211307 | EF211416 | EF211536 | EF211656 | EF211771 |
| WM176 | Eucalyptus citriodora,USA | B | α | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544846 | EF544792 | EF211209 | EF211308 | EF211417 | EF211537 | EF211657 | EF211772 |
| WM179 (R) | Immunocompetent human, reference strain of molecular type VGI, Sydney, Australia | B | α | 4 | 4 | VGI | 1 | 2 | Meyer (2003) | EF544847 | EF544793 | EF211210 | EF211309 | EF211418 | EF211538 | EF211658 | EF211773 |
| WM276 (=CBS10510) | Eucalyptus tereticornis, Mt. Annan, NSW, Australia | B | α | 4 | 4 | VGI | 2 | 1 | Kidd (2005) | EF544848 | EF544794 | EF211211 | EF211310 | EF211419 | EF211539 | EF211659 | EF211774 |
| WM830 | Immunocompetent, Human, Papua. New Guinea | B | a | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544849 | EF544795 | EF211212 | EF211311 | EF211420 | EF211540 | EF211660 | EF211775 |
| C. gattii AFLP5=VGIII |
| 380C | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (2001) | EF544850 | EF544796 | EF211213 | EF211312 | EF211421 | EF211541 | EF211661 | EF211776 |
| 384C | Patient | C | α | 5 | 5C | ND | 5 | 8 | Boekhout (2001) | EF544851 | EF544797 | EF211214 | EF211313 | EF211422 | EF211542 | EF211662 | EF211777 |
| CBS5758 | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544852 | EF544798 | EF211215 | AJ300929∥ | EF211423 | EF211543 | EF211663 | EF211778 |
| CBS6955 (T) (=NIH 191) | Human, type strain of Cryptococcus bacillisporus, CA, USA | C | a | 5 | 5C | ND | 5 | 7 | Boekhout (1997) | EF544853 | EF544799 | EF211216 | AJ300940∥ | EF211424 | EF211544 | EF211664 | EF211779 |
| CBS6993 (=NIH 18) | Human, CA, USA | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544854 | EF544800 | EF211217 | EF211314 | EF211425 | EF211545 | EF211665 | EF211780 |
| CBS6996 | Man | B | α | 5 | 5C | ND | 5 | 8 | Boekhout (1997) | EF544855 | EF544801 | EF211218 | AJ300939∥ | EF211426 | EF211546 | EF211666 | EF211781 |
| CBS8755 (=HOO58-I-682) | Detritus of almond tree, Colombia | C | α | 5 | 5A | ND | 5 | 5 | Boekhout (2001) | EF544856 | EF544802 | EF211219 | EF211315 | EF211427 | EF211547 | EF211667 | EF211782 |
| CN043 (=CBS11247) | Human, Auckland, New Zealand | B | α | 5 | 5 | VGIII | 5 | 6 | Katsu (2004) | EF544857 | EF544803 | EF211220 | EF211316 | EF211428 | EF211548 | EF211668 | EF211783 |
| WM161 (R) | Eucalyptus camaldulensis wood from hollow, reference strain of molecular type VGIII, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Meyer (2003) | EF544858 | EF544804 | EF211221 | EF211317 | EF211429 | EF211549 | EF211669 | EF211784 |
| WM726 | Eucalyptus citriodora, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544859 | EF544805 | EF211222 | EF211318 | EF211430 | EF211550 | EF211670 | EF211785 |
| WM728 | Eucalyptus sp. debris from car park of zoo, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544860 | EF544806 | EF211223 | EF211319 | EF211431 | EF211551 | EF211671 | EF211786 |
| C. gattii AFLP6=VGII |
| A1M F2866 (=CBS11240) | Dead wild Dall's porpoise lymph node, Shores of Gulf Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544861 | EF544807 | EF211224 | EF211320 | EF211432 | EF211552 | EF211672 | EF211787 |
| A1M F2932 (=CBS11235) | Immunocompetent male, lung, Kelowna, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544862 | EF544808 | EF211225 | EF211321 | EF211433 | EF211553 | EF211673 | EF211788 |
| A1M R265 (=CBS10514) | Immunocompetent male, Duncan, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544863 | EF544809 | EF211226 | EF211322 | EF211434 | EF211554 | EF211674 | EF211789 |
| A1M R269 (=CBS11236) | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544864 | EF544810 | EF211227 | EF211323 | EF211435 | EF211555 | EF211675 | EF211790 |
| A1M R271 (=CBS11237) | Immunocompetent male, Nanoose Bay, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544865 | EF544811 | EF211228 | EF211324 | EF211436 | EF211556 | EF211676 | EF211791 |
| A1M R368 (=CBS11238) | Immunocompetent male, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544866 | EF544812 | EF211229 | EF211325 | EF211437 | EF211557 | EF211677 | EF211792 |
| A1M R406 (=CBS11239) | Immunocompetent female, Nanaimo, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544867 | EF544813 | EF211230 | EF211326 | EF211438 | EF211558 | EF211678 | EF211793 |
| A1M R409 | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544868 | EF544814 | EF211231 | EF211327 | EF211439 | EF211559 | EF211679 | EF211794 |
| CBS1930 | Sick goat, Aruba | B | a | 6 | 6B | ND | 1 | 2 | Boekhout (1997) | EF544869 | EF544815 | EF211232 | AJ300919∥ | EF211440 | EF211560 | EF211680 | EF211795 |
| CBS6956 (=NIH 444, =ATCC32609) | Immunocompetent human, Seattle, WA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544870 | EF544816 | EF211233 | AJ300920∥ | EF211441 | EF211561 | EF211681 | EF211796 |
| CBS7750 | Eucalyptus camaldulensis bark debris, San Francisco, CA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544871 | EF544817 | EF211234 | AJ300922∥ | EF211442 | EF211562 | EF211682 | EF211797 |
| CBS8684 | Nest of wasp, Uruguay | B | α | 6 | 6B | ND | 1 | 2 | Boekhout (2001) | EF544872 | EF544818 | EF211235 | EF211328 | EF211443 | EF211563 | EF211683 | EF211798 |
| HEC11102 | Human, Rio de Janeiro, Brazil | B | a | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544873 | EF544819 | EF211236 | EF211329 | EF211444 | EF211564 | EF211684 | EF211799 |
| ICB184 (=MTPI2, =CBS11251) | Hollow trees, Piaui, Brazil | B | α | 6 | 6B | ND | 1 | 2 | Barreto de Oliveira (2004) | EF544874 | EF544820 | EF211237 | EF211330 | EF211445 | EF211565 | EF211685 | EF211800 |
| RAM2 | Eucalyptus camaldulensis, Arnhemland, NT, Australia | B | α | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544875 | EF544821 | EF211238 | EF211331 | EF211446 | EF211566 | EF211686 | EF211801 |
| WM178 (=IFM 50894) (R) | Immunocompetent human, lung, reference strain of molecular type VGII, Sydney, Australia | B | α | 6 | 6A | VGII | 1 | 2 | Meyer (2003) | EF544876 | EF544822 | EF211239 | EF211332 | EF211447 | EF211567 | EF211687 | EF211802 |
| C. gattii AFLP7=VGIV |
| B-5742 | Human, Punjab, India | C | α | 7 | 7 | VGIV | 3 | 9 | Katsu (2004) | EF544877 | EF544823 | EF211240 | EF211333 | EF211448 | EF211568 | EF211688 | EF211803 |
| B-5748 | HIV positive patient, India | C | α | 7 | 7 | ND | 3 | 9 | Diaz & Fell (2005) | EF544878 | EF544824 | EF211241 | EF211334 | EF211449 | EF211569 | EF211689 | EF211804 |
| M27055 | Clinical, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Latouche (2002) | EF544879 | EF544825 | EF211242 | EF211335 | EF211450 | EF211570 | EF211690 | EF211805 |
| WM779 (=IFM50896) (R) | Cheetah, reference strain of molecular type VGIV, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Meyer (2003) | EF544880 | EF544826 | EF211243 | EF211336 | EF211451 | EF211571 | EF211691 | EF211806 |
| C. neoformans var. grubii outgroup isolates |
| 125.91 (=CBS10512) | Cryptococcal meningitis patient, Tanzania | A | a | 1 | 1 | ND | NA | NA | Lengeler (2002) | EF544827 | EF544773 | EF211129 | EF211249 | EF211337 | EF211457 | EF211577 | EF211692 |
| H99 (=CBS8710, =CBS10515) (T) | Patient with Hodgkin's disease, type strain of Cryptococcus neoformans var. grubii, NY, USA | A | α | 1 | 1 | VNI | NA | NA | Franzot (1999) | EF544828 | EF544774 | EF211146 | EF211264 | EF211354 | EF211474 | EF211594 | EF211709 |
| WM714 | Cat paranasal, Australia | A | α | 1B | 1B | ND | NA | NA | Boekhout (2001) | EF544829 | EF544775 | EF211174 | EF211289 | EF211382 | EF211502 | EF211622 | EF211737 |
| C. neoformans var. neoformans outgroup isolates |
| CBS6886 (=NIH 430) | Dropping of pigeon | D | α | 2 | 2 | ND | NA | NA | Boekhout (1997) | EF544830 | EF544776 | EF211182 | AJ300886∥ | EF211390 | EF211510 | EF211630 | EF211745 |
| JEC20 (=CBS10511, =NIH-B4476) | Congenic pair with JEC21 that differs only in matingtype | D | a | 2 | 2 | ND | NA | NA | Kwon-Chung (1992) | EF544831 | EF544777 | EF211189 | EF211296 | EF211397 | EF211517 | EF211637 | EF211752 |
2AFLP genotype, molecular type, serotype and origin of Cryptococcus gattii and Cryptococcus neoformans isolates
| Isolate | Origin | Sero type | Mating type | Nuclear genotype* | AFLP geno type‡§ | Mole cular type¶ | MtLrRNA haplotype | ATP6 haplotype | References /sources | MtLrRNA | ATP6 | ITS | IGS | RPB1 | RPB2 | CNLAC1 | TEF1α |
| C. gattii AFLP4=VGI | | | |
| 48A (=CBS11230) | Lung of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544832 | EF544778 | EF211195 | AJ300934∥ | EF211403 | EF211523 | EF211643 | EF211758 |
| 503 2738 (=WM1251) | Human, Papua, New Guinea | B | α | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544833 | EF544779 | EF211196 | EF211302 | EF211404 | EF211524 | EF211644 | EF211759 |
| 56A (=CBS11231) | Gut of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544834 | EF544780 | EF211197 | AJ300932∥ | EF211405 | EF211525 | EF211645 | EF211760 |
| CBS883 (T) | Infected skin, syntype Cryptococcus hondurianus, Honduras | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544835 | EF544781 | EF211198 | EF211303 | EF211406 | EF211526 | EF211646 | EF211761 |
| CBS919 (T) | Meningoencephalic lesion, type strain of Torulopsis neoformans var. sheppei | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544836 | EF544782 | EF211199 | AJ300928∥ | EF211407 | EF211527 | EF211647 | EF211762 |
| CBS1622 | Man, Tumour, Lille, France | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102067** | EF102038** | EF102028** | EF102032** | EF102061** | EF102051** | EF102071** | EF102047** |
| CBS6289 (=CBS8273, =RV20186, =NIHB-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544837 | EF544783 | EF211200 | AJ300937∥ | EF211408 | EF211528 | EF211648 | EF211763 |
| CBS6290 | Man, Congo (Zaire) | B | α | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544838 | EF544784 | EF211201 | AJ300930∥ | EF211409 | EF211529 | EF211649 | EF211764 |
| CBS6992 (=NIH 17) | Man | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102068** | EF102039** | EF102029** | AJ300923∥ | EF102033** | EF102062** | EF102052** | EF102072** | EF102048** |
| CBS6998 (=NIH 365) | Human, Thailand | B | a | 4 | 4 | ND | 2 | 4 | Boekhout (1997) | EF544839 | EF544785 | EF211202 | AJ300925∥ | EF211410 | EF211530 | EF211650 | EF211765 |
| CBS7229 (T) | Meningitis, type strain Cryptococcus neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (1997) | EF544840 | EF544786 | EF211203 | AJ300926∥ | EF211411 | EF211531 | EF211651 | EF211766 |
| CBS7748 (=IFM50902) | Air in Eucalyptus camaldulensis hollow, Balranald, SA, Australia | B | α | 4 | 4B | VGI | 4 | 2 | Boekhout (1997) | EF544841 | EF544787 | EF211204 | AJ300927∥ | EF211412 | EF211532 | EF211652 | EF211767 |
| CBS8273 (=CBS6289, =RV20186, =NIH B-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4 | ND | 2 | 4 | Boekhout (1997) | EF544842 | EF544788 | EF211205 | EF211304 | EF211413 | EF211533 | EF211653 | EF211768 |
| E566 (=CBS11233) | E. camaldulensis tree #19 hollow 4, Renmark Australia | B | a | 4 | 4 | VGI | 1 | 2 | Halliday (2000) | EF544843 | EF544789 | EF211206 | EF211305 | EF211414 | EF211534 | EF211654 | EF211769 |
| RV20186 (=CBS6289, =CBS8273, =NIH B-3939) (T) | Cerebrospinal fluid, type strain of Cryptococcus gattii, Congo (Zaire) | B | a | 4A | 4A | ND | 2 | 4 | Gatti & Eeckels (1970) | EF544844 | EF544790 | EF211207 | EF211306 | EF211415 | EF211535 | EF211655 | EF211770 |
| RV54130 | Second isolate of C. neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544845 | EF544791 | EF211208 | EF211307 | EF211416 | EF211536 | EF211656 | EF211771 |
| WM176 | Eucalyptus citriodora,USA | B | α | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544846 | EF544792 | EF211209 | EF211308 | EF211417 | EF211537 | EF211657 | EF211772 |
| WM179 (R) | Immunocompetent human, reference strain of molecular type VGI, Sydney, Australia | B | α | 4 | 4 | VGI | 1 | 2 | Meyer (2003) | EF544847 | EF544793 | EF211210 | EF211309 | EF211418 | EF211538 | EF211658 | EF211773 |
| WM276 (=CBS10510) | Eucalyptus tereticornis, Mt. Annan, NSW, Australia | B | α | 4 | 4 | VGI | 2 | 1 | Kidd (2005) | EF544848 | EF544794 | EF211211 | EF211310 | EF211419 | EF211539 | EF211659 | EF211774 |
| WM830 | Immunocompetent, Human, Papua. New Guinea | B | a | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544849 | EF544795 | EF211212 | EF211311 | EF211420 | EF211540 | EF211660 | EF211775 |
| C. gattii AFLP5=VGIII |
| 380C | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (2001) | EF544850 | EF544796 | EF211213 | EF211312 | EF211421 | EF211541 | EF211661 | EF211776 |
| 384C | Patient | C | α | 5 | 5C | ND | 5 | 8 | Boekhout (2001) | EF544851 | EF544797 | EF211214 | EF211313 | EF211422 | EF211542 | EF211662 | EF211777 |
| CBS5758 | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544852 | EF544798 | EF211215 | AJ300929∥ | EF211423 | EF211543 | EF211663 | EF211778 |
| CBS6955 (T) (=NIH 191) | Human, type strain of Cryptococcus bacillisporus, CA, USA | C | a | 5 | 5C | ND | 5 | 7 | Boekhout (1997) | EF544853 | EF544799 | EF211216 | AJ300940∥ | EF211424 | EF211544 | EF211664 | EF211779 |
| CBS6993 (=NIH 18) | Human, CA, USA | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544854 | EF544800 | EF211217 | EF211314 | EF211425 | EF211545 | EF211665 | EF211780 |
| CBS6996 | Man | B | α | 5 | 5C | ND | 5 | 8 | Boekhout (1997) | EF544855 | EF544801 | EF211218 | AJ300939∥ | EF211426 | EF211546 | EF211666 | EF211781 |
| CBS8755 (=HOO58-I-682) | Detritus of almond tree, Colombia | C | α | 5 | 5A | ND | 5 | 5 | Boekhout (2001) | EF544856 | EF544802 | EF211219 | EF211315 | EF211427 | EF211547 | EF211667 | EF211782 |
| CN043 (=CBS11247) | Human, Auckland, New Zealand | B | α | 5 | 5 | VGIII | 5 | 6 | Katsu (2004) | EF544857 | EF544803 | EF211220 | EF211316 | EF211428 | EF211548 | EF211668 | EF211783 |
| WM161 (R) | Eucalyptus camaldulensis wood from hollow, reference strain of molecular type VGIII, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Meyer (2003) | EF544858 | EF544804 | EF211221 | EF211317 | EF211429 | EF211549 | EF211669 | EF211784 |
| WM726 | Eucalyptus citriodora, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544859 | EF544805 | EF211222 | EF211318 | EF211430 | EF211550 | EF211670 | EF211785 |
| WM728 | Eucalyptus sp. debris from car park of zoo, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544860 | EF544806 | EF211223 | EF211319 | EF211431 | EF211551 | EF211671 | EF211786 |
| C. gattii AFLP6=VGII |
| A1M F2866 (=CBS11240) | Dead wild Dall's porpoise lymph node, Shores of Gulf Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544861 | EF544807 | EF211224 | EF211320 | EF211432 | EF211552 | EF211672 | EF211787 |
| A1M F2932 (=CBS11235) | Immunocompetent male, lung, Kelowna, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544862 | EF544808 | EF211225 | EF211321 | EF211433 | EF211553 | EF211673 | EF211788 |
| A1M R265 (=CBS10514) | Immunocompetent male, Duncan, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544863 | EF544809 | EF211226 | EF211322 | EF211434 | EF211554 | EF211674 | EF211789 |
| A1M R269 (=CBS11236) | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544864 | EF544810 | EF211227 | EF211323 | EF211435 | EF211555 | EF211675 | EF211790 |
| A1M R271 (=CBS11237) | Immunocompetent male, Nanoose Bay, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544865 | EF544811 | EF211228 | EF211324 | EF211436 | EF211556 | EF211676 | EF211791 |
| A1M R368 (=CBS11238) | Immunocompetent male, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544866 | EF544812 | EF211229 | EF211325 | EF211437 | EF211557 | EF211677 | EF211792 |
| A1M R406 (=CBS11239) | Immunocompetent female, Nanaimo, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544867 | EF544813 | EF211230 | EF211326 | EF211438 | EF211558 | EF211678 | EF211793 |
| A1M R409 | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544868 | EF544814 | EF211231 | EF211327 | EF211439 | EF211559 | EF211679 | EF211794 |
| CBS1930 | Sick goat, Aruba | B | a | 6 | 6B | ND | 1 | 2 | Boekhout (1997) | EF544869 | EF544815 | EF211232 | AJ300919∥ | EF211440 | EF211560 | EF211680 | EF211795 |
| CBS6956 (=NIH 444, =ATCC32609) | Immunocompetent human, Seattle, WA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544870 | EF544816 | EF211233 | AJ300920∥ | EF211441 | EF211561 | EF211681 | EF211796 |
| CBS7750 | Eucalyptus camaldulensis bark debris, San Francisco, CA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544871 | EF544817 | EF211234 | AJ300922∥ | EF211442 | EF211562 | EF211682 | EF211797 |
| CBS8684 | Nest of wasp, Uruguay | B | α | 6 | 6B | ND | 1 | 2 | Boekhout (2001) | EF544872 | EF544818 | EF211235 | EF211328 | EF211443 | EF211563 | EF211683 | EF211798 |
| HEC11102 | Human, Rio de Janeiro, Brazil | B | a | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544873 | EF544819 | EF211236 | EF211329 | EF211444 | EF211564 | EF211684 | EF211799 |
| ICB184 (=MTPI2, =CBS11251) | Hollow trees, Piaui, Brazil | B | α | 6 | 6B | ND | 1 | 2 | Barreto de Oliveira (2004) | EF544874 | EF544820 | EF211237 | EF211330 | EF211445 | EF211565 | EF211685 | EF211800 |
| RAM2 | Eucalyptus camaldulensis, Arnhemland, NT, Australia | B | α | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544875 | EF544821 | EF211238 | EF211331 | EF211446 | EF211566 | EF211686 | EF211801 |
| WM178 (=IFM 50894) (R) | Immunocompetent human, lung, reference strain of molecular type VGII, Sydney, Australia | B | α | 6 | 6A | VGII | 1 | 2 | Meyer (2003) | EF544876 | EF544822 | EF211239 | EF211332 | EF211447 | EF211567 | EF211687 | EF211802 |
| C. gattii AFLP7=VGIV |
| B-5742 | Human, Punjab, India | C | α | 7 | 7 | VGIV | 3 | 9 | Katsu (2004) | EF544877 | EF544823 | EF211240 | EF211333 | EF211448 | EF211568 | EF211688 | EF211803 |
| B-5748 | HIV positive patient, India | C | α | 7 | 7 | ND | 3 | 9 | Diaz & Fell (2005) | EF544878 | EF544824 | EF211241 | EF211334 | EF211449 | EF211569 | EF211689 | EF211804 |
| M27055 | Clinical, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Latouche (2002) | EF544879 | EF544825 | EF211242 | EF211335 | EF211450 | EF211570 | EF211690 | EF211805 |
| WM779 (=IFM50896) (R) | Cheetah, reference strain of molecular type VGIV, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Meyer (2003) | EF544880 | EF544826 | EF211243 | EF211336 | EF211451 | EF211571 | EF211691 | EF211806 |
| C. neoformans var. grubii outgroup isolates |
| 125.91 (=CBS10512) | Cryptococcal meningitis patient, Tanzania | A | a | 1 | 1 | ND | NA | NA | Lengeler (2002) | EF544827 | EF544773 | EF211129 | EF211249 | EF211337 | EF211457 | EF211577 | EF211692 |
| H99 (=CBS8710, =CBS10515) (T) | Patient with Hodgkin's disease, type strain of Cryptococcus neoformans var. grubii, NY, USA | A | α | 1 | 1 | VNI | NA | NA | Franzot (1999) | EF544828 | EF544774 | EF211146 | EF211264 | EF211354 | EF211474 | EF211594 | EF211709 |
| WM714 | Cat paranasal, Australia | A | α | 1B | 1B | ND | NA | NA | Boekhout (2001) | EF544829 | EF544775 | EF211174 | EF211289 | EF211382 | EF211502 | EF211622 | EF211737 |
| C. neoformans var. neoformans outgroup isolates |
| CBS6886 (=NIH 430) | Dropping of pigeon | D | α | 2 | 2 | ND | NA | NA | Boekhout (1997) | EF544830 | EF544776 | EF211182 | AJ300886∥ | EF211390 | EF211510 | EF211630 | EF211745 |
| JEC20 (=CBS10511, =NIH-B4476) | Congenic pair with JEC21 that differs only in matingtype | D | a | 2 | 2 | ND | NA | NA | Kwon-Chung (1992) | EF544831 | EF544777 | EF211189 | EF211296 | EF211397 | EF211517 | EF211637 | EF211752 |
| Isolate | Origin | Sero type | Mating type | Nuclear genotype* | AFLP geno type‡§ | Mole cular type¶ | MtLrRNA haplotype | ATP6 haplotype | References /sources | MtLrRNA | ATP6 | ITS | IGS | RPB1 | RPB2 | CNLAC1 | TEF1α |
| C. gattii AFLP4=VGI | | | |
| 48A (=CBS11230) | Lung of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544832 | EF544778 | EF211195 | AJ300934∥ | EF211403 | EF211523 | EF211643 | EF211758 |
| 503 2738 (=WM1251) | Human, Papua, New Guinea | B | α | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544833 | EF544779 | EF211196 | EF211302 | EF211404 | EF211524 | EF211644 | EF211759 |
| 56A (=CBS11231) | Gut of a goat, Spain | B | α | 4A | 4A | ND | 2 | 4 | Diaz (2000) | EF544834 | EF544780 | EF211197 | AJ300932∥ | EF211405 | EF211525 | EF211645 | EF211760 |
| CBS883 (T) | Infected skin, syntype Cryptococcus hondurianus, Honduras | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544835 | EF544781 | EF211198 | EF211303 | EF211406 | EF211526 | EF211646 | EF211761 |
| CBS919 (T) | Meningoencephalic lesion, type strain of Torulopsis neoformans var. sheppei | B | α | 4 | 4B | ND | 4 | 2 | Boekhout (1997) | EF544836 | EF544782 | EF211199 | AJ300928∥ | EF211407 | EF211527 | EF211647 | EF211762 |
| CBS1622 | Man, Tumour, Lille, France | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102067** | EF102038** | EF102028** | EF102032** | EF102061** | EF102051** | EF102071** | EF102047** |
| CBS6289 (=CBS8273, =RV20186, =NIHB-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544837 | EF544783 | EF211200 | AJ300937∥ | EF211408 | EF211528 | EF211648 | EF211763 |
| CBS6290 | Man, Congo (Zaire) | B | α | 4A | 4A | ND | 2 | 4 | Boekhout (1997) | EF544838 | EF544784 | EF211201 | AJ300930∥ | EF211409 | EF211529 | EF211649 | EF211764 |
| CBS6992 (=NIH 17) | Man | B | α | 4 | 4B | ND | 2 | 3 | Boekhout (1997) | EF102068** | EF102039** | EF102029** | AJ300923∥ | EF102033** | EF102062** | EF102052** | EF102072** | EF102048** |
| CBS6998 (=NIH 365) | Human, Thailand | B | a | 4 | 4 | ND | 2 | 4 | Boekhout (1997) | EF544839 | EF544785 | EF211202 | AJ300925∥ | EF211410 | EF211530 | EF211650 | EF211765 |
| CBS7229 (T) | Meningitis, type strain Cryptococcus neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (1997) | EF544840 | EF544786 | EF211203 | AJ300926∥ | EF211411 | EF211531 | EF211651 | EF211766 |
| CBS7748 (=IFM50902) | Air in Eucalyptus camaldulensis hollow, Balranald, SA, Australia | B | α | 4 | 4B | VGI | 4 | 2 | Boekhout (1997) | EF544841 | EF544787 | EF211204 | AJ300927∥ | EF211412 | EF211532 | EF211652 | EF211767 |
| CBS8273 (=CBS6289, =RV20186, =NIH B-3939) | Subculture of type strain of Cryptococcus gattii (RV 20186) | B | a | 4A | 4 | ND | 2 | 4 | Boekhout (1997) | EF544842 | EF544788 | EF211205 | EF211304 | EF211413 | EF211533 | EF211653 | EF211768 |
| E566 (=CBS11233) | E. camaldulensis tree #19 hollow 4, Renmark Australia | B | a | 4 | 4 | VGI | 1 | 2 | Halliday (2000) | EF544843 | EF544789 | EF211206 | EF211305 | EF211414 | EF211534 | EF211654 | EF211769 |
| RV20186 (=CBS6289, =CBS8273, =NIH B-3939) (T) | Cerebrospinal fluid, type strain of Cryptococcus gattii, Congo (Zaire) | B | a | 4A | 4A | ND | 2 | 4 | Gatti & Eeckels (1970) | EF544844 | EF544790 | EF211207 | EF211306 | EF211415 | EF211535 | EF211655 | EF211770 |
| RV54130 | Second isolate of C. neoformans var. shanghaiensis, China | B | a | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544845 | EF544791 | EF211208 | EF211307 | EF211416 | EF211536 | EF211656 | EF211771 |
| WM176 | Eucalyptus citriodora,USA | B | α | 4 | 4B | ND | 2 | 1 | Boekhout (2001) | EF544846 | EF544792 | EF211209 | EF211308 | EF211417 | EF211537 | EF211657 | EF211772 |
| WM179 (R) | Immunocompetent human, reference strain of molecular type VGI, Sydney, Australia | B | α | 4 | 4 | VGI | 1 | 2 | Meyer (2003) | EF544847 | EF544793 | EF211210 | EF211309 | EF211418 | EF211538 | EF211658 | EF211773 |
| WM276 (=CBS10510) | Eucalyptus tereticornis, Mt. Annan, NSW, Australia | B | α | 4 | 4 | VGI | 2 | 1 | Kidd (2005) | EF544848 | EF544794 | EF211211 | EF211310 | EF211419 | EF211539 | EF211659 | EF211774 |
| WM830 | Immunocompetent, Human, Papua. New Guinea | B | a | 4 | 4 | VGI | 2 | 1 | Katsu (2004) | EF544849 | EF544795 | EF211212 | EF211311 | EF211420 | EF211540 | EF211660 | EF211775 |
| C. gattii AFLP5=VGIII |
| 380C | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (2001) | EF544850 | EF544796 | EF211213 | EF211312 | EF211421 | EF211541 | EF211661 | EF211776 |
| 384C | Patient | C | α | 5 | 5C | ND | 5 | 8 | Boekhout (2001) | EF544851 | EF544797 | EF211214 | EF211313 | EF211422 | EF211542 | EF211662 | EF211777 |
| CBS5758 | Unknown | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544852 | EF544798 | EF211215 | AJ300929∥ | EF211423 | EF211543 | EF211663 | EF211778 |
| CBS6955 (T) (=NIH 191) | Human, type strain of Cryptococcus bacillisporus, CA, USA | C | a | 5 | 5C | ND | 5 | 7 | Boekhout (1997) | EF544853 | EF544799 | EF211216 | AJ300940∥ | EF211424 | EF211544 | EF211664 | EF211779 |
| CBS6993 (=NIH 18) | Human, CA, USA | C | α | 5 | 5C | ND | 5 | 5 | Boekhout (1997) | EF544854 | EF544800 | EF211217 | EF211314 | EF211425 | EF211545 | EF211665 | EF211780 |
| CBS6996 | Man | B | α | 5 | 5C | ND | 5 | 8 | Boekhout (1997) | EF544855 | EF544801 | EF211218 | AJ300939∥ | EF211426 | EF211546 | EF211666 | EF211781 |
| CBS8755 (=HOO58-I-682) | Detritus of almond tree, Colombia | C | α | 5 | 5A | ND | 5 | 5 | Boekhout (2001) | EF544856 | EF544802 | EF211219 | EF211315 | EF211427 | EF211547 | EF211667 | EF211782 |
| CN043 (=CBS11247) | Human, Auckland, New Zealand | B | α | 5 | 5 | VGIII | 5 | 6 | Katsu (2004) | EF544857 | EF544803 | EF211220 | EF211316 | EF211428 | EF211548 | EF211668 | EF211783 |
| WM161 (R) | Eucalyptus camaldulensis wood from hollow, reference strain of molecular type VGIII, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Meyer (2003) | EF544858 | EF544804 | EF211221 | EF211317 | EF211429 | EF211549 | EF211669 | EF211784 |
| WM726 | Eucalyptus citriodora, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544859 | EF544805 | EF211222 | EF211318 | EF211430 | EF211550 | EF211670 | EF211785 |
| WM728 | Eucalyptus sp. debris from car park of zoo, San Diego, CA, USA | B | α | 5B | 5B | VGIII | 5 | 5 | Boekhout (2001) | EF544860 | EF544806 | EF211223 | EF211319 | EF211431 | EF211551 | EF211671 | EF211786 |
| C. gattii AFLP6=VGII |
| A1M F2866 (=CBS11240) | Dead wild Dall's porpoise lymph node, Shores of Gulf Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544861 | EF544807 | EF211224 | EF211320 | EF211432 | EF211552 | EF211672 | EF211787 |
| A1M F2932 (=CBS11235) | Immunocompetent male, lung, Kelowna, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544862 | EF544808 | EF211225 | EF211321 | EF211433 | EF211553 | EF211673 | EF211788 |
| A1M R265 (=CBS10514) | Immunocompetent male, Duncan, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544863 | EF544809 | EF211226 | EF211322 | EF211434 | EF211554 | EF211674 | EF211789 |
| A1M R269 (=CBS11236) | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544864 | EF544810 | EF211227 | EF211323 | EF211435 | EF211555 | EF211675 | EF211790 |
| A1M R271 (=CBS11237) | Immunocompetent male, Nanoose Bay, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544865 | EF544811 | EF211228 | EF211324 | EF211436 | EF211556 | EF211676 | EF211791 |
| A1M R368 (=CBS11238) | Immunocompetent male, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544866 | EF544812 | EF211229 | EF211325 | EF211437 | EF211557 | EF211677 | EF211792 |
| A1M R406 (=CBS11239) | Immunocompetent female, Nanaimo, Vancouver Island, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544867 | EF544813 | EF211230 | EF211326 | EF211438 | EF211558 | EF211678 | EF211793 |
| A1M R409 | Immunocompetent female, Victoria, Canada | B | α | 6 | 6A | VGIIa | 1 | 2 | Kidd (2004) | EF544868 | EF544814 | EF211231 | EF211327 | EF211439 | EF211559 | EF211679 | EF211794 |
| CBS1930 | Sick goat, Aruba | B | a | 6 | 6B | ND | 1 | 2 | Boekhout (1997) | EF544869 | EF544815 | EF211232 | AJ300919∥ | EF211440 | EF211560 | EF211680 | EF211795 |
| CBS6956 (=NIH 444, =ATCC32609) | Immunocompetent human, Seattle, WA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544870 | EF544816 | EF211233 | AJ300920∥ | EF211441 | EF211561 | EF211681 | EF211796 |
| CBS7750 | Eucalyptus camaldulensis bark debris, San Francisco, CA, USA | B | α | 6 | 6A | VGII | 1 | 2 | Boekhout (1997) | EF544871 | EF544817 | EF211234 | AJ300922∥ | EF211442 | EF211562 | EF211682 | EF211797 |
| CBS8684 | Nest of wasp, Uruguay | B | α | 6 | 6B | ND | 1 | 2 | Boekhout (2001) | EF544872 | EF544818 | EF211235 | EF211328 | EF211443 | EF211563 | EF211683 | EF211798 |
| HEC11102 | Human, Rio de Janeiro, Brazil | B | a | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544873 | EF544819 | EF211236 | EF211329 | EF211444 | EF211564 | EF211684 | EF211799 |
| ICB184 (=MTPI2, =CBS11251) | Hollow trees, Piaui, Brazil | B | α | 6 | 6B | ND | 1 | 2 | Barreto de Oliveira (2004) | EF544874 | EF544820 | EF211237 | EF211330 | EF211445 | EF211565 | EF211685 | EF211800 |
| RAM2 | Eucalyptus camaldulensis, Arnhemland, NT, Australia | B | α | 6 | 6B | VGII | 1 | 2 | Katsu (2004) | EF544875 | EF544821 | EF211238 | EF211331 | EF211446 | EF211566 | EF211686 | EF211801 |
| WM178 (=IFM 50894) (R) | Immunocompetent human, lung, reference strain of molecular type VGII, Sydney, Australia | B | α | 6 | 6A | VGII | 1 | 2 | Meyer (2003) | EF544876 | EF544822 | EF211239 | EF211332 | EF211447 | EF211567 | EF211687 | EF211802 |
| C. gattii AFLP7=VGIV |
| B-5742 | Human, Punjab, India | C | α | 7 | 7 | VGIV | 3 | 9 | Katsu (2004) | EF544877 | EF544823 | EF211240 | EF211333 | EF211448 | EF211568 | EF211688 | EF211803 |
| B-5748 | HIV positive patient, India | C | α | 7 | 7 | ND | 3 | 9 | Diaz & Fell (2005) | EF544878 | EF544824 | EF211241 | EF211334 | EF211449 | EF211569 | EF211689 | EF211804 |
| M27055 | Clinical, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Latouche (2002) | EF544879 | EF544825 | EF211242 | EF211335 | EF211450 | EF211570 | EF211690 | EF211805 |
| WM779 (=IFM50896) (R) | Cheetah, reference strain of molecular type VGIV, Johannesburg, South Africa | C | α | 7 | 7 | VGIV | 2 | 9 | Meyer (2003) | EF544880 | EF544826 | EF211243 | EF211336 | EF211451 | EF211571 | EF211691 | EF211806 |
| C. neoformans var. grubii outgroup isolates |
| 125.91 (=CBS10512) | Cryptococcal meningitis patient, Tanzania | A | a | 1 | 1 | ND | NA | NA | Lengeler (2002) | EF544827 | EF544773 | EF211129 | EF211249 | EF211337 | EF211457 | EF211577 | EF211692 |
| H99 (=CBS8710, =CBS10515) (T) | Patient with Hodgkin's disease, type strain of Cryptococcus neoformans var. grubii, NY, USA | A | α | 1 | 1 | VNI | NA | NA | Franzot (1999) | EF544828 | EF544774 | EF211146 | EF211264 | EF211354 | EF211474 | EF211594 | EF211709 |
| WM714 | Cat paranasal, Australia | A | α | 1B | 1B | ND | NA | NA | Boekhout (2001) | EF544829 | EF544775 | EF211174 | EF211289 | EF211382 | EF211502 | EF211622 | EF211737 |
| C. neoformans var. neoformans outgroup isolates |
| CBS6886 (=NIH 430) | Dropping of pigeon | D | α | 2 | 2 | ND | NA | NA | Boekhout (1997) | EF544830 | EF544776 | EF211182 | AJ300886∥ | EF211390 | EF211510 | EF211630 | EF211745 |
| JEC20 (=CBS10511, =NIH-B4476) | Congenic pair with JEC21 that differs only in matingtype | D | a | 2 | 2 | ND | NA | NA | Kwon-Chung (1992) | EF544831 | EF544777 | EF211189 | EF211296 | EF211397 | EF211517 | EF211637 | EF211752 |
Cultivation, DNA extraction, PCR reaction and sequencing
Cultivation and DNA extraction of C. neoformans and C. gattii isolates was carried out using an optimized protocol of Bolano (2001), which has been described previously (Bovers, 2006). PCR reactions were performed in a total volume of 50 μL containing 10 × PCR buffer (10 mM Tris-HCl, 50 mM KCl, 1.5 mM MgCl2, 0.01% gelatin, 0.1% Triton X-100, pH 8.3), 0.2 mM dNTPs, 0.6 μM of primer, 1.0 U Taq DNA polymerase (Gentaur, Brussels, Belgium) and 1–2 μL genomic DNA. Amplification of MtLrRNA was carried out using primers MtLrRNA-F (5′-GACCCTATGCAGCTTCTACTG-3′) and MtLrRNA-R (5′-TTATCCCTAGCGTAACTTTTATC-3′) (White, 1990). Amplification conditions were 94 °C for 3 min, followed by 35 cycles of 94 °C for 1 min, 50 °C for 1 min and 72 °C for 1 min, with a final extension step of 72 °C for 5 min. Mitochondrial ATP6 was amplified using primers ATP6-F (5′-ATTACATCTCCACTAGAACAATTC-3′) and ATP6-R (5′-AGTTCAATGGCATCCTTGATATAG-3′). Amplification conditions were 94 °C for 5 min, followed by 35 cycles of 94 °C for 1 min, 54 °C for 1 min and 72 °C for 2 min, with a final extension step of 72 °C for 5 min. Amplicons were purified with the GFX™ PCR DNA and Gel Band Purification Kit (Amersham Biosciences, Piscataway, NY) and used for sequencing. Sequencing reactions were performed with the BigDye v3.1 Chemistry kit (Applied Biosystems, Foster City, CA) using primers that had been used in the initial PCR reactions. Sequencing reactions were purified with Sephadex G-50 Superfine columns (Amersham Biosciences) and a MultiScreen HV plate (Millipore, Billerica, MA). An ABI 3700XL DNA analyzer (Applied Biosystems) was used to determine the sequences. GenBank accession numbers are listed in Table 2. Three isolates (CBS6289, CBS8273 and RV20186) were independently included in the analysis, but could be traced back to the same original isolate, and accordingly were found to have the same mitochondrial haplotypes. To confirm the correctness of our sequence data, especially related to the observed chimeric sequences, single colonies were selected, and processed for DNA extraction, PCR and sequence analysis. Furthermore, in an independent research project, a number of isolates from each lineage that were included in the present study were independently processed for the analysis of ATP6 sequences. Both gave the same sequences; hence, we are confident that we have been working with correct sequences (F. Hagen & T. Boekhout, unpublished data).
Alignment and phylogenetic analyses
The program seqman 5.03 (DNASTAR, Madison, WI) was used to assemble consensus sequences and these were checked manually. Sequences were aligned using clustalx version 1.8 (Thompson, 1997) and visually corrected using genedoc version 2.5.000 (http://www.nrbsc.org/downloads/). Neighbor-Joining (NJ) and Maximum Parsimony (MP) phylogenetic analyses were performed using paup* (phylogenetic analysis using parsimony) version 4.0b10 (Swofford, 2000). NJ analyses were carried out with the uncorrected (‘p’), Jukes–Cantor, Kimura two-parameter and HKY85 substitution models. Any ties that were encountered were broken randomly. Bootstrap analysis (Hillis & Bull, 1993) with 1000 replicates was used to determine the significance of branches. MP analyses were carried out (heuristic search, stepwise addition, random taxon addition, 1000 maximum trees) with tree bisection and reconstruction as the branch-swapping algorithm. All characters were unordered and of equal weight; gaps were treated both as missing and as a new character state. Bootstrap analysis was performed with 1000 replicates.
Results
ATP6
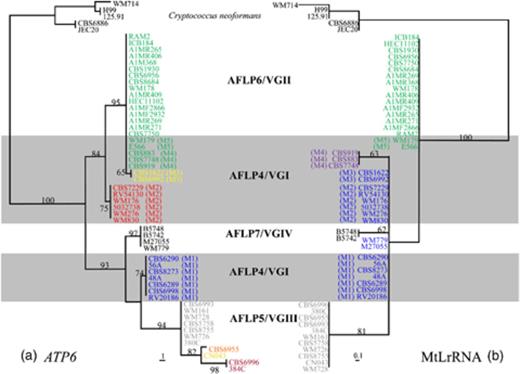
Analysis of partial ATP6 sequences showed that among 611 analyzed nucleotides, 34 nucleotides were polymorphic within C. gattii. Nine haplotypes could be identified (Fig. 1a). Haplotype 1 was present in AFLP4/VGI isolates 503 2738, CBS7229, RV54130, WM176, WM276 and WM830. All AFLP6/VGII isolates and AFLP4/VGI isolates WM179, E566, CBS883, CBS919 and CBS7748 possessed haplotype 2. Haplotype 3 was identified in AFLP4/VGI isolates CBS1622 and CBS6992. Haplotype 4 was found in AFLP4/VGI isolates 48A, 56A, CBS6289, CBS6290, CBS6998, CBS8273 and RV20186. AFLP5/VGIII isolates 380C, CBS5758, CBS6993, CBS8755, WM161, WM726 and WM728 possessed haplotype 5, whereas haplotype 6 was present in AFLP5/VGIII isolate CN043. Haplotype 7 was found in AFLP5/VGIII isolate CBS6955 and haplotype 8 was present in AFLP5/VGIII isolates 384C and CBS6996. All AFLP7/VGIV isolates possessed haplotype 9. The ATP6 haplotype of each isolate is indicated in Fig. 1a and Table 2. The alignment of the partial ATP6 sequences is given in Supporting Information, Fig. S1.
1
Phylogenetic tree of Cryptococcus gattii obtained by analysis of (a) partial ATP6 sequences. Presented is one of the four most parsimonious trees (length 83, consistency index 0.819, retention index 0.974) computed with gaps treated as missing data. Data consisted of 611 characters of which 64 characters were parsimony informative; (b) partial MtLrRNA sequences. Presented is one of three most parsimonious trees (length 15, consistency index 1.000, retention index 1.000) computed with gaps treated as missing data. Data consisted of 299 characters of which 13 characters were parsimony informative. Bootstrap values (1000 replicates) are indicated for the main branches. h, haplotype number; M, mitochondrial genotype. Haplotypes of both ATP6 and MtLrRNA are indicated by the colors given in Fig. 2. In addition, those not included in Fig. 2 are as follows: ATP6 – h5, grey; h6, ochre; h7, orange; h8, brown; h9, black; MtLrRNA – h3, black; h5, grey.
NJ and MP analyses showed a division of C. gattii into two clades. One clade consisted of haplotypes 1–3, and was found to be supported by bootstrap values ranging from 84% to 91% for NJ and MP analyses, respectively. The other clade contained haplotypes 4–9 and was found to be strongly supported by bootstrap values ranging from 93% to 99% for NJ and MP analyses. Haplotype 1 formed a separate cluster supported by bootstrap values ranging from 94% to 99% for NJ and from 75% to 76% for MP analyses. The cluster consisting of haplotypes 2 and 3 was strongly supported by bootstrap values ranging from 95% to 100% for NJ and MP analyses. Haplotype 4 formed a separate cluster supported by bootstrap values ranging from 97% to 99% for NJ and from 74% to 75% for MP analyses. Haplotypes 5–8 formed a cluster that was supported by bootstrap values ranging from 92% to 94% for NJ and MP analyses. Haplotype 5 formed a separate cluster in NJ analyses, supported by bootstrap values ranging from 87% to 97% in NJ and 94% in MP analyses. Haplotypes 6–8 also formed a strongly supported cluster with bootstrap values ranging from 93% to 95% for NJ and from 82 to 83% for MP analyses. A separate haplotype 8 cluster was strongly supported by bootstrap values ranging from 98% to 99% for NJ and MP analyses. Haplotype 9 formed a separate cluster strongly supported by bootstrap values ranging from 97% to 100%.
MtLrRNA
Analysis of partial MtLrRNA sequences resulted in the identification of five C. gattii haplotypes (Fig. 1b, Table 2). The alignment of the five haplotypes is shown in Fig. S2. Four polymorphic nucleotides, which are indicated within brackets, were present among 299 analyzed nucleotides. Haplotype 1 (CTAG) was present in all AFLP6/VGII isolates and in AFLP4/VGI isolates WM179 and E566; haplotype 2 (CTAC) was present in most of the AFLP4/VGI isolates and in AFLP7/VGIV isolates M27055 and WM779; haplotype 3 (CTTC) was present in AFLP7/VGIV isolates B5742 and B5748; haplotype 4 (CGAC) was present in AFLP4/VGI isolates CBS883, CBS919 and CBS7748; and haplotype 5 (GTAA) was present in all AFLP5/VGIII isolates. An overview of the MtLrRNA haplotype of all isolates is given in Table 2. Both NJ and MP analyses positioned haplotype 1 basal to the other haplotypes, but the topology of the tree of haplotypes 1–4 was not well supported. Haplotype 5 formed a separate cluster in NJ and MP analyses with bootstrap values ranging from 80% to 86%.
Combined analysis of mitochondrial data
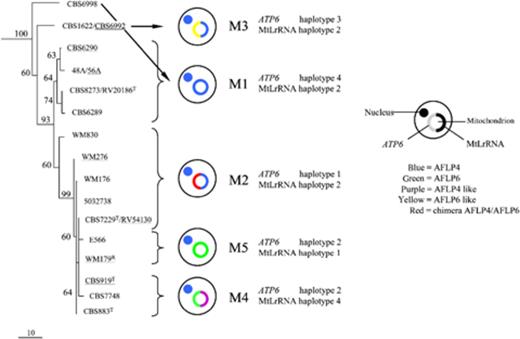
All AFLP6/VGII isolates contained MtLrRNA haplotype 1 and ATP6 haplotype 2, whereas AFLP7/VGIV isolates possessed MtLrRNA haplotypes 2 or 3 that differed by one nucleotide, and ATP6 haplotype 9. The AFLP5/VGIII isolates contained MtLrRNA haplotype 5 and ATP6 haplotypes 5–8 that formed one cluster. AFLP4/VGI isolates possessed MtLrRNA haplotypes 1–4 and ATP6 haplotypes 1–4. Surprisingly, these haplotypes did not cluster together. The AFLP4/VGI isolates could be divided into five mitochondrial genotypes based on the presence of ATP6 and MtLrRNA alleles (viz. M1–M5). Genotype AFLP4/VGI-M1 (MtLrRNA haplotype 2 and ATP6 haplotype 4, i.e. isolates 48A, 56A, CBS6289, CBS6290, CBS6998, CBS 8273, RV 20186) corresponded to the previously described genotypic subgroup AFLP4A (Boekhout, 2001). This genotype formed a clade with AFLP5/VGIII and AFLP7/VGIV isolates in the ATP6 analyses (Fig. 1a) similar to the topology obtained by analyses of six concatenated nuclear regions (see Bovers, 2008b, Fig. 3). Genotype AFLP4/VGI-M1 thus appears to be the core AFLP4/VGI genotype, because they contain AFLP4/VGI-typical alleles of both the nuclear and mitochondrial loci, and in the phylogenetic analysis of the nuclear genes these isolates appeared basal. Genotype M2 of AFLP4/VGI (MtLrRNA haplotype 2 and ATP6 haplotype 1, i.e. isolates 503 2738, CBS7229, RV54130, WM176, WM276 and WM830) possessed the core AFLP4/VGI MtLrRNA sequence. Interestingly, the ATP6 sequence of this genotype, i.e. haplotype 1, was found to be a chimerical sequence in all the isolates studied. This sequence was partially identical to the sequence found in AFLP6/VGII isolates (i.e. h2) and partially identical to the sequence of isolates with a core AFLP4/VGI mitochondrial sequence (i.e. h4, Fig. S3). Genotype AFLP4/VGI-M3 (MtLrRNA haplotype 2 and ATP6 haplotype 3, i.e. isolates CBS1622, CBS6992) possessed the core AFLP4/VGI MtLrRNA sequence and an ATP6 sequence that differed only at one nucleotide position from the ATP6 sequence found in AFLP6/VGII isolates. Genotype AFLP4/VGI-M4 (MtLrRNA haplotype 4 and ATP6 haplotype 2, i.e. isolates CBS883, CBS919, CBS7748) possessed a MtLrRNA sequence that differed one nucleotide from the core AFLP4/VGI MtLrRNA sequence and an ATP6 sequence identical to that of AFLP6/VGII isolates. Genotype AFLP4/VGI-M5 (MtLrRNA haplotype 1 and ATP6 haplotype 2, i.e. isolates E566, WM179 possessed MtLrRNA and ATP6 sequences identical to those found in AFLP6/VGII isolates. Isolate WM179 and isolate E566 that both belong to the mitochondrial genotype M5, have MATα and MATa, respectively. This indicates that the presence of the AFLP6/VGII allele in AFLP4/VGI isolates does not depend on the MAT locus. Thus, the mitochondrial genome of part of the AFLP4/VGI isolates contained sequences that were partially identical to those found in AFLP6/VGII isolates. The incongruence between the mitochondrial genotypes of AFLP4/VGI and the topology of the AFLP4/VGI clade as derived from the analysis of six concatenated nuclear regions (Bovers, 2008b) is illustrated in Fig. 2.
2
Comparison of mitochondrial genotypes and a phylogenetic tree of genotype AFLP4/VGI of Cryptococcus gattii obtained by analysis of six concatenated nuclear regions (RPB1, RPB2, CNLAC1, TEF1α, IGS1 and internal transcribed spacer). Presented is one of the 60 most parsimonious trees (length 847, consistency index 0.902, retention index 0.961) computed with gaps treated as missing data. Data consisted of 3932 characters of which 459 characters were parsimony informative. Bootstrap values (1000 replicates) are indicated. The mitochondrial genotype as well as the ATP6 and MtLrRNA haplotypes are shown. T, type strain; R, molecular type reference strain (Meyer, 2003). Haploid isolates are underlined.
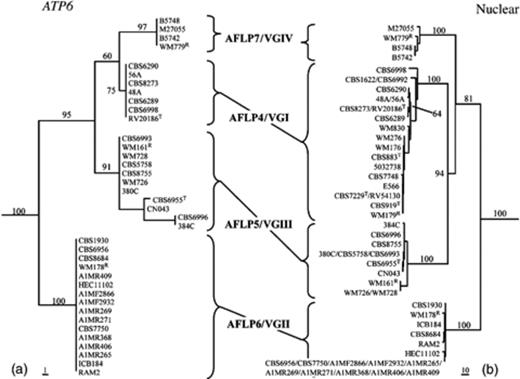
Phylogenetic analyses of partial ATP6 sequences were also carried out without the presence of the AFLP4/VGI isolates that contained sequences that are partially identical to those found in AFLP6/VGII isolates. These AFLP4/VGI isolates were excluded because their presence might obscure the genotypic structure of C. gattii. The topology obtained is depicted in Fig. 3. The cluster that contained isolates with core AFLP4/VGI alleles and AFLP7/VGIV isolates formed a sister group to the AFLP5/VGIII isolates, and this AFLP4/VGI-AFLP7/VGIV-AFLP5/VGIII clade was strongly supported with bootstrap values ranging from 98% to 99% for NJ and from 94% to 95% for MP analyses. The AFLP6/VGII isolates clustered basal to the other C. gattii genotypic groups in both the mitochondrial and nuclear analyses with strong support.
3
Phylogenetic tree of Cryptococcus gattii obtained by analysis of (a) partial ATP6 sequences. AFLP4/VGI isolates that possessed mitochondrial sequences identical to sequences found in AFLP6/VGII isolates were excluded from the analysis. Presented is the most parsimonious tree (length 79, consistency index 0.861, retention index 0.978) computed with gaps treated as missing data. Data consisted of 611 characters of which 64 characters were informative of parsimony; (b) six concatenated nuclear regions (RPB1, RPB2, CNLAC1, TEF1α, IGS1 and internal transcribed spacer). Presented is one of the 60 most parsimonious trees (length 847, consistency index 0.902, retention index 0.961) computed with gaps treated as missing data. Data consisted of 3932 characters of which 459 characters were informative of parsimony. Bootstrap values (1000 replicates) are indicated for the main branches. T, type strain; R, molecular type reference strain (Meyer, 2003).
Discussion
Phylogenetic analyses of partial mitochondrial sequences of C. gattii showed that AFLP6/VGII isolates clustered basal to all other C. gattii genotypic groups. These results correspond to the topology that was found after the analysis of six concatenated nuclear regions (Bovers, 2008b; Fig. 3).
Surprisingly, two AFLP4/VGI isolates (AFLP4/VGI-M5) were found that possessed MtLrRNA and ATP6 sequences identical to those found in AFLP6/VGII isolates. In addition, three AFLP4/VGI isolates (AFLP4/VGI-M4) possessed a MtLrRNA sequence that differed by one nucleotide from the core AFLP4/VGI sequence, but the ATP6 sequence was identical to the sequence found in all AFLP6/VGII isolates. Furthermore, two AFLP4/VGI isolates (AFLP4/VGI-M3) possessed the core AFLP4/VGI MtLrRNA sequence, whereas the ATP6 sequence differed by one nucleotide from the sequence found in AFLP6/VGII isolates. In addition, six AFLP4/VGI isolates (AFLP4/VGI-M2) possessed the core AFLP4/VGI MtLrRNA sequence, but here the ATP6 sequence appeared to be a chimera between core AFLP4/VGI and AFLP6/VGII sequences.
Our results show that AFLP4/VGI isolates exist that possess mitochondrial genomes that consist completely of AFLP6/VGII sequences or that contain a combination of AFLP4/VGI and AFLP6/VGII sequences. It could be argued that the presence of AFLP6/VGII mitochondrial sequences in AFLP4/VGI isolates is a retained ancestral character state, because genotype AFLP6/VGII was found to be the ancestral lineage (Bovers, 2008b). However, in that case AFLP6/VGII mitochondrial sequences would be expected to occur in other genotypic groups of C. gattii as well, especially in AFLP7/VGIV as this genotypic group is most closely related to AFLP6/VGII (Bovers, 2008b). We have to keep in mind, however, that the number of available AFLP7/VGII isolates is small (i.e. only four strains are currently know to belong to this group); thus, it is possible that nucleotide variation in this lineage remains to be detected in the future when more isolates will become available. In the case that ancestral mitochondrial sequences remained preserved in the more derived lineages, viz., AFLP7/VGIV, AFLP5/VGIII and AFLP4/VGI, one would expect that the same or similar sequences would be present in the phylogenetically intermediate genotypes because the patterns of divergence of the various genotypic groups is well supported by a phylogenetic analysis of six nuclear genes (Bovers, 2008b). Moreover, one would expect that some polymorphisms would be present due to ongoing evolution since the initial divergence of the various genotypic groups of C. gattii. In contrast with these suppositions, we observed only AFLP4/VGI isolates that possessed ATP6 sequences identical to those found in AFLP6/VGII isolates. These sequences did not occur in any of the other, phylogenetically intermediate, genotypes, viz., AFLP7/VGIV and AFLP5/VGIII. This suggests that the presence of AFLP6/VGII mitochondrial sequences in AFLP4/VGI isolates is not a retained ancestral character state but, more likely, is a consequence of mitochondrial recombination. The likelihood of mitochondrial recombination is further supported by the presence of the MtLrRNA haplotype 2 allele of AFLP4/VGI in two isolates of AFLP7/VGIV. If this would be the result of retaining ancestral sequences, this process would contradict the well-supported phylogeny based on six nuclear genes in which genotype AFLP4/VGI is more derived than AFLP7/VGIII (Bovers, 2008b).
AFLP4/VGI and AFLP6/VGII isolates have been isolated from the same geographic regions (Sorrell, 1996; Kidd, 2003, 2005, 2007; Campbell, 2005a, b), which indicates that these genotypic groups occur in the same areas and may physically interact. Interestingly, AFLP4/VGI isolates that possessed mitochondrial genomes with (partial) AFLP6/VGII sequences were all isolated from countries near the Pacific Ocean, whereas isolates with core AFLP4/VGI sequences originated from Africa and Europe. Although the number of isolates included in our study is small, this may indicate that the atypical AFLP4/VGI isolates originated in the region of the Pacific Ocean.
If mitochondrial recombination has occurred, one has to assume a process of somatic fusion or mating to occur between cells of the recombining genotypes. To the best of our knowledge, somatic fusion without mating has never been reported in C. neoformans or in C. gattii. Mating usually occurs between isolates of opposite mating type (MATa and MATα) (Kwon-Chung, 1975, 1976a, b), but has also been reported between isolates of identical mating type (Lin, 2005). During mating, the nucleus of the MATα cell migrates into the conjugation tube and the recipient MATa cell generates a hypha (McClelland, 2004). Previous reports indicated that mitochondria in C. neoformans were uniparentally inherited from the MATa parent (Xu, 2000; Yan & Xu, 2003), probably because the MATa cell produces the hypha. However, recent studies have shown that in some cases mitochondrial inheritance from the MATα may occur as well (Yan & Xu, 2003; Toffaletti, 2004; Yan, 2004, 2007a, b) resulting in the presence of MATα parental mitochondria in the progeny. Recent studies on other fungal species have shown that strictly uniparental or biparental mitochondrial inheritance is rare (Yan & Xu, 2005), and it has therefore been suggested to treat mitochondrial inheritance as a quantitative rather than a qualitative trait (Birky, 1995). In cells of the mitochondrial genotype M5, which combine a nuclear genome of the genotype AFLP4/VGI with the presence of the mitochondrial alleles ATP6 and MtLrRNA from AFLP6/VGII, cells of both MATa (i.e. isolate E566) and MATα (i.e. isolate WM179) seem to have functioned as a donor for mitochondrial genes. Therefore, in these cases, the transfer of mitochondria does not seem to be regulated by the mating type. Mitochondrial inheritance following AFLP4/VGI × AFLP6/VGII mating may have resulted in a cell with both types of mitochondria. Subsequent mitotic divisions and mitochondrial selection could have resulted in isolates that possessed a complete AFLP6/VGII mitochondrial genome. In addition, recombination between the two types of mitochondria that were present may have resulted in mitochondrial genomes with both AFLP4/VGI and AFLP6/VGII sequences. It is important to note that recombination between mitochondria has been observed in other fungi under laboratory conditions, for example Agaricus bisporus (De la Bastide & Horgen, 2003), Agrocybe aegerita (Barroso & Labarère, 1997), Neurospora intermedia (Yang & Griffiths, 1992), Saccharomyces cerevisiae (Nakagawa, 1992), as well as in natural populations, for example A. aegerita (Barroso, 1995), Armillaria gallica (Saville, 1998), Candida albicans (Anderson, 2001; Jacobsen, 2008) and Neurospora crassa (Taylor, 1986). In addition, mitochondrial recombination has been observed in laboratory crossings of C. neoformans isolates (Toffaletti, 2004; Yan, 2007b).
Here we suggest the occurrence of mitochondrial recombination among environmental, veterinary and clinical isolates of C. gattii. Recombination between two mitochondrial genomes from different genotypes is only possible after cell–cell conjugation occurs. However, in that case one might expect the occurrence of hybrid nuclear genomes within the populations, and this is not the case with the loci that we have studied (Bovers, 2008b). Hybrid isolates between the two C. neoformans varieties and between C. neoformans and C. gattii have been described (Tanaka, 1999; Cogliati, 2000; Boekhout, 2001; Bovers, 2006, 2008c), as has DNA exchange between serotype A and serotype D isolates of C. neoformans (Kavanaugh, 2006). The resulting hybrid isolates are diploid or aneuploid (Tanaka, 1999; Cogliati, 2000; Lengeler, 2001; Bovers, 2006, 2008c) and possess alleles from both parents. However, all AFLP4/VGI isolates that had been studied using flow cytometry were found to be haploid (M. Bovers & H. Hoogveld, unpublished data) and nuclear AFLP6/VGII sequences have not been found in these AFLP4/VGI isolates (Bovers, 2008b), which indicates that the isolates, most likely, are not hybrids. Possibly, mating between different C. gattii genotypic groups does not result in the formation of hybrid nuclear genomes, but may lead to transfer of mitochondria and subsequent mitochondrial recombination. Moreover, the predominant uniparental inheritance of mitochondria as well as the small size of the mitochondrial genome increases the chance of incorporation of foreign alleles by drift and selection (Martinsen, 2001; Funk & Omland, 2003; Ballard & Whitlock, 2004). The predominant uniparental inheritance and the haploid nature of mitochondria both decrease the effective population size and thereby increase the chance of fixation by drift (Funk & Omland, 2003; Ballard & Whitlock, 2004). In addition, the small genome size decreases the probability of counter selection because negative gene interactions are less likely to occur (Martinsen, 2001; Ballard & Whitlock, 2004). These processes could explain why mitochondrial regions, but not nuclear regions, were incorporated in a different genetic background. Another possibility is that smaller regions of the AFLP6/VGII genome have been incorporated in parts of the nuclear genome of AFLP4/VGI isolates, but that these have escaped detection.
Mitochondria play a central role in energy production and in responses to stresses (Osiewacz & Kimpel, 1999; Ahkter, 2003). These processes play a key role in the fitness of an organism, and the mitochondrial genotype may therefore influence fitness as well. A different mitochondrial genotype may have a positive effect on fitness, as was observed in S. cerevisiae when mitochondria of wine yeasts were transferred to a laboratory yeast, resulting in an increased viability and increased ethanol and temperature tolerance (Jiménez & Benítez, 1988). However, the mitochondrial genotype does not always affect fitness, and no difference in virulence or high-temperature growth rate could be found for strains with mitochondria derived from both C. neoformans varieties (Toffaletti, 2004). In addition, the mitochondrial genotype may also have a negative effect on fitness. Several mitochondrial proteins are produced in the nucleus (Osiewacz & Kimpel, 1999; Burton, 2006) and these nuclear encoded proteins interact with mitochondrial encoded proteins, for example in the electron transport system. The presence of a mitochondrial genotype different from the nuclear genotype may therefore negatively affect fitness as both genomes have coevolved (Burton, 2006). Cryptococcus gattii isolates with different mitochondrial genotypes should be further studied to investigate whether the mitochondrial genotype affects their fitness.
Our results indicate that conjugation may have occurred between AFLP4/VGI and isolates of both AFLP6/VGII and AFLP7/VGIV leading to the generation of cells with an AFLP4/VGI nuclear genotype and a partial AFLP6/VGII or AFLP7/VGIV mitochondrial genotype. Our study is the first to report on mitochondrial inheritance in C. gattii and provides evidence for mitochondrial recombination occurring in nature. Furthermore, the high percentage of AFLP4/VGI isolates studied that possessed a recombined mitochondrial genome (65%) indicates that recombination occurs frequently in nature. Recently, recombination has been demonstrated to occur in natural populations of genotype AFLP4/VGI of C. gattii using AFLP analysis (Saul, 2008). Future research is needed to elucidate the genetic mechanism of the intergenotypic transfer of mitochondria in C. gattii and the effect this has on phenotypic properties. The occurrence of mitochondrial recombination between isolates of genotypes AFLP4/VGI and AFLP6/VGII may also be important, because strains of the latter are involved in the ongoing Vancouver Island outbreak of C. gattii (Kidd, 2004; MacDougall, 2007). Plasmogamy between cells belonging to different genotypes may allow the exchange and introgression of genetic elements in either genetic background, as has also been observed between isolates of serotype A and serotype D of C. neoformans (Kavanaugh, 2006), and such processes may contribute to the formation of C. gattii strains that differ in virulence. It is likely that such genetic transfer events may have an effect on the virulence properties, and this alternative hypothesis concurs with the same sex mating hypothesis (Fraser, 2005; Lin, 2005) that has been proposed to explain the origin of hypervirulence of isolates involved in the ongoing Vancouver Island outbreak of C. gattii.
Acknowledgements
Isolates were kindly donated by the following researchers: J.W. Kronstad, K.J. Kwon-Chung, M.S. Lazera, D. Swinne, W. Meyer and J.M. Torres-Rodríguez. We would like to thank Cañan Bayrakdar and Hans Hoogveld for technical assistance. Work of M.B. was supported by the ‘Odo van Vloten fonds’. E.E.K. and F.H. were funded by the Strategy Fund of the Royal Netherlands Academy of Arts and Sciences (RNAAS-KNAW).
Authors' contributions
M.B. and F.H. equally contributed to the work, M.B. and F.H. performed the work, T.B. designed the research, and M.B., F.H., E.E.K. and T.B. wrote the paper.
Supporting Information
Additional Supporting Information may be found in the online version of this article:
Fig. S1. Alignment of Cryptococcus gattii partial ATP6 sequences.
Fig. S2. Alignment of Cryptococcus gattii partial MtLrRNA sequences.
Fig. S3. Alignment of Cryptococcus gattii partial ATP6 sequences for haplotypes 1, 2 and 4.
Please note: Wiley-Blackwell is not responsible for the content or functionality of any supporting information supplied by the authors. Any queries (other than missing material) should be directed to the corresponding author for the article.
References
(
2003
)
Role of alternative oxidase gene in pathogenesis of Cryptococcus neoformans
.
Infect Immun
71
:
5794
–
5802
.
(
2001
)
mtDNA recombination in a natural population
.
J Bacteriol
183
:
865
–
872
.
(
2004
)
The incomplete natural history of mitochondria
.
Mol Ecol
13
:
729
–
744
.
(
2004
)
Cryptococcus neoformans shows a remarkable genotypic diversity in Brazil
.
J Clin Microbiol
42
:
1356
–
1359
.
(
1997
)
Genetic evidence for nonrandom sorting of mitochondria in the basidiomycete Agrocybe aegerita
.
Appl Environ Microbiol
63
:
4686
–
4691
.
(
1995
)
Wide distribution of mitochondrial genome rearrangements in wild strains of the cultivated basidiomycete Agrocybe aegerita
.
Appl Environ Microbiol
61
:
1187
–
1193
.
(
1995
)
Uniparental inheritance of mitochondrial and chloroplast genes: mechanisms and evolution
.
P Natl Acad Sci USA
92
:
11331
–
11338
.
(
2003
)
Molecular analysis of Cryptococcus neoformans mitochondrial cytochrome b gene sequences
.
J Clin Microbiol
41
:
5572
–
5576
.
(
1997
)
Molecular typing of Cryptococcus neoformans: taxonomic and epidemiological aspects
.
Int J Syst Bacteriol
47
:
432
–
442
.
(
2001
)
Hybrid genotypes in the pathogenic yeast Cryptococcus neoformans
.
Microbiology
147
:
891
–
907
.
(
2001
)
Rapid methods to extract DNA and RNA from Cryptococcus neoformans
.
FEMS Yeast Res
1
:
221
–
224
.
(
2006
)
Unique hybrids between fungal pathogens Cryptococcus neoformans and Cryptococcus gattii
.
FEMS Yeast Res
6
:
599
–
607
.
(
2008a
)
Diversity of the Cryptococcus neoformans–Cryptococcus gattii species complex
.
Rev Iberoam Mycol
25
:
S4
–
S12
.
(
2008b
)
Six monophyletic lineages identified within Cryptococcus neoformans and Cryptococcus gattii by multi-locus sequence typing
.
Fungal Genet Biol
45
:
400
–
421
.
(
2008c
)
AIDS patient death caused by novel Cryptococcus neoformans×C. gattii hybrid
.
Emerg Infect Dis
14
:
1105
–
1108
.
(
2006
)
The sorry state of F2 hybrids: consequences of rapid mitochondrial DNA evolution in allopatric populations
.
Am Nat
168
:
S14
–
S24
.
(
2005
)
The PRP8 inteins in Cryptococcus are a source of phylogenetic and epidemiological information
.
Fungal Genet Biol
42
:
452
–
463
.
(
1998
)
First isolation of Cryptococcus neoformans var. gattii, serotype C, from the environment in Colombia
.
Med Mycol
36
:
341
–
344
.
(
2005a
)
Clonality and recombination in genetically differentiated subgroups of Cryptococcus gattii
.
Eukaryot Cell
4
:
1403
–
1409
.
(
2005b
)
Clinical and environmental isolates of Cryptococcus gattiifrom Australia retain sexual fecundity
.
Eukaryot Cell
4
:
1410
–
1419
.
(
2002
)
Molecular genetic analyses of mating pheromones reveal intervariety mating or hybridization in Cryptococcus neoformans
.
Infect Immun
70
:
5225
–
5235
.
(
2000
)
Sequence analysis and ploidy determination of Cryptococcus neoformans var. neoformans
.
J Mycol Med
10
:
171
–
176
.
(
2003
)
Mitochondrial inheritance and the detection of non-parental mitochondrial DNA haplotypes in crosses of Agaricus bisporus homokaryons
.
Fungal Genet Biol
38
:
333
–
342
.
(
2005
)
Use of a suspension array for rapid identification of the varieties and genotypes of the Cryptococcus neoformans species complex
.
J Clin Microbiol
43
:
3662
–
3672
.
(
2000
)
Molecular sequence analyses of the intergenic spacer (IGS) associated with rDNA of the two varieties of the pathogenic yeast, Cryptococcus neoformans
.
Syst Appl Microbiol
23
:
535
–
545
.
(
2005
)
Comparative analysis of the intergenic spacer regions and population structure of the species complex of the pathogenic yeast Cryptococcus neoformans
.
FEMS Yeast Res
5
:
1129
–
1140
.
(
2000
)
Epidemiology: surveillance of fungal infections
.
Med Mycol
38
:
173
–
182
.
(
1990
)
Natural habitat of Cryptococcus neoformans var. gattii
.
J Clin Microbiol
28
:
1642
–
1644
.
(
2006
)
Molecular epidemiology of clinical and environmental isolates of the Cryptococcus neoformans species complex reveals a high genetic diversity and the presence of the molecular type VGII mating type a in Colombia
.
FEMS Yeast Res
6
:
625
–
635
.
(
2000
)
Biodiversity and systematics of basidiomycetous yeasts as determined by large-subunit rDNA D1/D2 domain sequence analysis
.
Int J Syst Evol Microbiol
50
:
1351
–
1371
.
(
2001
)
First isolation of Cryptococcus neoformans var. gattii from a native jungle tree in the Brazilian Amazon rainforest
.
Mycoses
44
:
137
–
140
.
(
1999
)
Cryptococcus neoformans var. grubii: separate varietal status for Cryptococcus neoformans serotype A isolates
.
J Clin Microbiol
37
:
838
–
840
.
(
2003
)
Recapitulation of the sexual cycle of the primary fungal pathogen Cryptococcus neoformans var. gattii: implications for an outbreak on Vancouver Island, Canada
.
Eukaryot Cell
2
:
1036
–
1045
.
et al.
(
2005
)
Same-sex mating and the origin of the Vancouver Island Cryptococcus gattii outbreak
.
Nature
437
:
1360
–
1364
.
(
2003
)
Species-level paraphyly and polyphyly: frequency, causes and consequences, with insights from animal mitochondrial DNA
.
Annu Rev Ecol Evol S
34
:
397
–
423
.
(
1970
)
An atypical strain of Cryptococcus neoformans (Sanfelice) Vuillemin 1894. Part 1. Description of the disease and of the strain
.
Ann Soc Belg Med Tr
50
:
689
–
693
.
(
2005
)
Isolation and characterization of Cryptococcus neoformans varieties recovered from natural sources in Bogotá, Colombia and study of ecological conditions in the area
.
Microbial Ecol
49
:
282
–
290
.
(
2000
)
A molecular study of mating type and recombination in Cryptococcus neoformans var. gattii. PhD Thesis
,
University of Sydney
,
Sydney, Australia
.
(
1993
)
An empirical test of bootstrapping as a method for assessing confidence in phylogenetic analysis
.
Syst Biol
42
:
182
–
192
.
(
2004
)
Cryptococcus neoformans infections at Vancouver Hospital and Health Sciences Centre (1997–2002): epidemiology, microbiology and histopathology
.
J Med Microbiol
53
:
935
–
940
.
(
2008
)
Mitochondrial haplotypes and recombination in Candida albicans
.
Med Mycol
46
:
647
–
654
.
(
1988
)
Yeast cell viability under conditions of high temperature and ethanol concentrations depends on the mitochondrial genome
.
Curr Genet
13
:
461
–
469
.
(
2004
)
The internal transcribed spacers and 5.8S rRNA gene show extensive diversity among isolates of the Cryptococcus neoformans species complex
.
FEMS Yeast Res
4
:
377
–
388
.
(
2006
)
Recent evolution of the human pathogen Cryptococcus neoformans by intervarietal transfer of a 14 gene fragment
.
Mol Biol Evol
23
:
1879
–
1890
.
(
2003
)
Isolation of two molecular types of Cryptococcus neoformans var. gattii from insect frass
.
Med Mycol
41
:
171
–
176
.
(
2004
)
A rare genotype of Cryptococcus gattii caused the cryptococcosis outbreak on Vancouver Island (British Colombia, Canada)
.
P Natl Acad Sci USA
101
:
17258
–
17263
.
(
2005
)
Comparative gene genealogies indicate that two clonal lineages of Cryptococcus gattii in British Columbia resemble strains from other geographical areas
.
Eukaryot Cell
4
:
1629
–
1638
.
(
2007
)
Characterization of environmental sources of the human and animal pathogen Cryptococcus gattii in British Columbia, Canada, and Pacific Northwest USA
.
Appl Environ Microbiol
73
:
1433
–
1443
.
(
2002
)
Cryptococcus neoformans in the koala (Phascolarctos cinereus): colonization by C. neoformans var. gattii and investigation of environmental sources
.
Med Mycol
40
:
263
–
272
.
(
1975
)
A new genus, Filobasidiella, the perfect state of Cryptococcus neoformans
.
Mycologia
67
:
1197
–
1200
.
(
1976a
)
Morphogenesis of Filobasidiella neoformans, the sexual state of Cryptococcus neoformans
.
Mycologia
68
:
821
–
833
.
(
1976b
)
A new species of Filobasidiella, the sexual state of Cryptococcus neoformans B and C serotypes
.
Mycologia
68
:
942
–
946
.
(
1984
)
Epidemiological differences between the two varieties of Cryptococcus neoformans
.
Am J Epidemiol
120
:
123
–
130
.
(
2006
)
Do major species concepts support one, two or more species within Cryptococcus neoformans?
FEMS Yeast Res
6
:
574
–
587
.
(
1982
)
Taxonomic studies on Filobasidiella species and their anamorphs
.
Antonie van Leeuwenhoek
48
:
25
–
38
.
(
1992
)
Genetic association of mating types and virulence in Cryptococcus neoformans
.
Infect Immun
60
:
602
–
605
.
(
2002
)
Proposal to conserve the name Cryptococcus gattii against C. hondurianus and C. bacillisporus (Basidiomycota, Hymenomycetes, Tremellomycetidae)
.
Taxon
51
:
804
–
806
.
(
2002
)
Isolation and characterisation of the phospholipase B gene of Cryptococcus neoformans var. gattii
.
FEMS Yeast Res
2
:
551
–
561
.
(
2003
)
Isolation and characterisation of the phospholipase B gene of Cryptococcus neoformans var. gattii
.
Appl Environ Microbiol
69
:
2080
–
2086
.
(
1998
)
Cryptococcus neoformans var. gattii– evidence for a natural habitat related to decaying wood in a pottery tree hollow
.
Med Mycol
36
:
119
–
122
.
(
2000
)
Possible primary ecological niche of Cryptococcus neoformans
.
Med Mycol
38
:
379
–
383
.
(
2001
)
Serotype AD strains of Cryptococcus neoformans are diploid or aneuploid and are heterozygous at the mating-type locus
.
Infect Immun
69
:
115
–
122
.
(
2002
)
Identification of the MATa mating-type locus of Cryptococcus neoformans reveals a serotype A MATa strain thought to have been extinct
.
P Natl Acad Sci USA
97
:
14455
–
14460
.
(
2005
)
Sexual reproduction between partners of the same mating type in Cryptococcus neoformans
.
Nature
434
:
1017
–
1021
.
(
2006
)
Multilocus sequence typing reveals three genetic subpopulations of Cryptococcus neoformans var. grubii (serotype A), including a unique population in Botswana
.
Genetics
172
:
2223
–
2238
.
(
2007
)
Spread of Cryptococcus gattii in British Columbia, Canada, and detection in the Pacific Northwest, USA
.
Emerg Infect Dis
13
:
42
–
50
.
(
2001
)
Hybrid populations selectively filter gene introgression between species
.
Evolution
55
:
1325
–
1335
.
(
2004
)
Uniqueness of the mating system in Cryptococcus neoformans
.
Trends Microbiol
12
:
208
–
212
.
and the IberoAmerican Cryptococcal, study, group
(
2003
)
Molecular typing of IberoAmerican Cryptococcus neoformans isolates
.
Emerg Infect Dis
9
:
189
–
195
.
(
1992
)
An endonuclease with multiple cutting sites, Endo.SceI, initiates genetic recombination at its cutting site in yeast mitochondria
.
EMBO J
11
:
2707
–
2715
.
(
1999
)
Mitochondrial-nuclear interactions and lifespan control in fungi
.
Exp Gerontol
34
:
901
–
909
.
(
2003
)
Decayed wood of Syzygium cumini and Ficus religiosa living trees in Delhi/New Delhi metropolitan area as natural habitat of Cryptococcus neoformans
.
Med Mycol
41
:
199
–
209
.
(
1996
)
Characterization of Cryptococcus neoformans by random DNA amplification
.
Lett Appl Microbiol
23
:
312
–
316
.
(
2008
)
Evidence of recombination in mixed-mating-type and alpha-only populations of Cryptococcus gattii sourced from single eucalyptus tree hollows
.
Eukaryot Cell
7
:
727
–
734
.
(
1998
)
mtDNA recombination in a natural population
.
P Natl Acad Sci USA
95
:
1331
–
1335
.
(
1996
)
Natural environmental sources of Cryptococcus neoformans var. gattii
.
J Clin Microbiol
34
:
1261
–
1263
.
(
1995
)
Clinical and host differences between infections with the two varieties of Cryptococcus neoformans
.
Clin Infect Dis
21
:
28
–
34
.
(
2002
)
Multispecies outbreak of cryptococcosis on southern Vancouver Island, British Columbia
.
Can Vet J
43
:
792
–
794
.
(
2000
)
PAUP* 4.0: Phylogenetic Analysis Using Parsimony
.
Sinauer Associates
,
Sunderland, MA
.
(
1999
)
Ploidy of serotype AD strains of Cryptococcus neoformans
.
Jpn J Med Mycol
40
:
31
–
34
.
(
1986
)
Evolution and mitochondrial DNA in Neurospora crassa
.
Evolution
40
:
716
–
739
.
(
1997
)
The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools
.
Nucleic Acids Res
24
:
4876
–
4882
.
(
2004
)
Cryptococcus neoformans mitochondrial genomes from serotype A and D strains do not influence virulence
.
Curr Genet
46
:
193
–
204
.
(
2007
)
First contemporary case of human infection with Cryptococcus gattii in Puget Sound: evidence for spread of the Vancouver Island outbreak
.
J Clin Microbiol
45
:
3086
–
3088
.
and European Confederation of medical Mycology.
(
2006
)
Cryptococcus neoformans isolates from a 30-month ECMM survey of cryptococcosis in Europe
.
FEMS Yeast Res
6
:
614
–
619
.
(
1990
)
Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics
.
PCR Protocols: A Guide to Methods and Applications
(
,
,
, eds), pp.
315
–
322
.
Academic Press
,
San Diego
.
(
2000
)
Uniparental mitochondrial transmission in sexual crosses in Cryptococcus neoformans
.
Curr Microbiol
40
:
269
–
273
.
(
2003
)
Mitochondria are inherited from the MATa parent in crosses of the basidiomycete fungus Cryptococcus neoformans
.
Genetics
163
:
1315
–
1325
.
(
2005
)
Fungal mitochondrial inheritance and evolution
.
Evolutionary Genetics of Fungi
(
, ed), pp.
221
–
252
.
Horizon Bioscience
,
Norfolk, UK
.
(
2004
)
SXI1α controls uniparental mitochondrial inheritance in Cryptococcus neoformans
.
Curr Biol
14
:
743
–
744
.
(
2007a
)
The mating type-specific homeodomain genes SXI1α and SXI2a coordinately control uniparental mitochondrial inheritance in Cryptococcus neoformans
.
Curr Genet
51
:
187
–
195
.
(
2007b
)
Environment factors can influence mitochondrial inheritance in the fungus Cryptococcus neoformans
.
Fungal Genet Biol
44
:
315
–
322
.
(
1992
)
Male transmission of linear plasmids and mitochondrial DNA in the fungus Neurospora
.
Genetics
134
:
1055
–
1062
.
Author notes
© 2009 Federation of European Microbiological Societies. Published by Blackwell Publishing Ltd. All rights reserved
PDF